The Atom - Chp 2
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
Diffusion
the spreading of particles from a region of high concentration to a region of low concentration. The movement of these particles provides evidence for the existence of atoms
Describe an experiment which demonstrates that matter is made from particles
one piece of cotton wool soaked in ammonia (NH₃) and one piece of cotton wool soaked in hydrochloric acid (HCl), both put into a glass pipe at opposite ends, at the same time
a white cloud of ammonium chloride (NH₄Cl) forms closer to the HCl end
the HCl is denser than NH₃, and moves more slowly which is why the white cloud is closer to that end
since the substances did not come into direct contact with eachother but a new substance was formed, this proves that the particles must have diffused through the glass pipe and then reacted with each other
What did the Greek Philosophers do?
around 400BC, they were the first to propose that matter was composed of small particles, they believed that they were indivisible, the greek word atomos gave us the English word atom
What did Dalton do?
he was the first to put forward idea of an atom
Dalton's Atomic Theory
- All matter is made up of very small particles
- All atoms are indivisible, they cannot be broken down into simpler particles
What did Crookes do?
he discovered cathode rays
What did Thompson do?
discovered electron
discovered charge/mass ratio of an electron
put forward the plum pudding model of an atom
What did Rutherford do?
discovered the nucleus
discovered the proton
proposed new structure of the atom, consisting of a nucleus and electron cloud
Millikan
- discovered the charge and mass of an electron
- disproved the plum pudding model
Describe Crookes's cathode ray experiment - diagram
Crookes passed electric current through air at low pressure
the glass tube used is called a vacuum tube
rays travelled from the negative electrode and to the positive electrode
the rays caused the glass to fluoresce and a shadow of a Maltese cross was formed at the other end of the tube
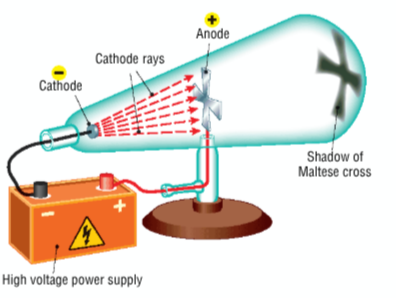
Conclusion of Crookes's cathode ray experiment
found that rays were produced from the cathode, which he called cathode rays
shadow proves that they travel in straight lines
did not realise that these rays were streams of negatively charged particles called electrons
How did Crookes investigate the properties of cathode rays?
he mounted a light paddle wheel on rails in front of the cathode
the current was switched on, the paddle wheel rotated away from the cathode and travelled down the tube, and concluded that the vanes were being struck by particles coming from the cathode
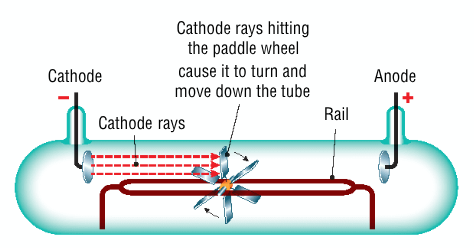
What are the properties of cathode rays?
they travel in straight lines
causes glass to fluoresce
they possess enough energy to move a paddle wheel
Describe how Thomson modified Crookes's experiment - diagram
passed the cathode rays through a doughnut shaped anode, to get a focused beam
the cathode beam passed between two parallel metal plates, and struck a fluorescent screen at the end of the tube
when the plates were charged, the cathode beam was deflected towards the positive plate
Thomson concluded that cathode rays were made up of negative particles called electrons (George Stoney)
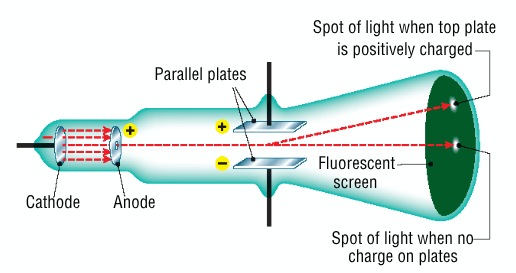
Conclusion of Thomson's modified experiment
focused cathode rays were attracted towards the positively charged plate, meaning that the cathode rays were negatively charged
How did Thomson work out the charge/mass ratio?
- placed a large electromagnet outside the glass tube and adjusted the strength of the magnetic field
- electrons were also deflected by this and adjusted the magnet until it was back down to its original position
- from this he calculated the e/m ratio
What is charge measured in?
Coulombs
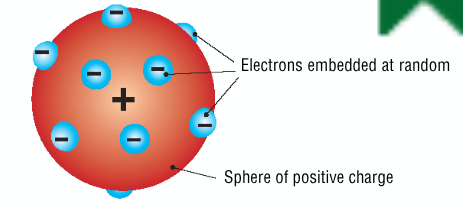
Describe + draw Thompsons plum pudding model
described as a sphere of positive charge with negative electrons embedded in it at random
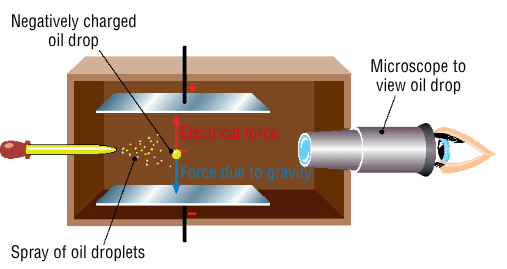
Describe Millikan's oil drop experiment
sprayed oil droplets between two charged plates
the air was ionised using x-ray
electrons lost in the air picked up by oil droplets
adjusted charge until the droplets were suspended in mid air
the electrical force pulling the droplet up was balanced by the weight of the droplet due to gravity, using this he was able to calculate the charge of an electron
using Thomson's charge/mass ratio he was then able to calculate the mass of an electron
Millikan's oil drop experiment - diagram
Describe Rutherford's experiment which proved Thomsons model of the atom wrong - diagram
he bombarded a thin piece of gold foil with alpha particles
used a phosphorescent screen made of zinc sulfide to detect the alpha particles
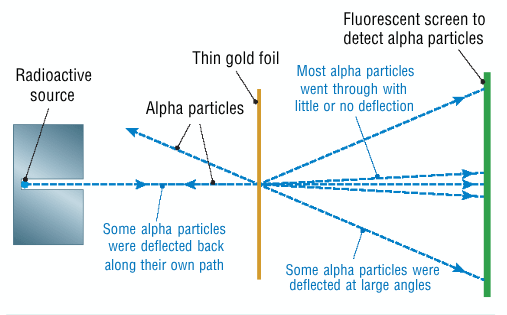
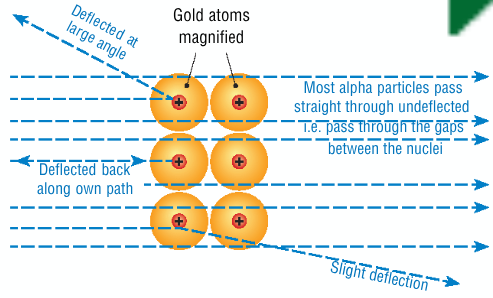
What 3 things were observed in Rutherford's experiment + what are the implications of these observations
most particles go through the gold, atom is mainly empty space
some particles are deflected through wide angles when it gets too close to the nucleus of a gold atom, nucleus has a positive charge
very rarely a few particles will bounce straight back, meaning that an atoms mass is located in a small space
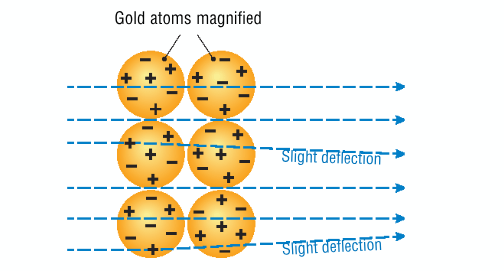
How would Rutherford’s experiment have been different if the plum pudding model was accurate?
the alpha particles would only have been slightly deflected and most would have gone straight through
Why did Rutherford’s experiment only work for heavier atoms?
- the alpha particles were breaking up the nuclei of the lighter particles
- the alpha particles were repelled by the heavier atoms before they reached their nucleus
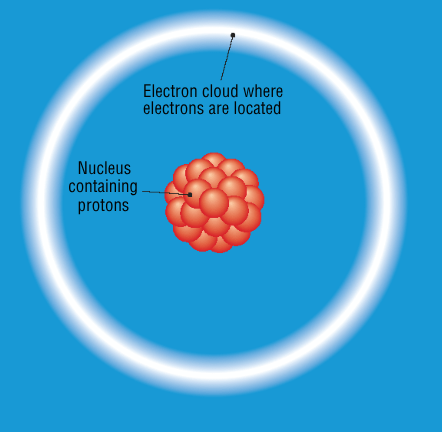
What was the new model for an atom proposed by Rutherford?
consisted of a nucleus containing protons and an electron cloud surrounding nucleus
What did Chadwick do?
- discovered the neutron
Describe Chadwick's experiment to discover the neutron
he fired alpha particles at beryllium
neutrons were knocked out of the nucleus of the beryllium atoms
the neutrons were difficult to detect due to their neutral charge
the neutrons knocked protons out of the paraffin wax, which was easy to detect the positive charge
he found that these particles had about the same mass as the proton, and named them neutrons as they were neutral
What unit measures the mass of sub-atomic particles?
atomic mass units, A.M.U.
Electron: mass + charge
charge: -1
mass: 1/1840 amu
Proton: mass + charge
charge: +1
mass: 1
Neutron: charge + mass
charge: 0
mass: 1
What makes neutrons such a significant discovery?
- brought about the development of nuclear fission
- if there were no neutrons in the nucleus it would collapse because the repulsive forces of the protons repel eachother
Cathode rays
- streams of negatively charge particles called electrons
- they travel in straight lines from the cathode to the anode,
- are deflected by electric and magnetic fields
- and have sufficient energy to move a small object such as a paddle wheel