Chapter 4 PETE 314
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
The first law of thermodynamics resulted from a study of ______.
Friction Heating
This study lead to the definition of an absteact quantity called ________
Energy
T/F Energy follows the balance principle, with neither creation nor destruction
True
All energy quantities are measured ____________
Relative to some arbitrary datum
The first law of thermodynamics is ________________; it rests on the ability to predict the outcome of all the expereriments run to test it.
not derivable or provable
The first law of thermodynamics can be used to _____________
Solve an enourmous number of problems
The first law of thermodynamics says nothing about _____________. That is covered by the second law of thermodynamics.
the direction of an energy change
The first law of thermodynamics _________
States that energy cannot be created nor destroyed, it can only be converted from one form to another
Energy is always measured as a _______ quantity.
Relative
By the early 1800’s, scientists knew that a moving body possessed what we would call ____________
kinetic energy
If an object came to rest by sliding across a surface, it loses its kinetic energy but it and the surface become ________
Hotter
There idea that “there is a quantity called energy that is conserved“ was introduced by _______ , ________ , and __________
Rumford, Joule, Mayer
Examples of absolute quantities
Temperature, Entropy, Length, Mass
What is an absolute quantity?
Quantities for which there is defined a standard unit of measurement and the meaning of a zero amount of the quantity is clear.
What is a relative quantity?
A quantity that is realtive to some conveinent benchmarkE
Examples of relative quantities
Elevation, Velocity
No one has yet found a way to measure or to calculate ________ of energies
absolute values
Consider a 1kg ball of steel, what forms of energy can it possess?
Internal, Potential, Kinetic, Surface, Electrostatic, Magnetic, Nuclear
How many forms of energy exist?
7
A body that is hot possess more ___________ than the same body does when cold
Internal Energy
How can energy be transferred from one body to another?
Touching
Doing work upon one another
Radiation
What is the general balance equation?
Accumulation = Creation - Destruction + Flow in - Flow Out
What is the mass balance equation?
Increase of mass in boundary = Flow in - Flow Out
Derive the energy balance equation

Define the Kinetic Energy term in the energy balance equation
ft * lb / lbm
OR
J/kg
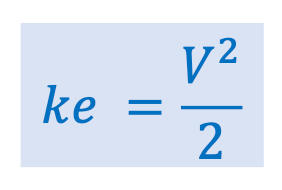
Define the potential energy term for the energy balance equation and its units.
ft * lb / lbm
OR
J/kg
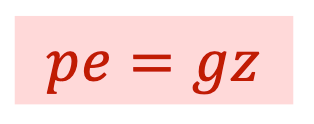
What is Injection work
Define BTU
Define the Cal