Chemistry: KMT and Gas Laws
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
Kinetic Molecular Theory (KMT)
Based upon the idea that particles of matter are always in motion.
gas
Which state of matter’s particle arrangement is being describe:
Total disorder; much empty space; particles have complete freedom of motion; particles far apart.
liquid
Which state of matter’s particle arrangement is being describe:
Disorder; particles are free to move relative to each other; particles close together
crystalline solid
Which state of matter’s particle arrangement is being describe:
Ordered arrangement; particles are essentially in fixed positions; particles close together
molecules
Kinetic Molecular Theory is an important theory for explaining the behavior of _________ in matter.
moving particles
Kinetic Molecular theory shows how all matter is made up of ________ _________ too small to be seen with the smallest microscope.
gases
Kinetic Molecular Theory helps explain physical properties and behavior of which state of matter?
small particles
The Kinetic Molecular theory states all matter is composed of what?
kinetic energy
The Kinetic Molecular theory states that the particles in matter are in constant random motion; they possess what kind of energy?
gas
The Kinetic Molecular theory states that _____ particles move in a straight line, and they collide with each other or the wall of their container; the collision between particles are perfectly elastic, which means “no loss of energy.”
do not
The Kinetic Molecular theory states that gas particles _____ attract or repel each other.
temperature
The Kinetic Molecular theory states that the average kinetic energy of the particles depends on the __________ of the gas (gas particles only - this is Kelvin temperature)
kinetic energy
The energy due to the motion of a particle
velocity
Increase in kinetic energy means more particle motion, greater ________ (speed).
slowly
Do bigger particles move faster or slowly?
Temperature
A measure of the average kinetic energy of the particles in a substance.
yes
If the 2 substances have the same temperature, do they have the same KE regardless of particles present?
direct (Increase T, increase KE; Decrease T, decrease KE)
Does temperature and KE have a direct or indirect relationship?
fahrenheit, celsius, kelvin
Name the 3 units of temperature.
farenheit
Unit of temperature used in the U.S. (based on average human body temperature)
celsius
Unit of temperature used in the U.K. (based on movement of water)
Kelvin
Unit of temperature used in science (based on movement of particles)
K = C + 273
Formula for converting Celsius to Kelvin.
C = K - 273
Formula for converting Kelvin to Celsius.
standard temperature
the freezing point of pure water at sea level in air at standard pressure (1 atm)a
absolute zero
Is a theoretical temperature, it is the lowest possible temperature where nothing could be colder and no heat energy remains in the substance.
no (they are completely at rest)
During the absolute zero temperature, do atoms transmit thermal energy?
pressure
The amount of force applied over a surface area
pressure = force/area
Formula for pressure.
increase
In a rigid container, there is an increase in temperature and a _________ in pressure.
decrease
In a rigid container, there is an decrease in temperature and a _________ in pressure.
Newtons (N)
What unit is used to measure force (a push or pull)?
m2, cm2, mm2, km2
What units are used to measure area (length x width)?
torr, kPa (kiloPascals), bar, atm (atmosphere), Pa, mmHg (millimeter of Mercury), psi (pounds per square inch)
What are the standard units of pressure?
gas molecules
Can apply pressure on the walls of a container. Ex: Shake up a coke bottle…. The bottle becomes harder because you’ve increased the pressure inside the bottle.
Barometer
A device used to measure air pressure.
Internal Pressure/Gas (vapor) Pressure
Depends on the number of collisions with the inside wall of the container
direct
Do pressure and temperature have a direct or inverse relationship?
Atmospheric Pressure (air pressure)/External Pressure
Results from the collisions of air molecules with the outside of objects.
yes (no volume = lots of empty space)
Can gases be compressed?
diffusion
Gases undergo _______ - random motion.
diffusion
Is movement of molecules from a region of high concentration to a region of low concentration.
mass; temperature
Diffusion depends on the ______ (or size) of the particles and ___________.
Lighter
________ (Lighter or Heavier?) molecules travel faster, have more frequent collisions and thus diffuse more rapidly. Higher temperatures will cause particles to diffuse rapidly as well.
properties of gases
What was one of the earliest phenomena that chemists studied?
gas laws
Laws that bring together temperature, volume, and pressure and helps us see how those factors are related.
constant
The gas laws state that the number of gas particles remains _________.
closed
Gas laws are only relevant in __________ systems.
Robert Boyle
Anglo-Irish philosopher, chemist, physicist and alchemist. He studied the relationship the volume and pressure of a sample of gas. He’s known for his book The Skeptical Chemist. He refuted Aristotle’s idea that all matter was composed of earth, air, fire, and water.
Boyle’s Law
Law that states if the temperature is constant, when the pressure goes up, the volume goes down. This is an inverse relationship.
a curve
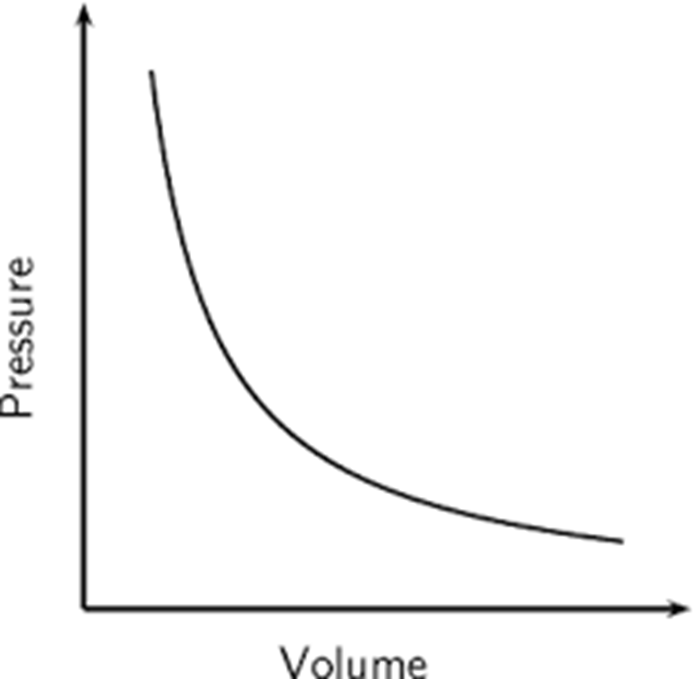
What shape is the graph of an inverse curve?
P1 x V1 = P2 x V2
What is Boyles Law formula?
Jacques Charles
French physicist
He was on the second balloon ever to lift a human being off the surface of the earth
Studied the effect of temperature on the volume of a gas at constant pressure
Charles’ Law
The law that states if the pressure is constant, when the temperature goes up, the volume goes up (elastic container). This is a direct relationship.
a straight line
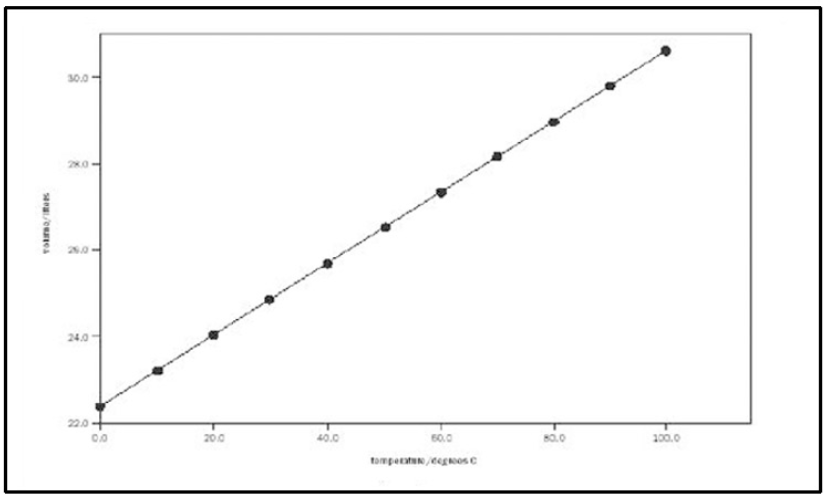
What does the graph of a direct relationship look like (for Charles’ Law)?
V1/T1 = V2/T2
What is Charles’ Law formula?
Joseph-Louis Gay-Lussac
French Chemist
Charles never published his work, so this chemist repeated Charles’ work and published the results.
He followed up the work changing the container to a rigid container
Gay-Lussac’s Law
Law that states if the volume is constant, when the temperature goes up, the pressure will go up (rigid container). This a direct relationship.
a straight line
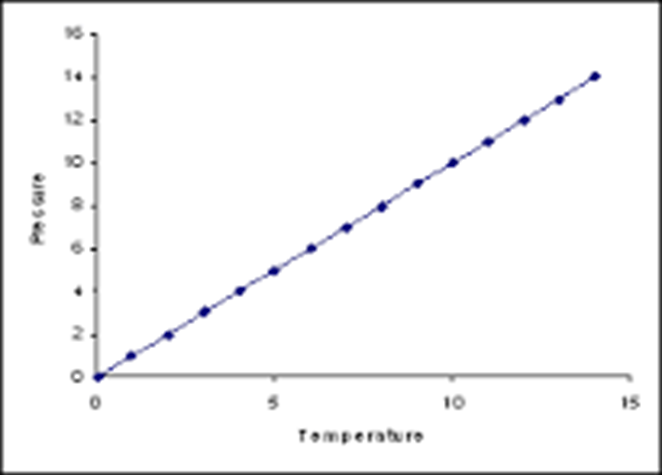
What does the graph of a direct relationship look like (Gay-Lussac’s Law)?
P1/T1 = P2/T2
Gay Lussac’s formula.