2 biomolecules - carbohydrates and lipids
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
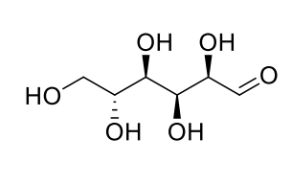
how might this change into 2 other sugars? what are they called?
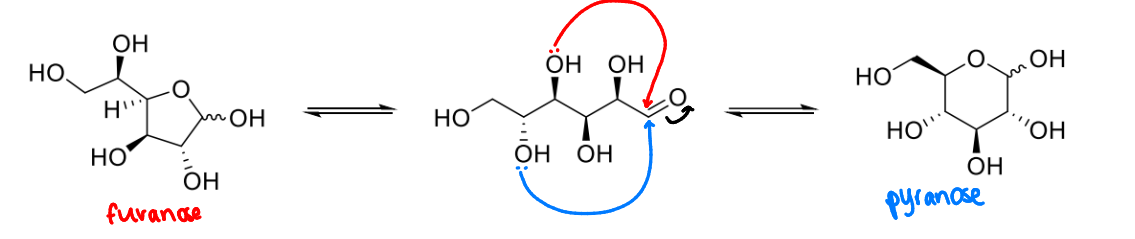
what is the anomeric carbon?
the carbon attached to 2 oxygen atoms
numbering of anomeric carbon
anomeric carbon is always carbon-1
how are sugars numbered
start with anomeric carbon as 1 then number clockwise
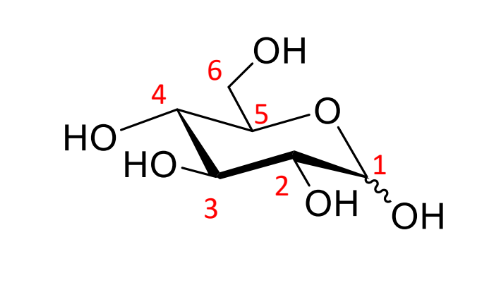
show the two possible stereochemistries
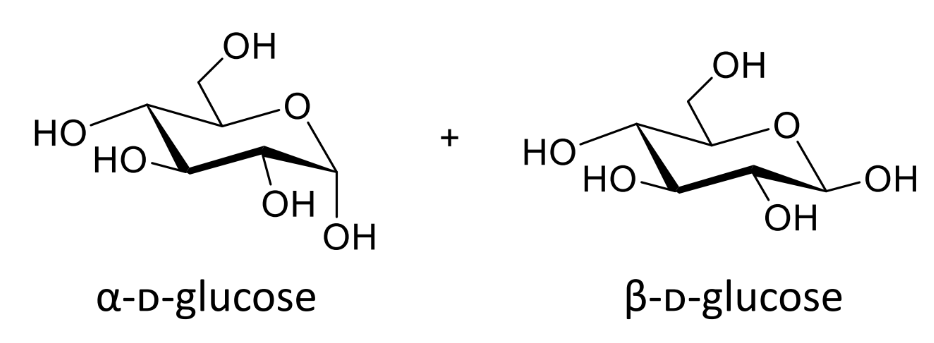
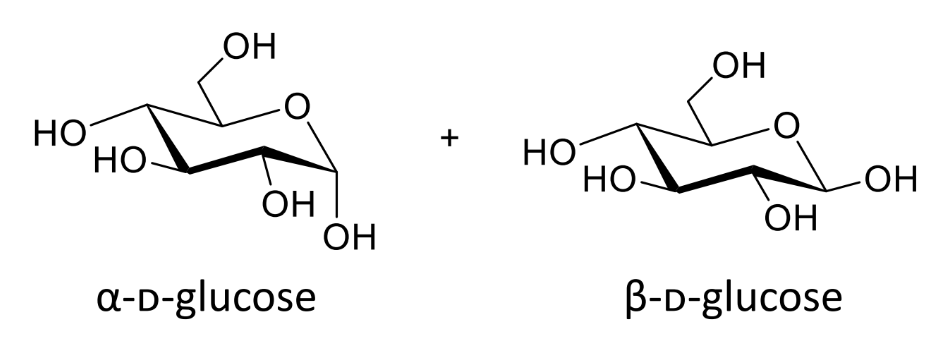
are these the same molecule
no - different molecules not conformations as havent ring flipped
what is mutarotation
the ratio of α and β will change until an equilibrium is reached
does mutarotation require a catalyst
no
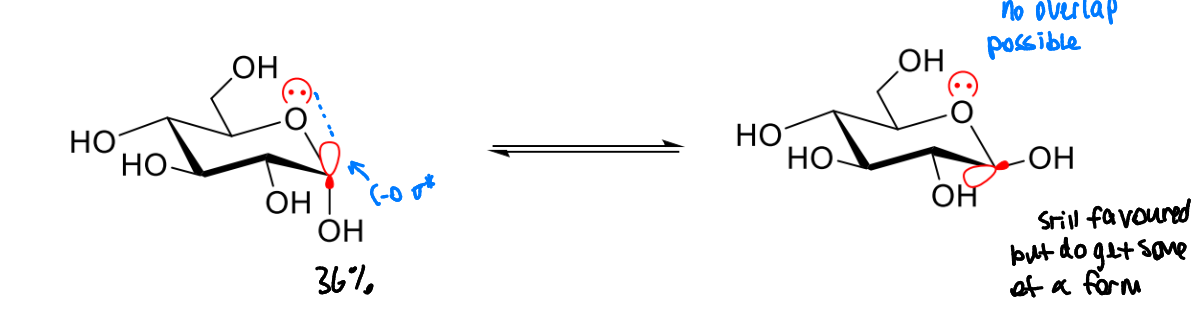
is axial or equatorial favoured in sugars
equatorial not always favoured due to the anomeric effect
what is the anomeric effect
an axial lone pair from the oxygen in the ring that is attached to the anomeric carbon can donate into the empty C-O σ* can increase stability
more of the α isomer may be seen than would be expected in most molecules
the β isomer is still favoured
what are simple sugar molecules called
monosaccharides
how can monosaccharides react
what are the bonds called
react and polymerise through acetal linkages at their anomeric positions
these are called glycosidic linkages
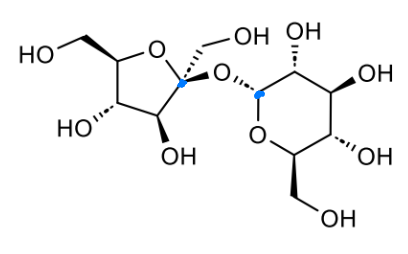
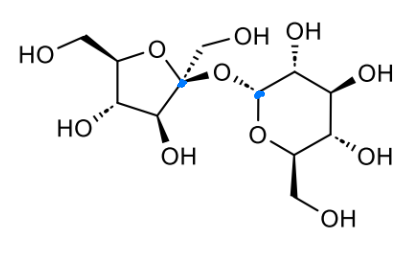
what is this made from
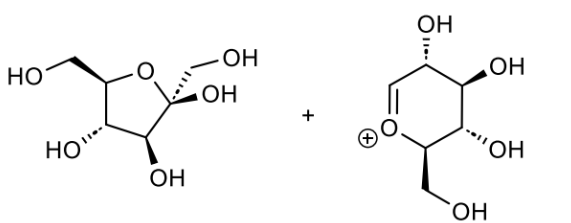
name the bond
at anomeric position, O points down → α
α-1,4-glycosidic linkage
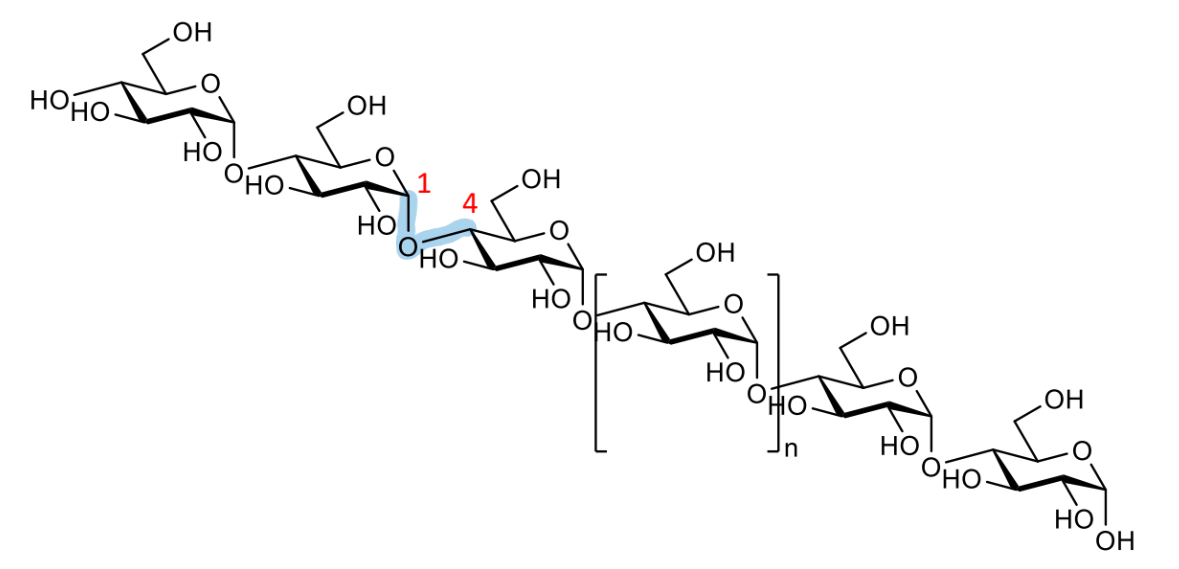
how can polysaccharides form branched molecules
what are 2 examples of this
after formation of one glycosidic linkage, the polysaccharides still have free -OH groups so can interact with further chains
forms branched carbohydrates and dendrimers
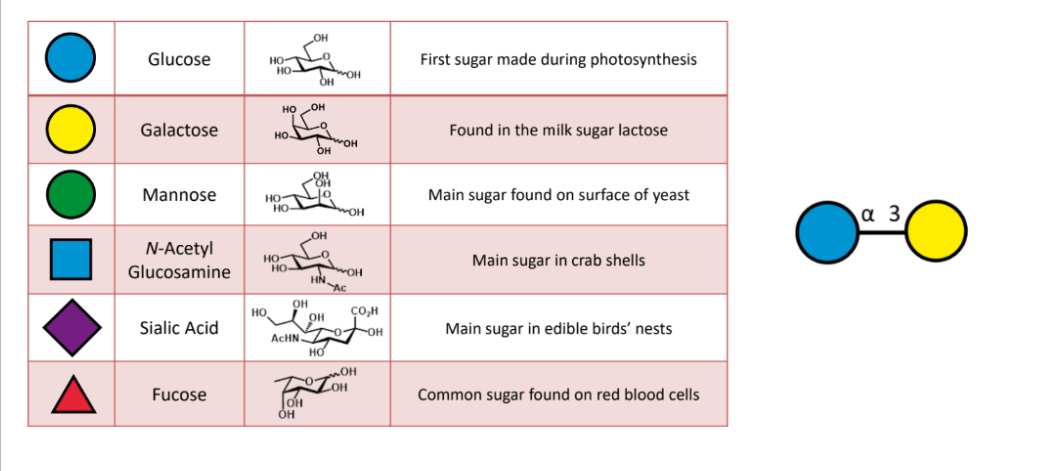
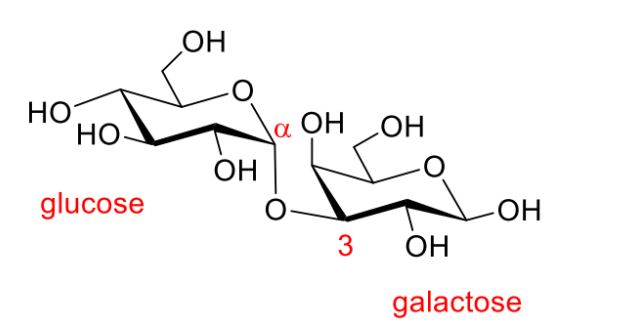
draw out this molecule








what are lipids important for
important component of cell membranes along with carbohydrates
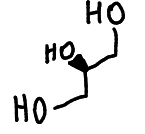
structure of glycerol
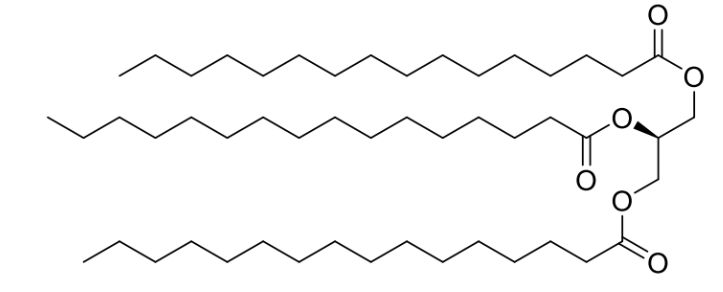
most common type of lipids?
what are they known as and what is their main function
esters of glycerol - glycerol is esterified at all 3 OH positions
these lipid esters are known as triglycerides and mainly function as an energy store
what are the most common lengths of fatty acid and what are their names
C14 = myristic
C16 = palmitic
C18 = stearic
what are phospholipids
replace one of the 3 fatty acid chains of a triglyceride with a polar head group - positively charged
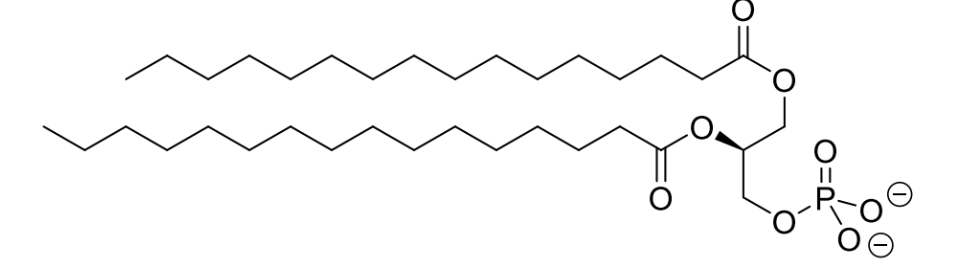
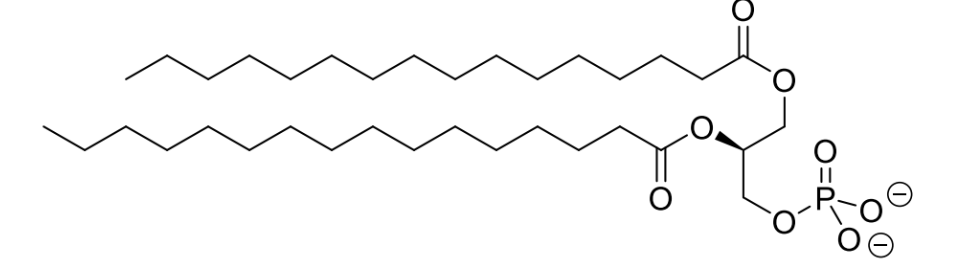
which areas are hydrophilic or hydrophobic
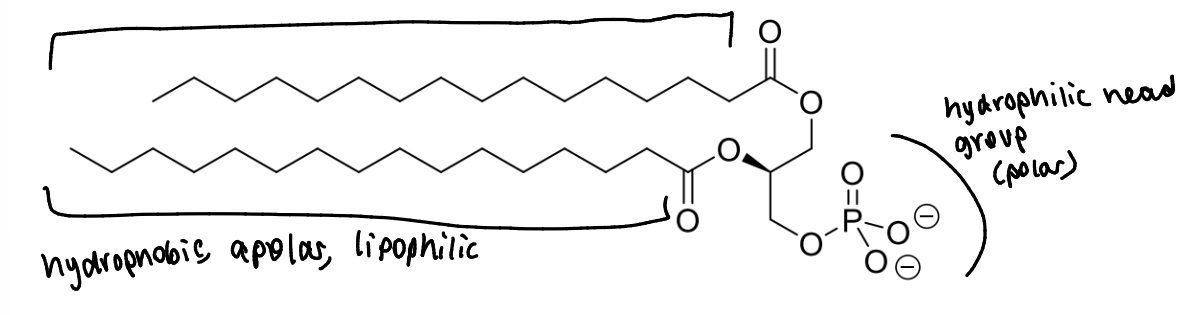
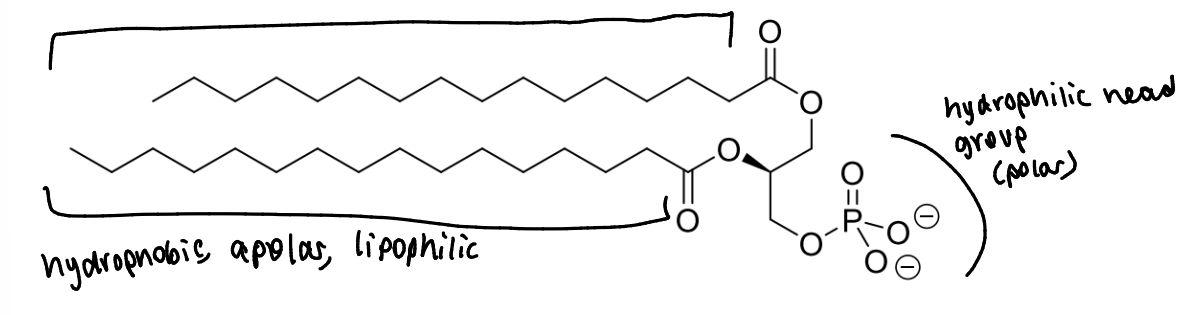
what is the effect of these different areas of the molecule
what is this driven by
polar head and lipophilic tail of phospholipids leads to the spontaneous formation of bilayers in an aqueous
driven by the charge-charge interactions and the hydrophobic effect
what is the significance of the phospholipid bilayer
constitutes a major component of the cell membrane