biological chemistry
1/107
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
108 Terms
Glycine abbreviations
Gly
G
Alanine abbreviations
Ala
A
Valine abbreviations
Val
V
Leucine abbreviations
Leu
L
Isoleucine abbreviations
Ile
I
Hydrophobic amino acids
Glycine, Alanine, Valine, Leucine, Isoleucine
Cysteine abbreviations
Cys
C
Methionine abbreviations
Met
M
Thiolated amino acids
Cysteine, Methionine
Serine abbreviations
Ser
S
Threonine abbreviations
Thr
T
Tyrosine abbreviations
Tyr
Y
hydroxylated amino acids
Serine, Threonine, Tyrosine
Lysine abbreviations
Lys
K
Arginine abbreviations
Arg
R
Histidine abbreviations
His
H
Amine amino acids
Lysine, Arginine, Histidine
Aspartic acid abbreviations
Asp
D
Asparagine abbreviations
Asn
N
Glutamic acid abbreviations
Glu
E
Glutamine abbreviations
Gln
Q
acidic amino acids
aspartic acid, asparagine, glutamic acid, glutamine
tryptophan abbreviations
Trp
W
Phenylalanine abbreviations
Phe
F
Proline abbreviations
Pro
P
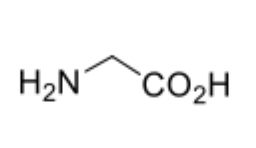
glycine
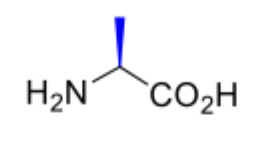
Alanine
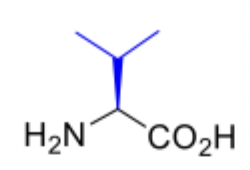
Valine
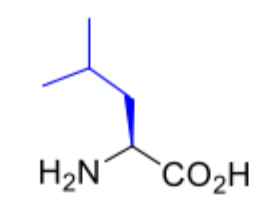
Leucine
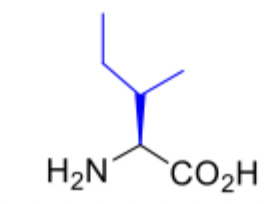
Isoleucine
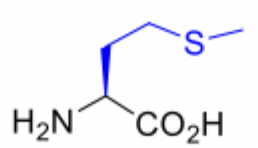
Methionine
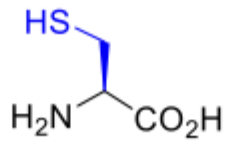
Cysteine
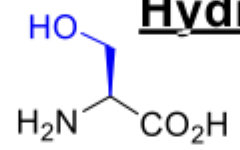
Serine
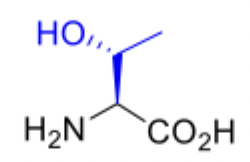
Threonine
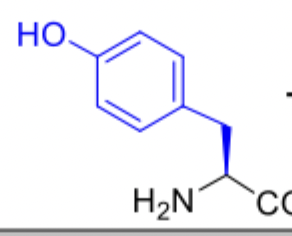
Tyrosine
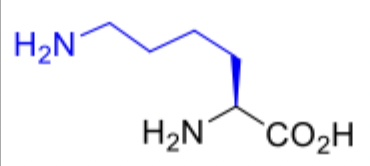
Lysine
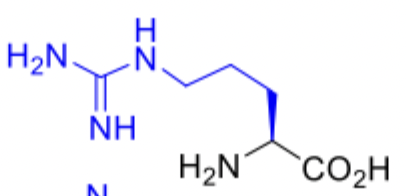
Arginine
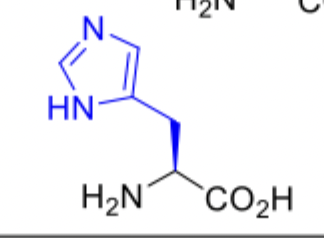
Histidine
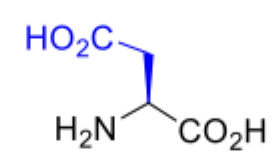
Aspartic Acid
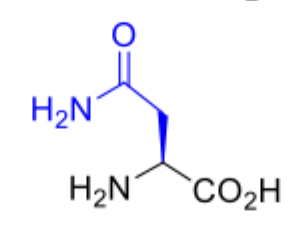
Asparagine
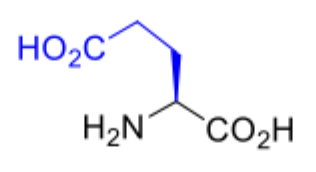
Glutamic Acid
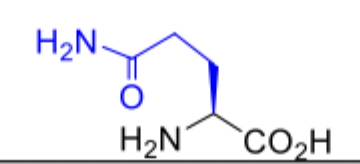
Glutamine
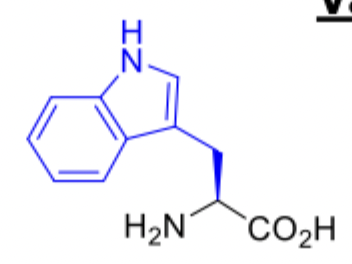
Tryptophan
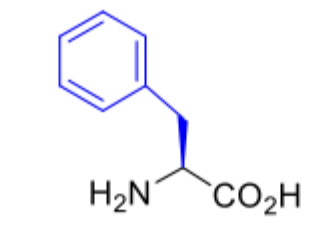
phenylalanine
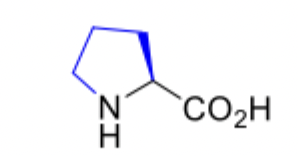
proline
General rules for fischer projections of amino acids (positioning and what determines L/D)
Carboxylate (C1) at the top and side chain (R) at the bottom (highest oxidized carbon goes to the top)
Vertical bonds represent receding bonds
Horizontal (across) bonds represent bonds coming out of the plane
The position of the amine group determines L or D - amine group to the right= D; amine group to the left = L
Fischer projections- how to find the highest oxidized carbon
Carbon bonded to the fewest hydrogens or the most heteroatoms — each bond to an electronegative atom increases OS by 1
Which amino acid has R absolute configuration
Cysteine
What is the stereochemistry of all amino acids (L or D)?
L
What is an isoelectric point
The pH at which an amino acid is neutral
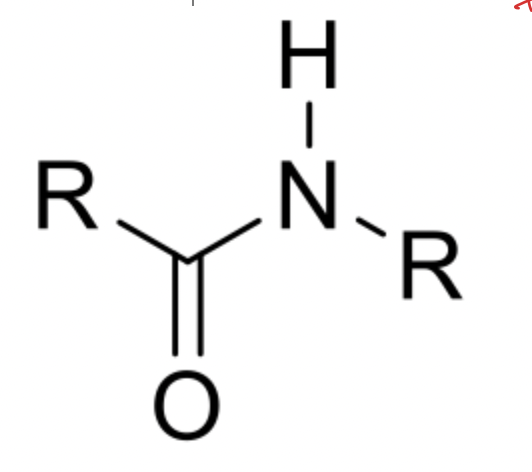
Characteristics of amide bonds
Amide bonds don’t rotate
Amide bonds are planar
What does an amide bond look like? (draw)
Primary structure of a peptide
Linear sequence of amino acids
Secondary structure
Hydrogen bonds between amino acids
Alpha helix
Beta-pleated sheet
Tertiary Structure
How the domains of secondary structure fold relative to each other
Types of bonds that hold together tertiary structure
salt linkages, hydrogen bonds, disulfide bridges/linkages, van der waals interactions, p-p (pi orbital) stacking, hydrophobic effect
Quaternary structure
individual folded peptides come together
What do enzymes do and how?
Catalysts that lower the activation barrier of reaction by reducing both enthalpy and entropy of activation
How do enzymes affect the equilibrium position of a reaction?
They don’t affect it — enzymes don’t alter equilibrium
what are enzymes
biological catalysts made up of a peptide chain folded into a 3d globular protein structure
stereochemistry of enzymes and their reactions
Chiral and carry out reactions on single enantiomers
What is Km?
Equilibrium constant and measure of an affinity of a substrate for an enzyme
KM~ k-1/k1
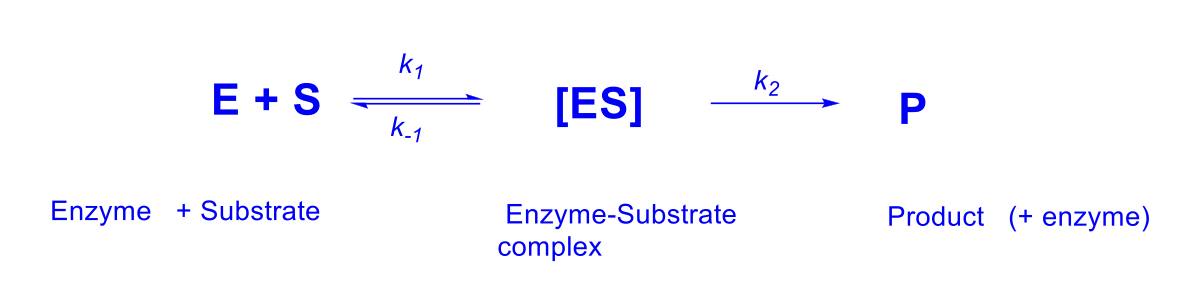
What is Vmax
The maximum rate at saturating the enzyme-substrate complex — approximates k2 at saturating substrate concentrations
What is the difference between saturated and unsaturated fatty acids?
Unsaturated fatty acids have double bonds, where saturated fatty acids do not
what kind of double bonds do unsaturated fatty acids have?
Cis/Z
what are the building blocks of phospholipids
choline, phosphate, glycerol, fatty acids
what does amphiphile mean
compound with a hydrophilic component and a hydrophobic component - polarized - polar heads separate hydrophobic tails from aqueous extracellular and intracellular environment in lipid bilayer
what is cholesterol?
a lipid, steroid - not a fatty acid
what is an aldose
a sugar whose ring-opened form is an aldehyde
what is a ketose
a sugar whose ring opened form is a ketone
what is an anomeric center
The carbon in the center of the hemiacetal on a sugar — the carbon is the carbonyl carbon in the ring opened form
What is the difference between alpha and beta anomers
Alpha anomers - substituents are trans across the cyclic ether (alpha-across)
Beta anomers - substituents are cis across the cyclic ether
Mutarotation of glucose
The change in optical rotation to go to equilibrium value (52.5º) when pure alpha and beta anomers are dissolved in water (via interconversion to opposite anomer)
Ratio of a to b anomers for equilibrium of D glucose in water is 64 %(b) to 36% (a)
hemiacetal vs acetal
hemiacetal has one hydroxyl group (OH) and one allkoxy group (OR) — when a sugar has a hemiacetal is is capable of ring opening and sits in an equilibrium between cyclic and ring opened stat
acetal has two alkoxy groups - means that the ring structure is ‘locked’
What do sugars become when reacted with methanol
methylglycosides (acetals)
What is the Purdie-Irvine reaction?
When sugars are reacted with methyl iodide and silver oxide to form sugar methyl ethers
Rule for deducing L or D from a Fischer projection (sugars)
If the OH of the lowest stereogenic center is to the left - L
OH of lowest stereogenic center is to the right - D
What is the difference between alpha and beta linkages
Alpha linkages have the oxygen connected to anomeric carbon pointing down/axial/trans (alpha - axial)
Beta linkages have oxygen pointing up/equatorial/cis — create much more rigid chains
difference between DNA and RNA structures
RNA is ribonucleic acid- so has ribose sugar where DNA is deoxyribonucleic acid, so it is missing an oxygen at the 2’ carbon of the sugar — deoxyribose sugar
RNA has Uracil base instead of Thymine
What are DNA and RNA chains made up of
Base- sugar - phosphate
what is the difference between a nucleoside and a nucleotide
Nucleosides lack a phosphate
What is the difference between purines and pyrimidines
Purine nucleotides have 2 rings and pyrimidines only have 1
Chargaff’s rule
A+G = T+C
The percentage of A is equal to the percentage of T
The percentage of G is equal to the percentage of C
Watson Crick hydrogen bonding pairs
A with T
C with G
What direction do DNA chains run in
5"‘——> 3’
central dogma
DNA»RNA»Protein
Transcription
DNA to mRNA
translation
mRNA to protein
3 (main) kinds of RNA
messenger RNA (mRNA) - carry code from DNA to assembly of protein on ribosome
transfer RNA (tRNA) - carry individual amino acids (bound as esters) for protein biosynthesis
ribosomal RNA (rRNA) - structural material of the ribosome
how do tRNAs work
they carry amino acids bonded to each as an ester. When they co-locate on the ribosome they tranfer their amino acids and form peptide bonds in sequence (dictated by mRNA) and form a specific protein chain.
amino acids with ionizable side chains
Arginine (R), Lysine (K), Histidine (H), Aspartic acid (D), Glutamic acid (E), Tyrosine (Y), and Cysteine (C)
Which amino acids positive at pH 7
Lysine and arginine
Which amino acids are negative at pH 7
Aspartic acid and glutamic acid
Strecker synthesis of amino acids- broad 3 steps and what reagents are used?
Starts with an aldehyde forms aminonitrile intermediate with NH4Cl/NaCN which then forms a racemic amino acid using H2O/HCl
Specific steps of strecker synthesis -
NH3 attacks in at the carbonyl carbon of the aldehyde to form an aminal
OH is protonated to form water and water leaves as leaving group, and an imine is formed
Imine is protonated at NH and CN- attacks in at carbon to form aminonitrile
The nitrile is hydrolyzed by water in acidic medium to form a carboxylic acid (primary amide)
Water substitutes into primary amide then undergoes proton tranfer to form the racemic amino acid
Convention for listing amino acids in a peptide chain (which way do you list)
From the N-terminus (amino group) to the C-terminus (carboxyl group)
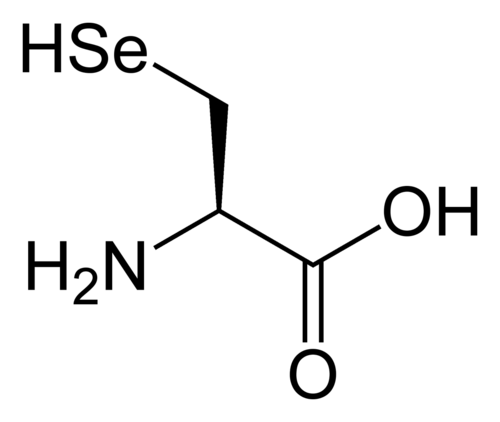
Selenocysteine (21st proteinogenic amino acid)
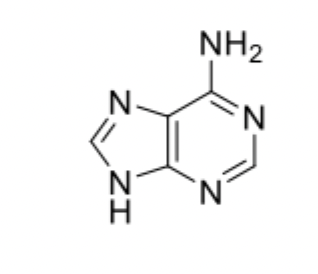
Adenine
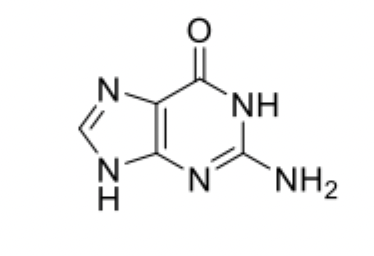
Guanine
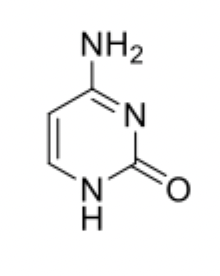
Cytosine