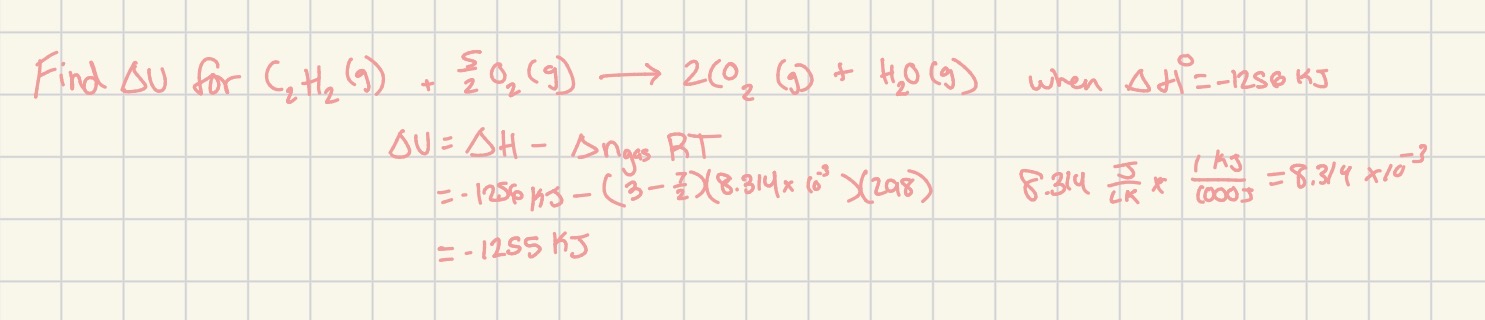chem quiz 4
1/3
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
What are the 4 ways to calculate enthelpy?
Calorimeter
Hess's law
Heats of formation
Bond energy
What is heSs's law?
you're given multiple equations that need to add up to the overall equation
Manipulate the supplemental equations and supplemental enthalpies when needed
Add up new enthalpies to get the overall enthalpy
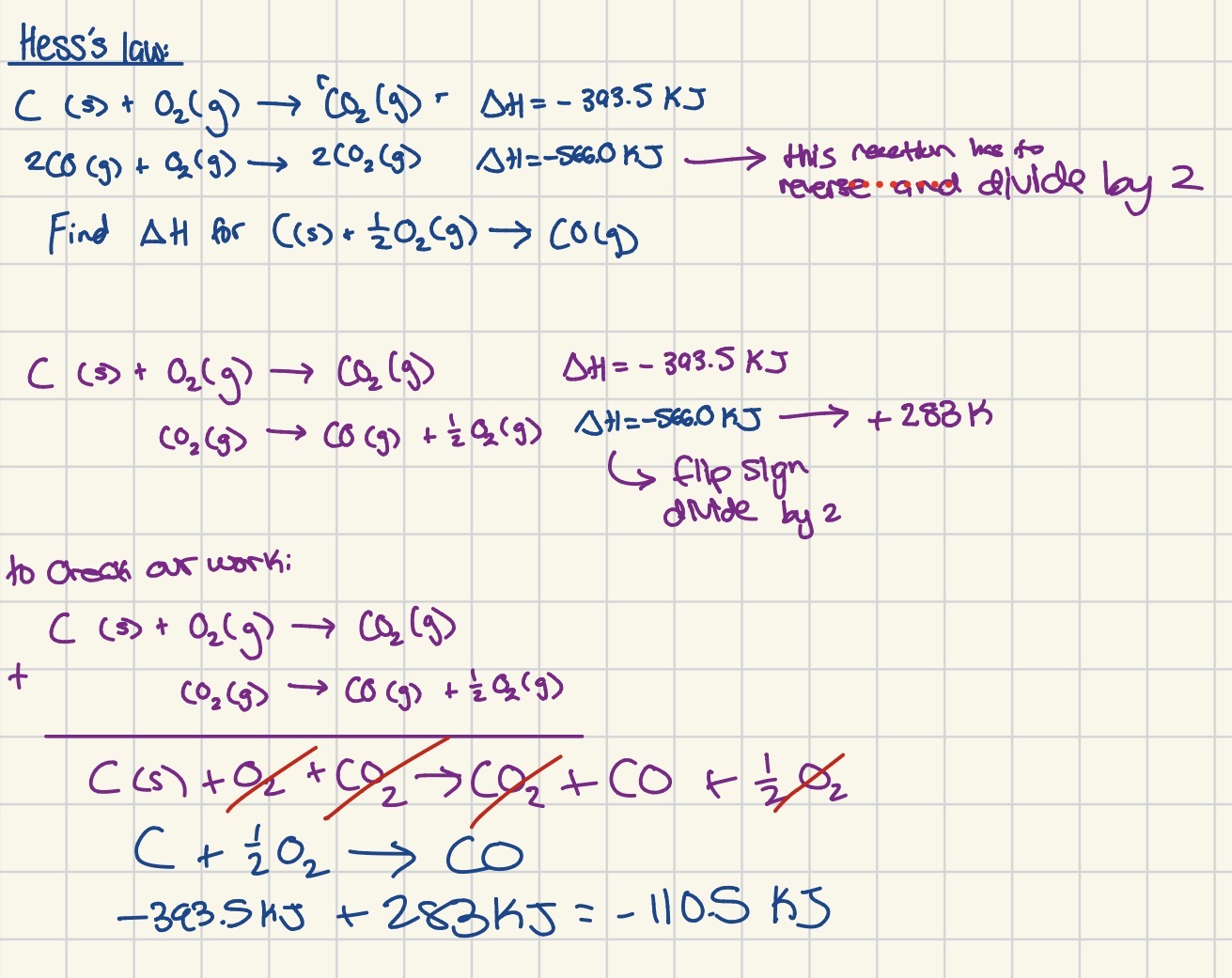
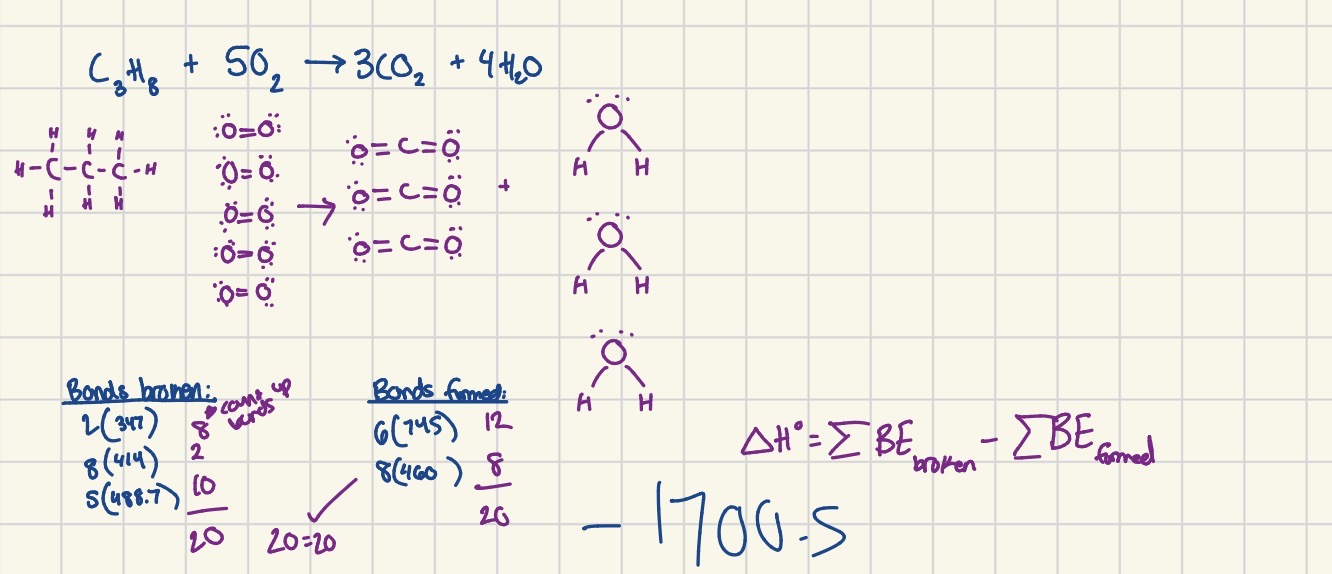
What is enthalpy (heat of formation)?
The energy associated with the formation of one mole of a compound from its elements at standard state (1 ATM and 298K)
Delta H* rxn = (sum of delta H products) - (sum of delta H reactants)
What is the first law of thermodynamics?
Work done on system means W > 0
Work done by system means W < 0
Change in u (internal system energy)= (delta H) - (change in n gas)RT
Where change in n gas = (sum of product coefficients) - (sum of reactant coefficients)