Reactions Unit Test (Mrs. Herrick Honors Chemistry)
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Chemical Reaction
A process involving the rearrangement of atoms or molecules to form new substances with different properties
Chemical Change
A process where one or more substances are altered into one or more new substances with different compositions and properties
Chemical Equation
A written representation of the process that occurs in a chemical reaction
Reactants
Substances that start a chemical reaction
Product
The substances that are formed as a result of the reaction
(s)
Solid
(l)
Liquid
(g)
Gas
(aq)
Aqueous solution
Aqueous Solution
A solution in which the solvent is water
Precipitate
A solid substance that forms when two aqueous solutions react and produce an insoluble compound
Single Replacement Reactions
Occurs when one element replaces another element in a compound
Oxidation
The loss of electrons from an atom or molecule during a chemical reaction
Reduction
A chemical reaction that involves the gain of electrons by a molecule, atom, or ion
Synthesis Reactions
A type of chemical reaction where two or more simple substances combine to form a more complex substance
Decomposition Reaction
A type of chemical reaction where one compound breaks down into two or more simpler substances
Combustion Reaction
Involves a substance reacting rapidly with oxygen, often resulting in the formation of carbon dioxide and water
Coefficients
Numerical values placed in front of chemical formulas in chemical equations to indicate the proportion of reactants and products
Subscript
Used after an element symbol to indicate the number of atoms present in a molecule or compound
List signs or evidence of a chemical change
Color changes
Temperature changes
Bubble production
Odor emission
Formation of a precipitate
Complete Ionic Equation
Shows all of the ions present as either reactants or products in a chemical reaction
Net Ionic Equation
A chemical equation that shows only the molecules or ions participating in a chemical reaction, getting rid of the spectator ions
7 Diatomic Elements
Br
I
N
Cl
H
O
F
Spectator Ions
Ions that exist in the same form on both sides of a chemical equation. They do not participate directly in the reaction
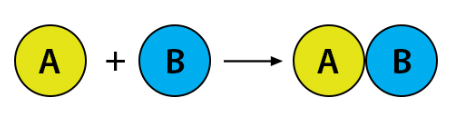
What type of reaction is taking place?
Synthesis
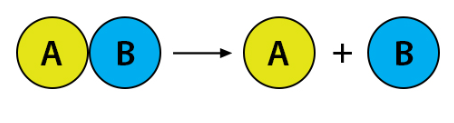
What type of reaction is taking place?
Decomposition
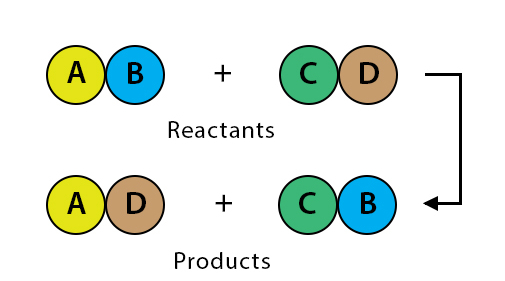
What type of reaction is taking place?
Double replacement
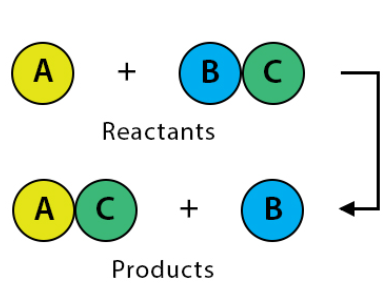
What type of reaction is taking place?
Single replacement
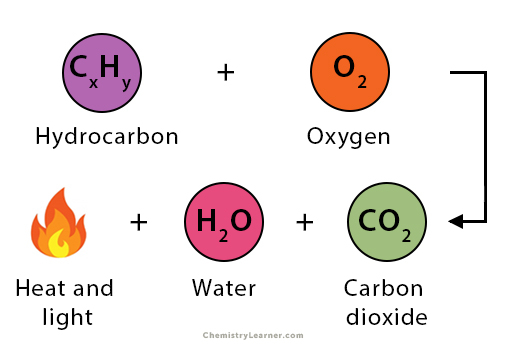
What type of reaction is taking place?
Combustion
Solute
Substance dissolved in a solution
Solvent
The most plentiful substance in a solution
Is a chemical reaction taking place?: A tomato smells rotten
Yes
Is a chemical reaction taking place?: A drinking glass breaks into smaller pieces
No
Is a chemical reaction taking place?: A piece of ice melts
No
Is a chemical reaction taking place?: A solid “growth” forms on a piece of aluminum metal when added to copper II chloride
Yes
Is a chemical reaction taking place?: Candle wax burns
Yes
What substances were involved when a rusty solid formed over the aluminum wire during the Copper Reaction Lab?
Cu (s)
What substances were involved when the blue precipitate turned black once heated during the Copper Reaction Lab?
Cu(OH)₂ (s) → CuO (s)
What substances were involved when a blue green solution formed when acid was added to the mixture during the Copper Reaction Lab?
CuSO₄ (aq)