Biochem SI
1/138
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
139 Terms
What the name of a chemical that is attracted to regions of positive charge, such as a nucleus?
Neocleophile
What is the name of a chemical that is a electron deficient and loving?
electrophile
What kind of inhibitor most resembles the substrate when it binds to the enzyme?
Competitive
fill in the chart
Competitive Uncompetitive Noncompetitive
Vmax: Vmax: Vmax:
Km: Km: Km:
Competitive Uncompetitive Noncompetitive
Vmax: Unchanged Vmax: Decreases Vmax: decreases
Km: increased Km: decreases Km: unchanged
Which type of reversible inhibition takes place when the inhibitor binds to the enzyme substrate complex? What happens when the substrate concentration is increased?
uncompetitive, nothing
What type of reversible inhibition takes place when the inhibitor binds directly to the active site of the enzyme, reducing the rate of catalysis? What happens when the substrate concentration is increased?
competitive, increases substrate concentration overcomes inhibition
What type of reversible inhibition takes place when the inhibitor bind to an enzyme at the same time at different binding sites? What happens when the substarte concentration is increased?
non competitive, nothing
What type of irreversible inhibitor is structurally similar to the enzyme’s substrate and modifies the enzyme?
Affinity labels
What type of irreversible inhibitor binds to the enzyme as a substarte, converting it to an irreversible inhibitor that can no longer catalyze?
Suicide inhibitors
What type of irreversible inhibitor react with specific R groups of enzyme?
Group specific reagent
What type of irreversible inhibitors are designed to strongly bind to active site but not react?
T. S. analogs

What is a real-life example of a suicide inhibitor in medicine?
Penecillin
What is the physiology role of myoglobin? Hemoglobin?
Myoglobin stores oxygen and Hemoglobin transports oxygen.
Fill in the following chart with the characteristics of myoglobin and hemoglobin:
Myoglobin Hemoglobin
What type of kinetics?
How many subunits?
What structure?
Physiological role?
Fill in the following chart with the characteristics of myoglobin and hemoglobin:
Myoglobin Hemoglobin
What type of kinetics? michaelis Sigmoidal
How many subunits? one four
What structure? tertiary quaternary
Physiological role? store oxygen transport oxygen
What does it mean to say hemoglobin displays cooperativity in respect to oxygen binding and relase?
hemoglobin responds upon the binding of pos and neg effectors. Pos effectors tell it to hold onto oxygen, and neg effectors tell it to dump oxygen.
When Fe 2+ binds with oxygen to form Fe 3+, there will be a slight shift in ions. This shift in ions causes a shift from the tensed state to relaxed state. What is the known as?
Co operative bind
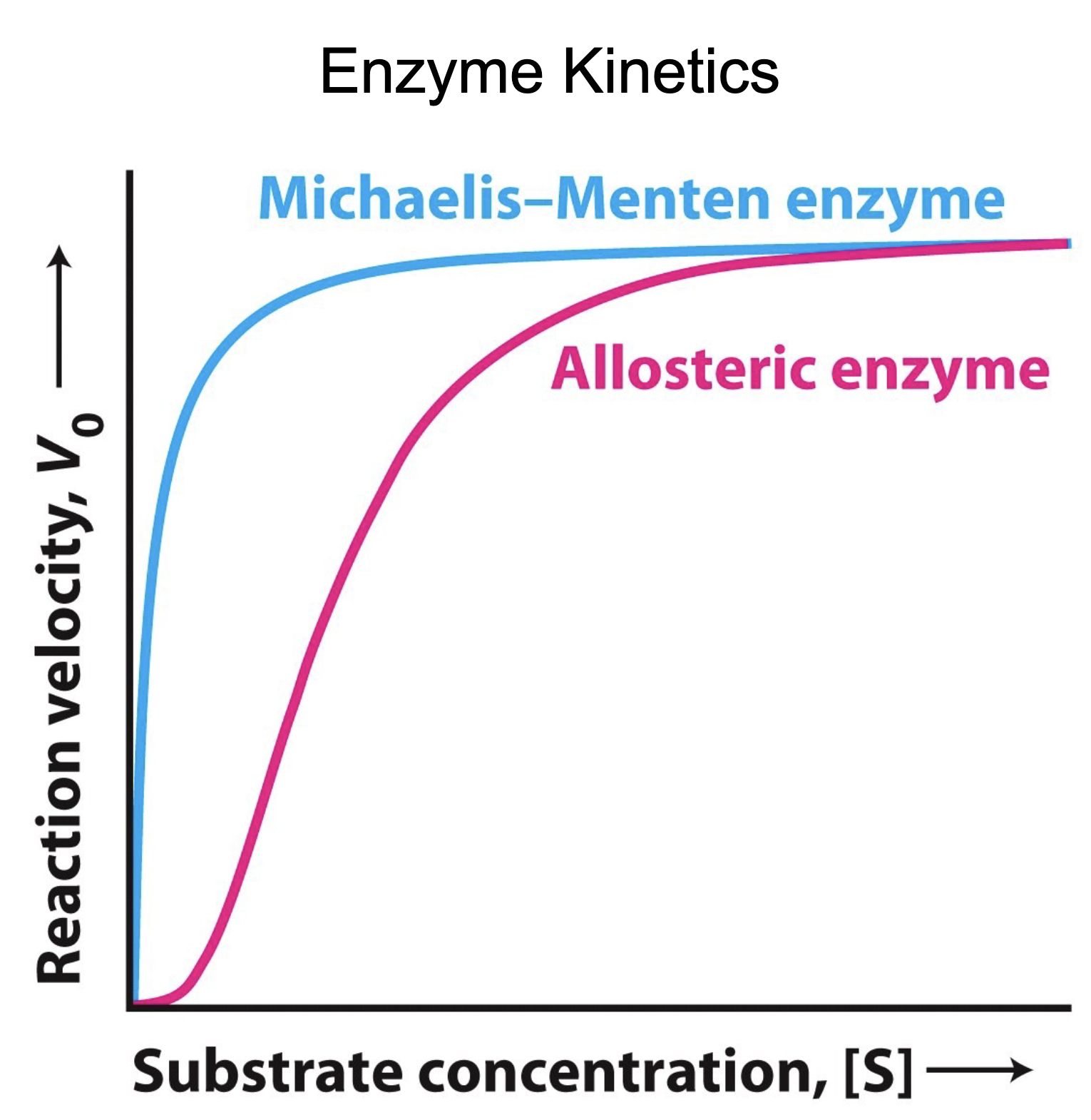
In the curve below, which represents myglobin and hemoglobin?
myoglobin is blue
hemoglobin is red allosteric
Tensed form binds with oxygen ___?___
Relaxed form binds with oxygen ____?____
Poorly
well
Deoxyhemoglobin corresponds to which allosteric state/form? Oxyhemoglobin?
Deoxyhemoglobin is tensed form
Oxyhemoglobin is relaxed form
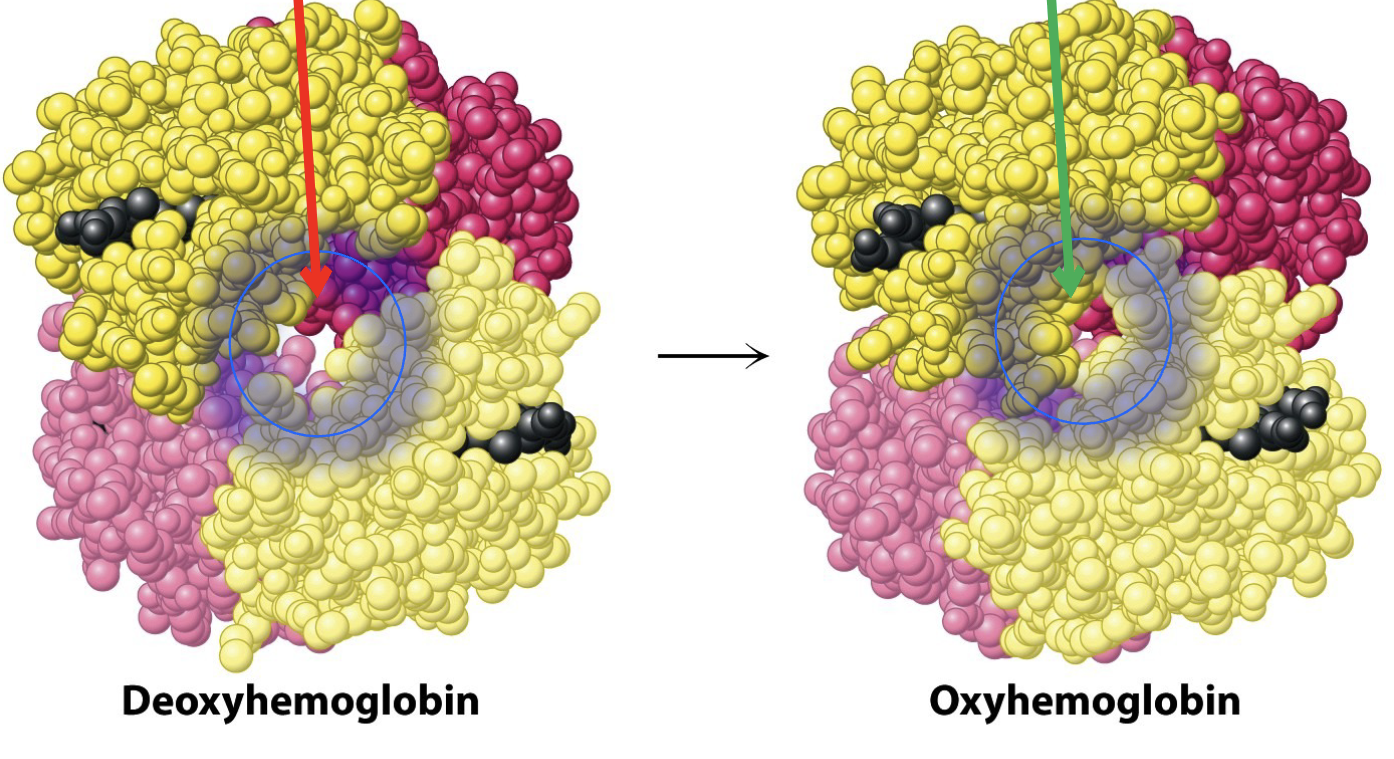
Using the photo which form does BPG readily bind to?
Deoxyhemoglobin bc there is room to bind
Is BPG considered a positive or negative effector? Why?
Negative because stabilized the tensed state/form and facilitates the release of oxygen
(True/False) Fetal hemoglobin binds to BPG well.
False fetal hemoglobin needs oxygen
Oxygen flows from maternal ___?___to___?___deoxyhemoglobin
Oxyhemoglobin to fetal
Which of the following has a higher affinity for oxygen?
Fetal hemoglobin
Regulation of oxygen binding of hemoglobin by hydrogen ions and carbon dioxide is known as?
Bohr Effect
What condition is caused by a mutation in hemoglobin?
sickle cell anemia
Body tissues:___?___CO2 → ___?___pH, favoring the __?___. So, hemoglobin__?___.
Lungs:__?___CO2→__?____pH, favoring the __?___. So, hemoglobin__?___O2.
Body tissues: high CO2 → low pH, favoring the tensed. So, hemoglobin dumps.
Lungs: low CO2→ high pH, favoring the relaxed. So, hemoglobin picks up O2.
Carbohydrates are carbon-based molecules that are rich in what kind of functional group?
Hydroxyl
The simplest monosaccharide is made up of __?___ Carbons.
3
Define the following carbon structures:
Triose:
Tetrose:
Pentose:
Hexose:
Triose: 3 carbons
Tetrose: 4 carbons
Pentose: 5 carbons
Hexose: 6 carbons
Match glucose and fructose to their corresponding ring:
Furan ring →
Pyran Ring →
Furan ring → fructose (5 carbons)
Pyran Ring → glucose (6 carbons)
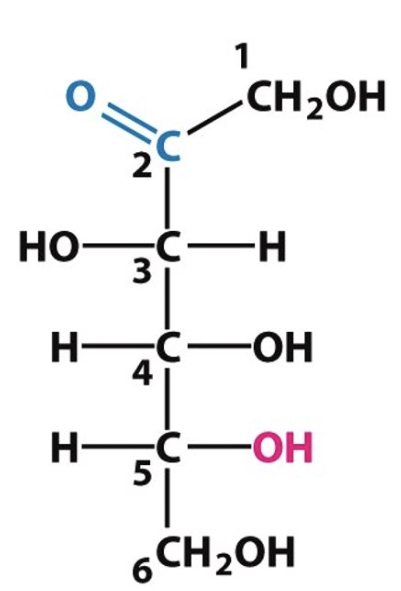
Identify the following structures: (fructose/glucose), (pentose/hexose)
Fructose, hexose
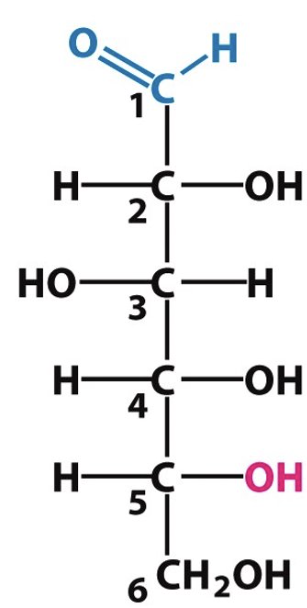
(fructose/glucose), (pentose/hexose)
glucose, hexose
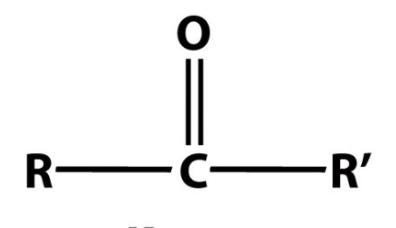
Identify the following functional group:
ketone
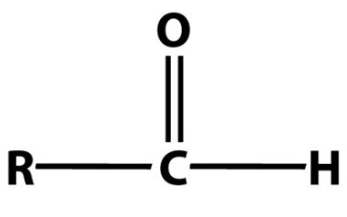
Identify the following functional group:
aldehyde
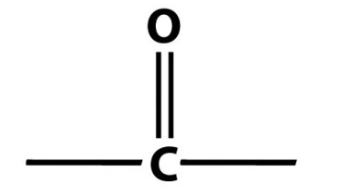
Identify the following functional group:
carbonyl group
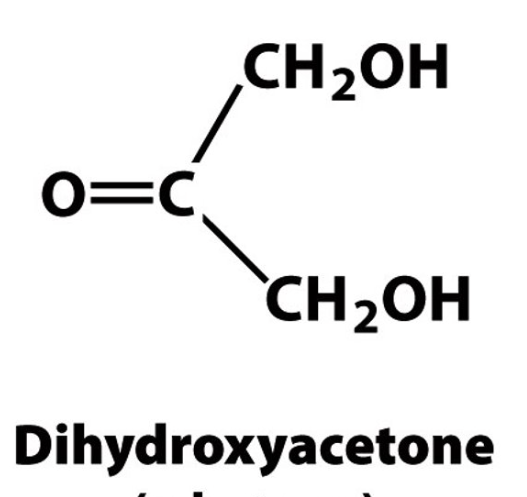
Identify the monosaccaride based on its structure.
ketose
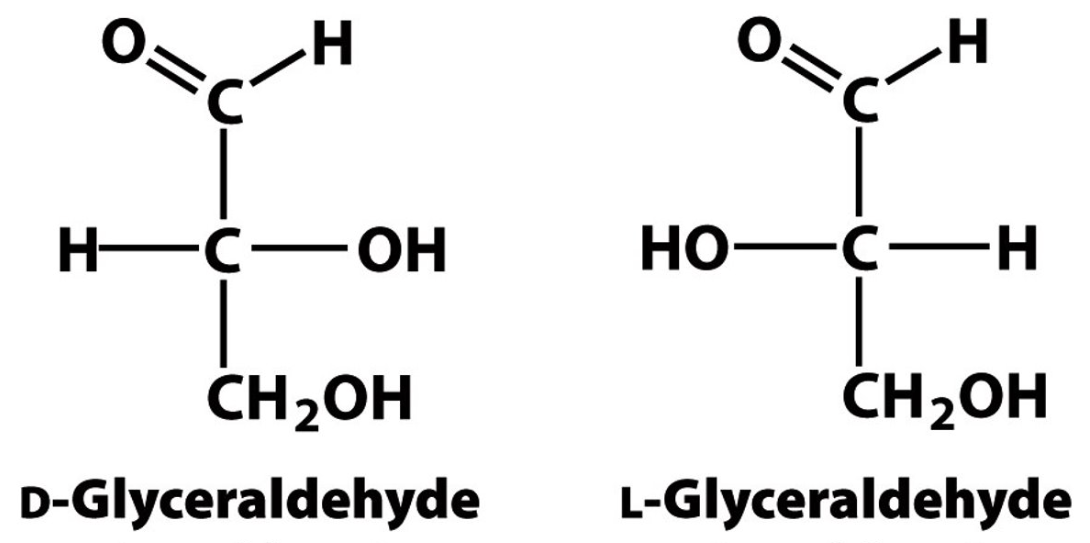
Identify the monosaccaride based on its structure.
aldose
Isomers have the (same/different) molecular formula, but (same/different) structure.
same, different
What kind of isomers have the same molecular formula, but differ in the order to attach of atoms?
Constitutional isomers
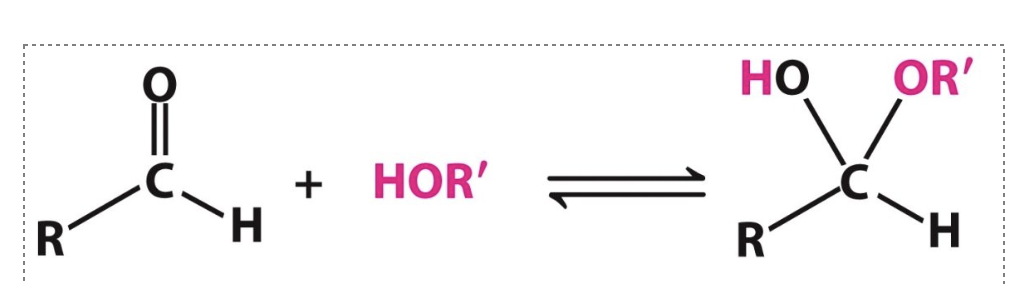
Label the following reaction:
___?____ reacts with ___?_____ to make _____?____
aldehyde, alcohol, hemiacetal
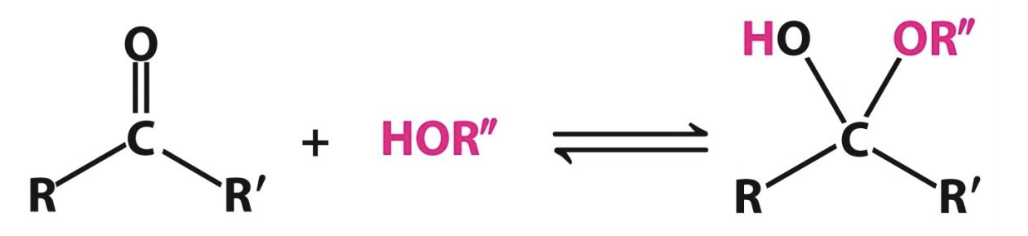
Label the following reaction:
___?____ reacts with ___?_____ to make _____?____
ketone, alcohol, hemiketal
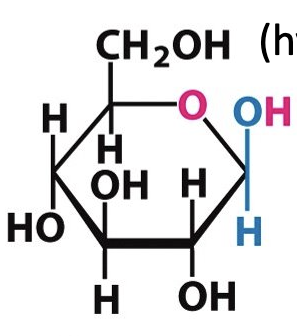
Number the carbons in this molecule:
6
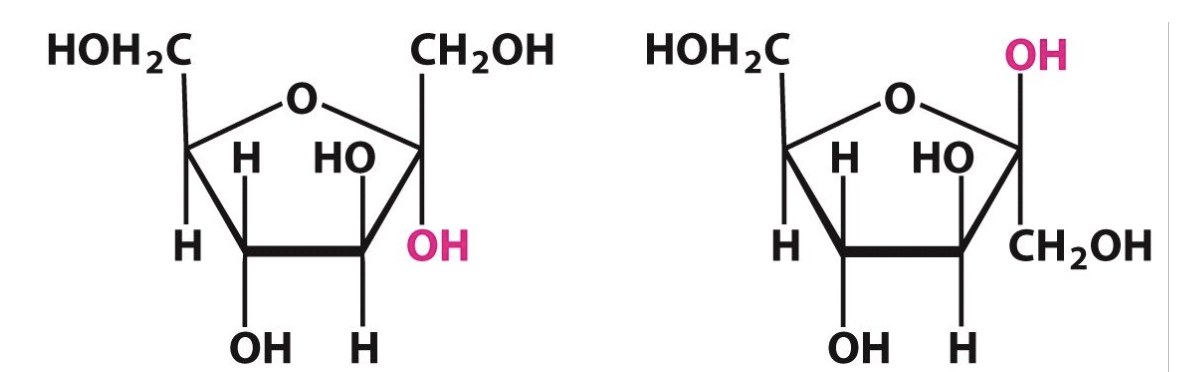
Label the following sugars as having either alpha or beta configerations:
Alpha = below
Beta = above

d and l
Refers to the direction of rotation (d: dextrorotatory , l = levorotatory)
+ and -
Refers to the direction in which molecule rotates plane polarized light.
+ clockwise (dextrorotatory)
– anticlockwise (levorotatory)
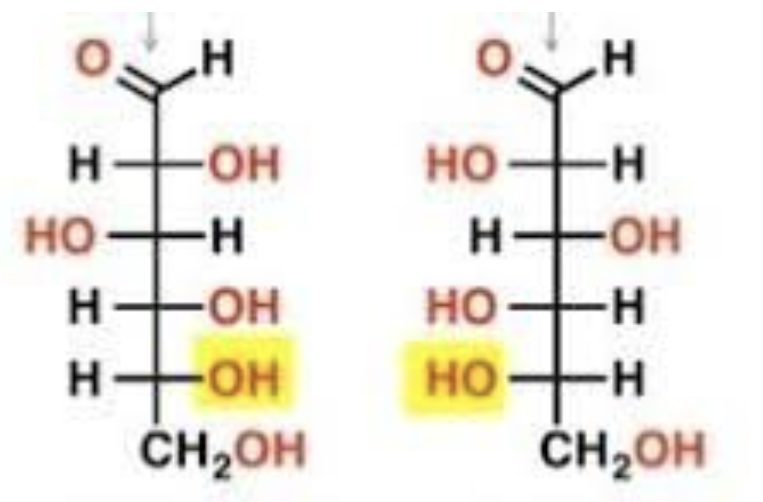
D-Glucose, L-Glucose
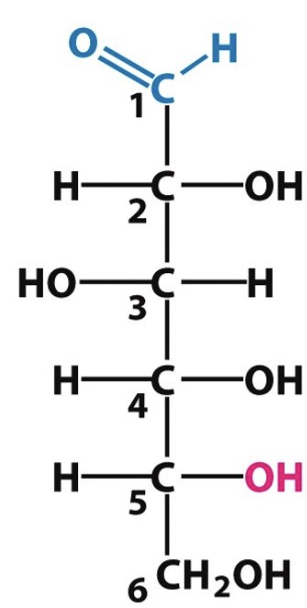
Label the following as either D or L configuration:
D
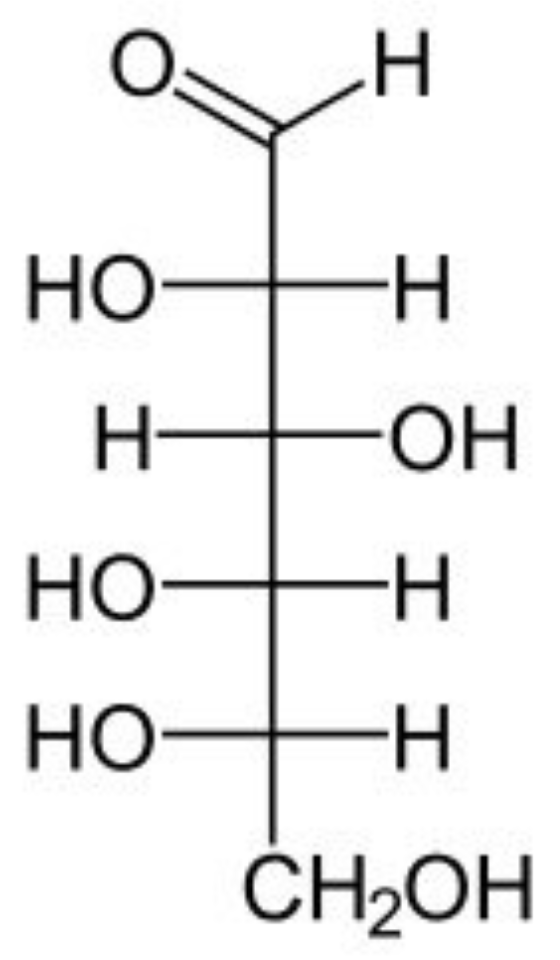
Label the following as either D or L configuration:
L
What are joined together by glycosidic linkages?
Carbohydrates (monosaccharides)
Glucose is a ____?_____ sugar
Reducing
What molecule is released in glycosidic linkages when two monosaccharides connect to form a disaccharide?
Water
What is the function of glycosyltransferase?
They catalyze the information of glycosidic bonds
It takes sugar and attaches it to another molecule creating a glycosidic bond.
Sticks to sugar to make bigger molecule think of it like glue.
The form that glucose is stored in is in the form of:
Starch and glycogen
How is glucose stored in animal cells?
Glycogen

How is glucose stored in plant cells?
Starch
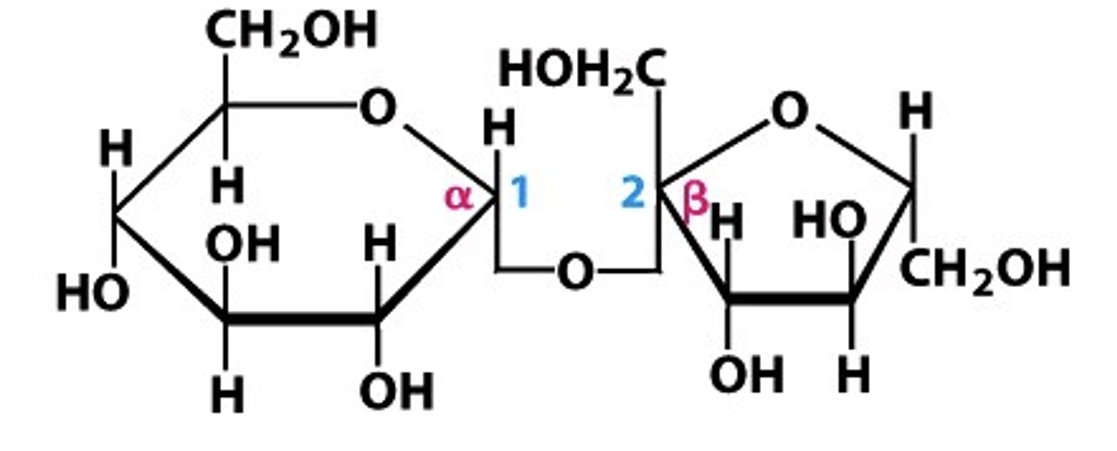
Label the following with alpha or beta, the carbon numbers, and the type of bond:
alpha 1, 2 glycosidic bond
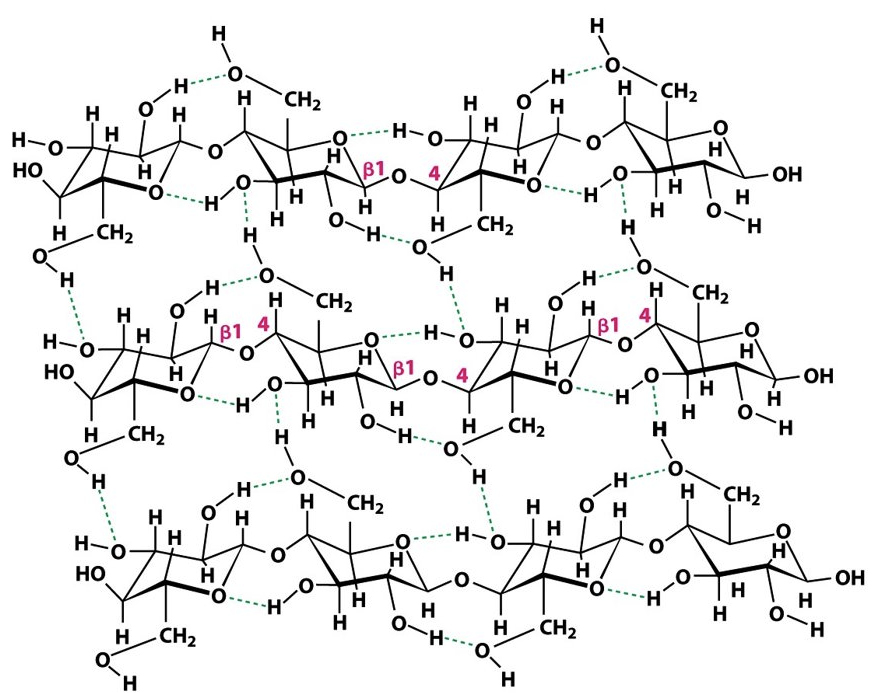
Identify the following structures: (glycogen/cellulose, branched/unbranched)
cellulose, unbranched
mess = unbranched
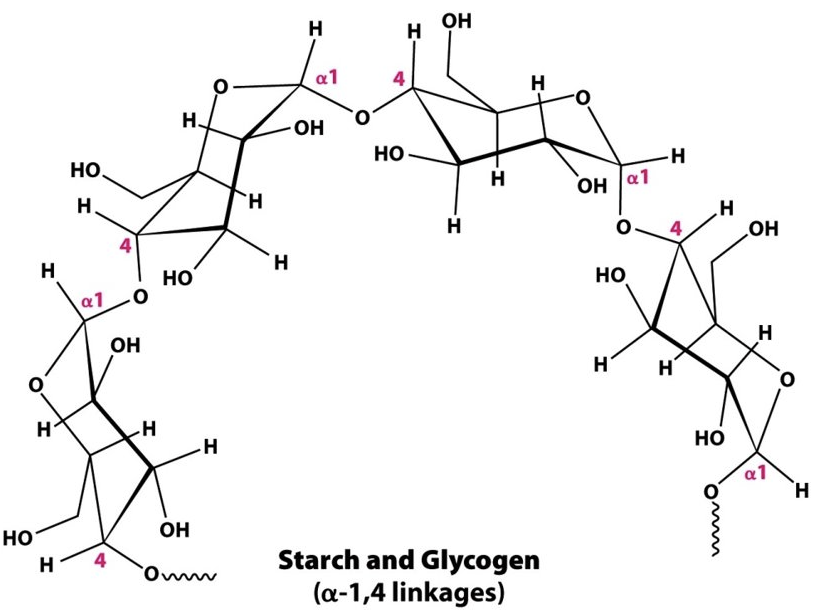
Identify the following structures: (glycogen/cellulose, branched/unbranched)
gylcogen, branched
organized = branched
The largest protein component by weight, component of the cell membrane and take part in cell adhension
glycoprotein
the protein attached to glycosaminoglycan and play a structural role
proteoglycon
the protein that is mainly carbohydrate and is attached to the carbohydrate by N-acetylgalactoseamine. Often act as lubricant.
mucins
What are blood type groups based on? They reflect the specificity of what?
Blood groups are based on protein glycosylation patternThe human ABO blood groups reflect the specificity of glycosyltransferases.
All of the blood groups share the oligosaccharide foundation called O.
In A, N-acetylgalactosamine is added to the O by a specific glycosyltransferase.
In B, galactose is added by another transferase.
(True/Fasle) fatty acids can be saturated and unsaturated
True
Unsaturated fatty acids have a ___?____ melting point than saturated fatty acids.
Lower
Which biomolcule does not have a polymer?
Lipids
(True/False) fatty acids have a polar and a nonpolar part.
true
When there are more (single/double) bonds, the melting point is lower.
Double
Double bonds are always separated by at least __?__ Carbon
1
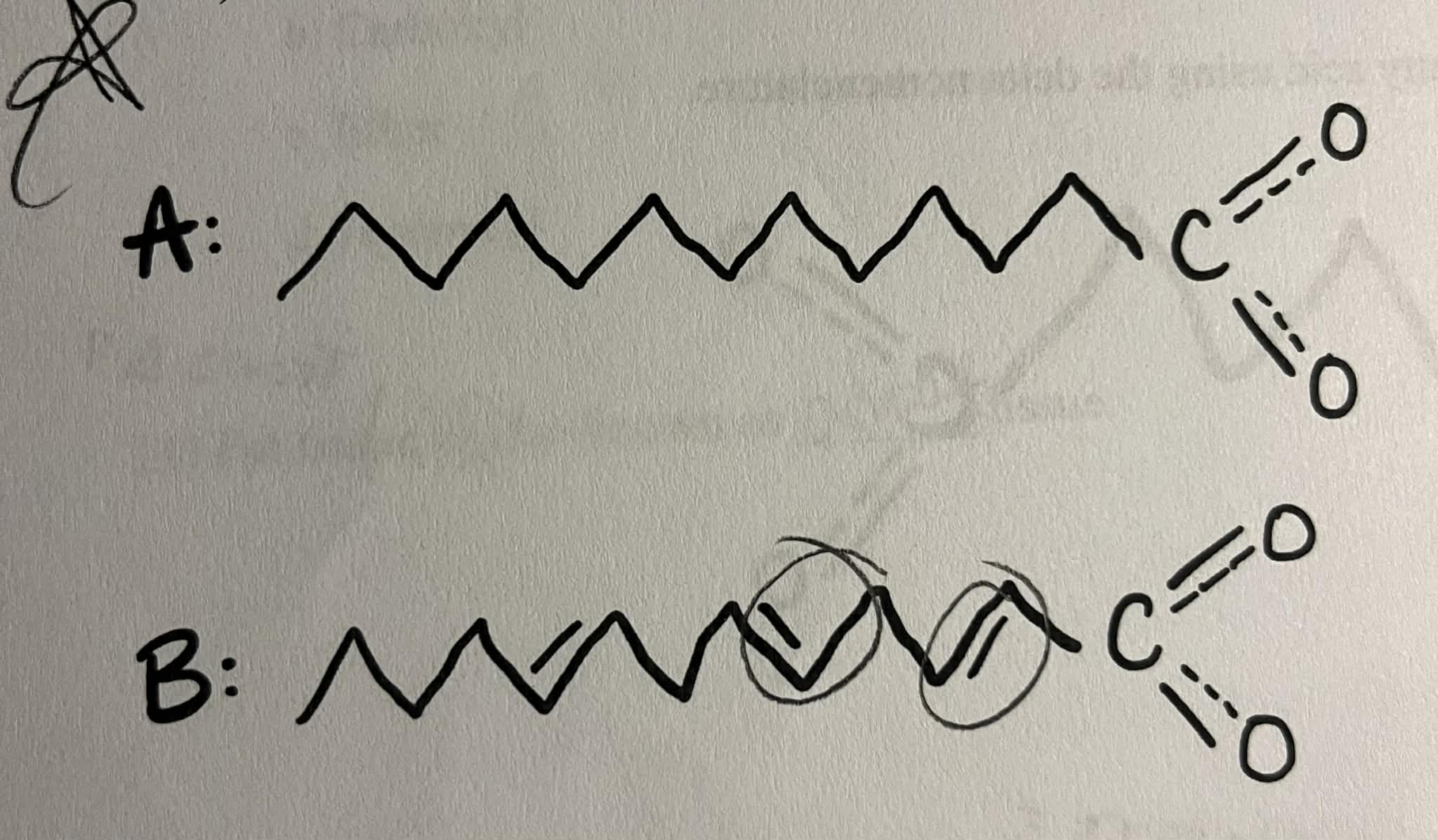
Identify which of the following lipids is saturated and unsaturated.
A: sat = single
B: unsat = double

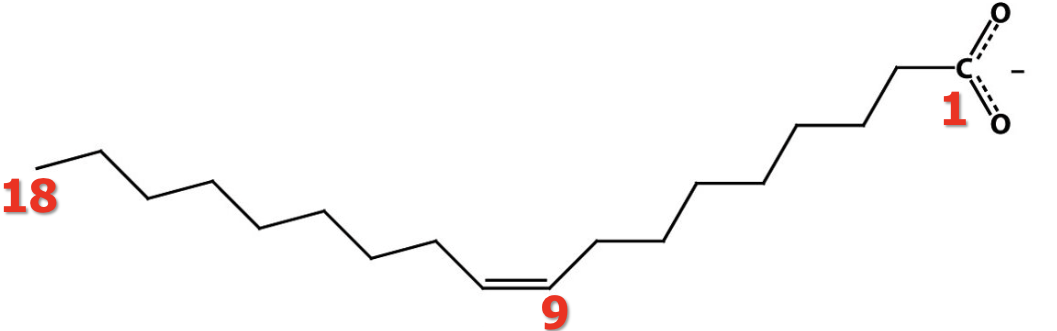
Is this cis or trans configuration?
cis
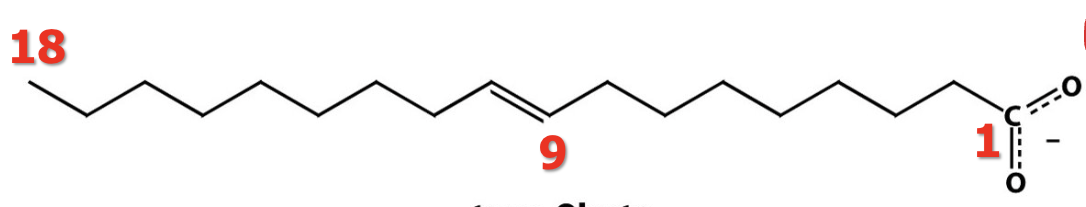
Is this cis or trans configuration?
trans
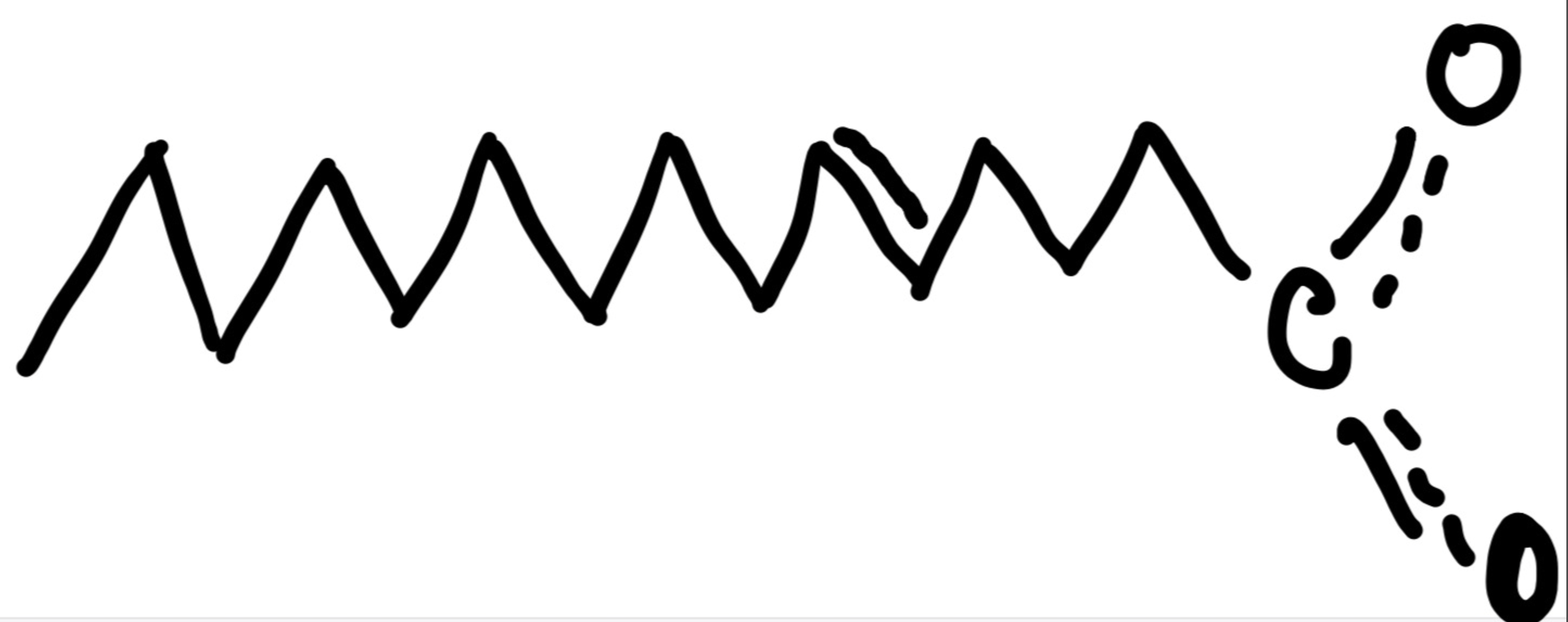
Number the following fatty acids using the omega nomenclature.
Omega 10 fatty acid
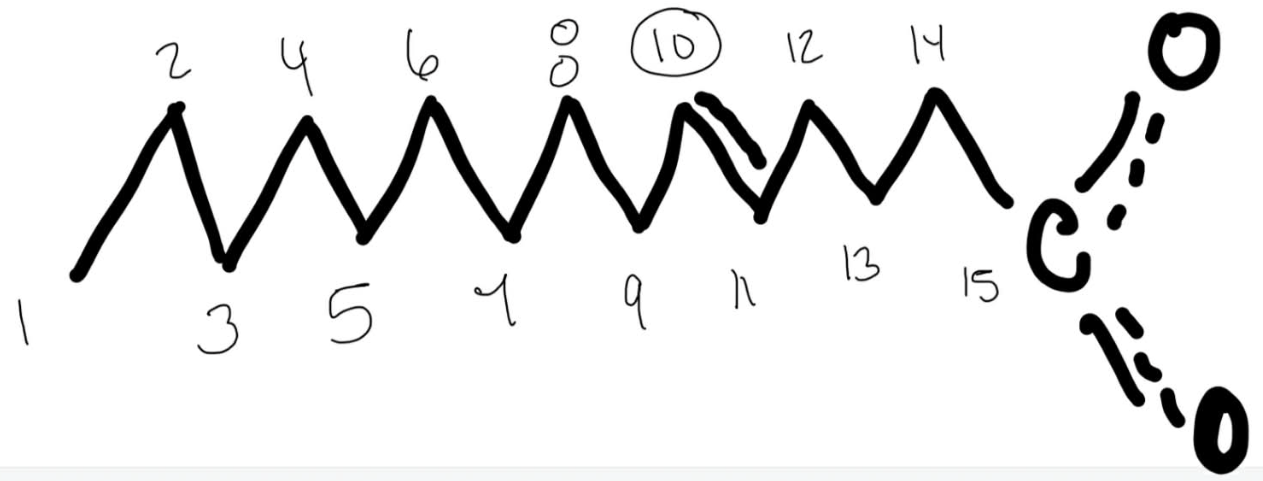
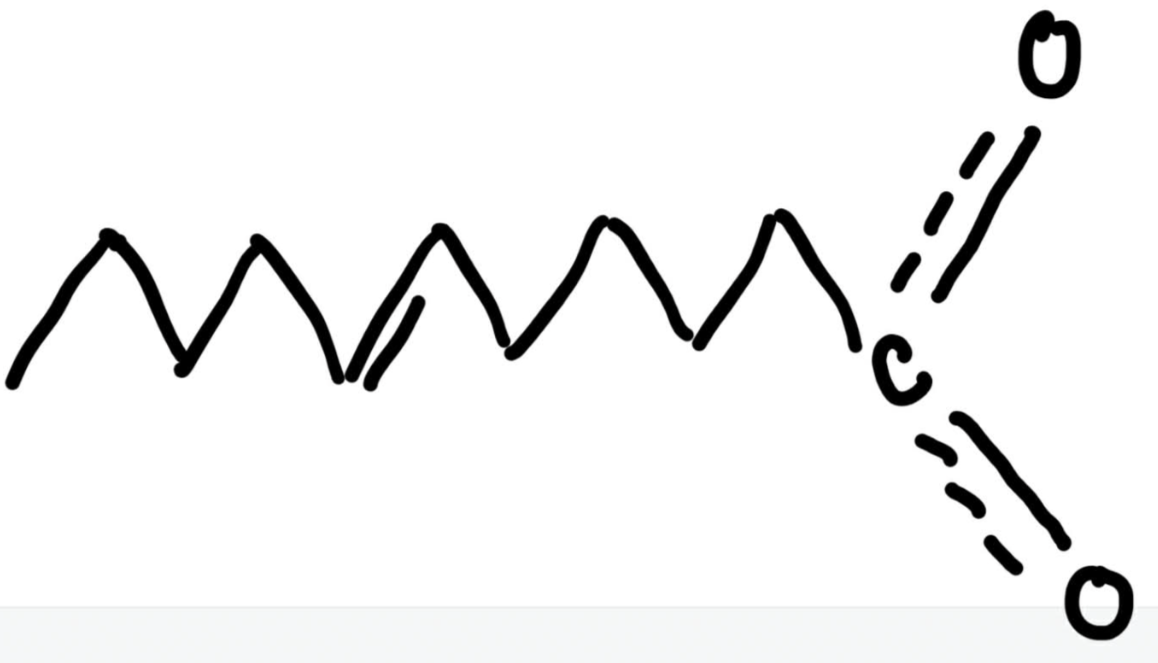
Number the following fatty acid using the delta nomenclature.
12:1 Trans delta 7
*Delta nomenclature = systematic nomenclature
Systematic nomenclature is a standardized, rule-based method for naming chemical substances, ensuring each compound has a unique and unambiguous name derived from its structure.
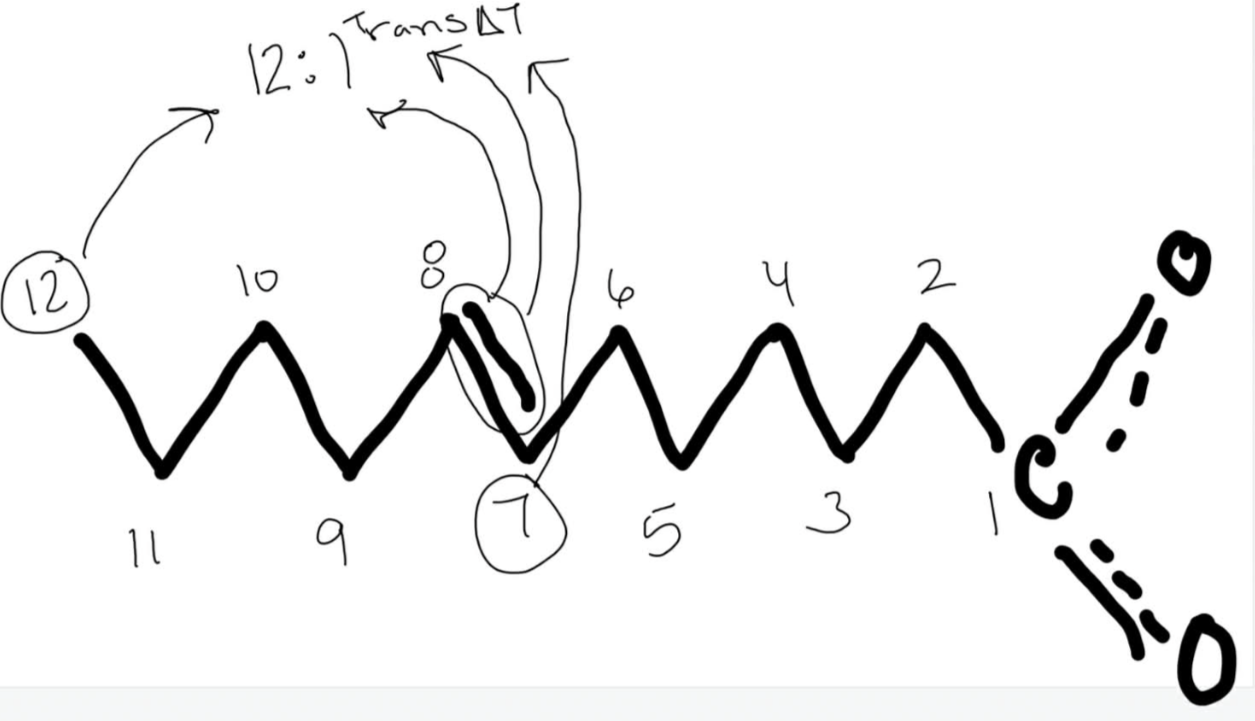
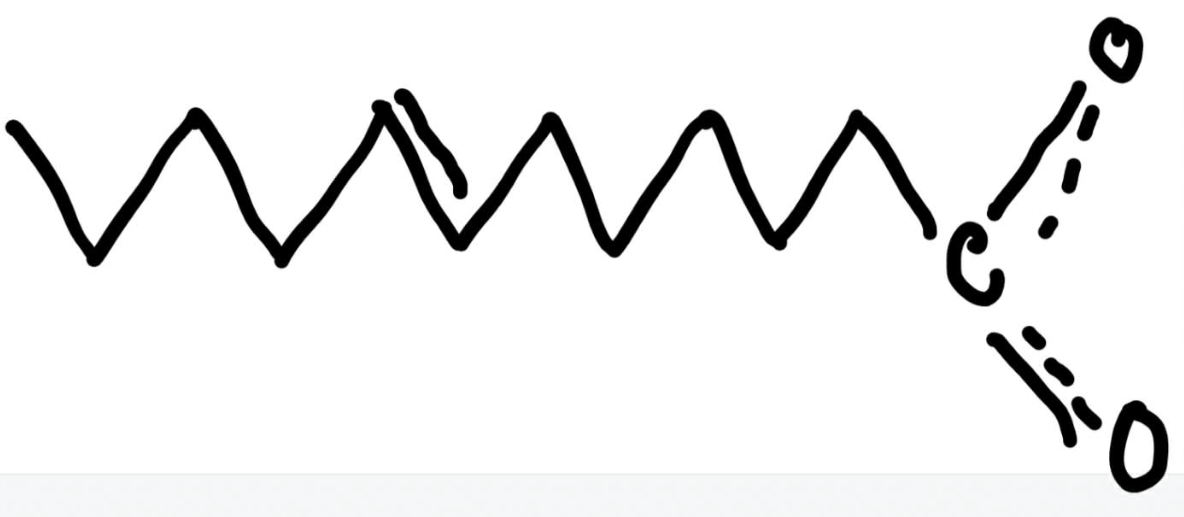
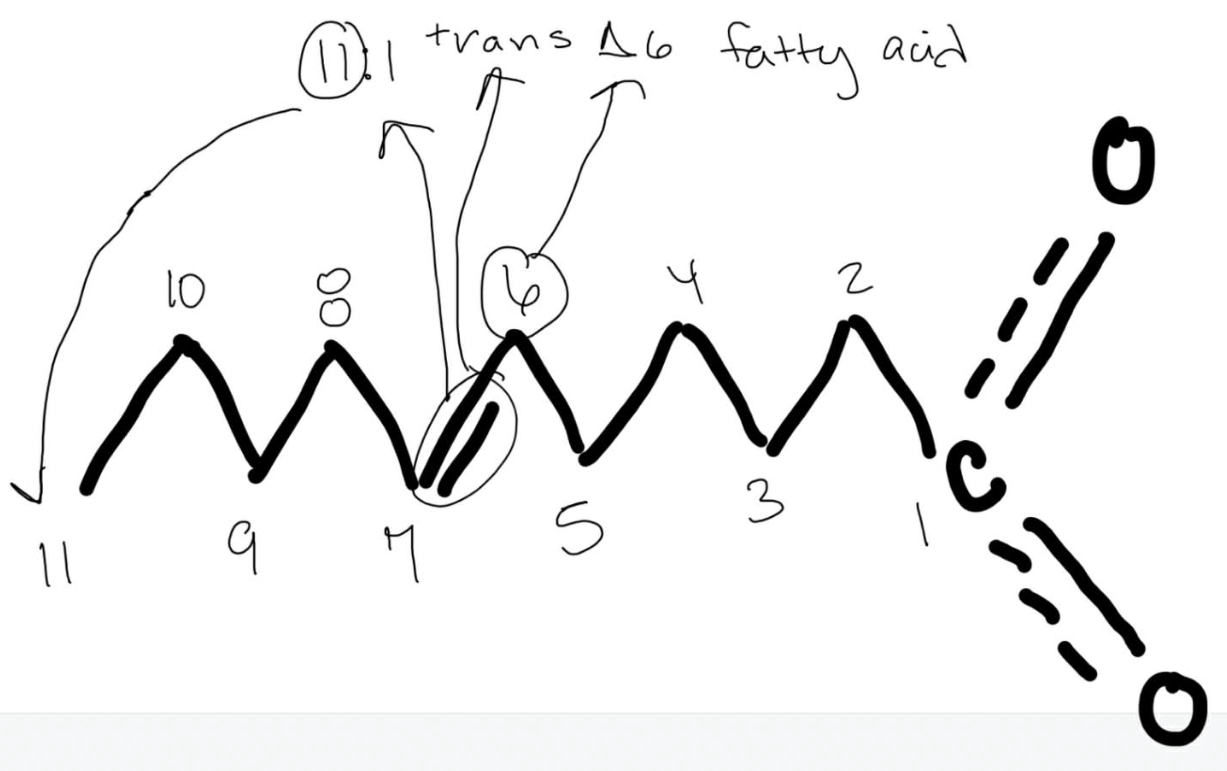
Number the unsaturated fatty acid below and identify the double bond using delta nomenclature.
11:1 trans delta 6 fatty acid
What is commenly known as the storage lipid?
Triacylglycerols: they are energy rich and are designed to stored energy for later use
____?____ bonds attach fatty acids to glycerol.
Ester
Fat issue is also known as ___?_____ tissue.
adipose
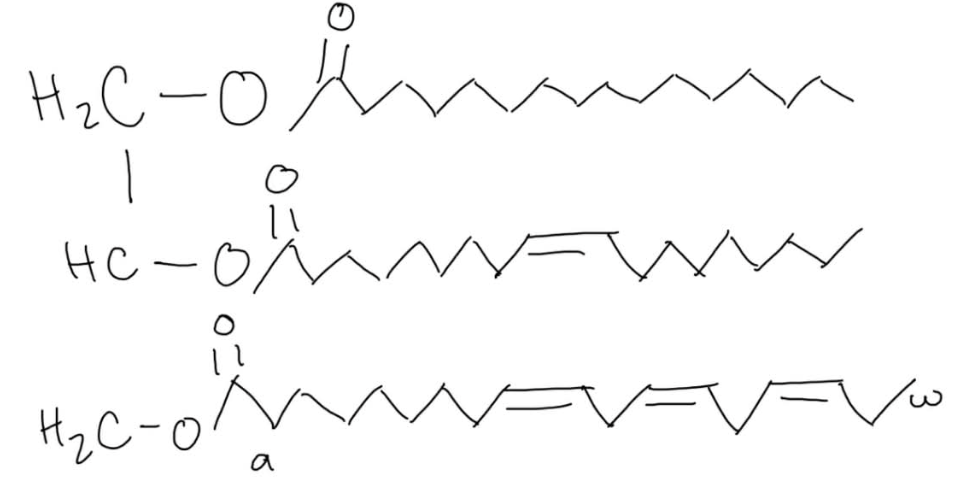
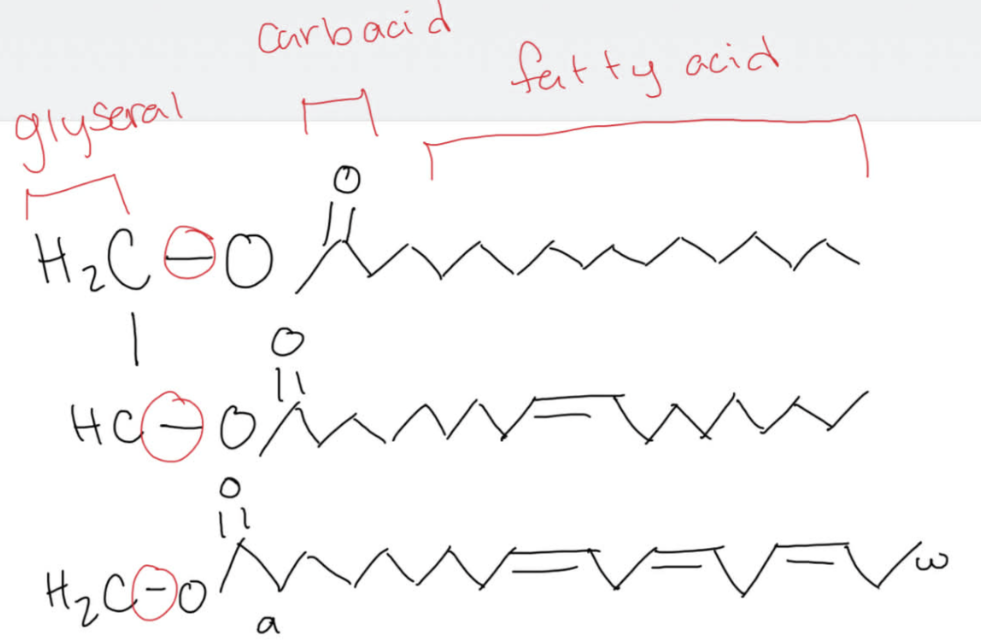
please circle the ester bond in the following triacylglycerol.
(True/False) Carbohydtraes are more enegy rich than fatty acids.
False. fatty acids have more carbon hydrogen bonds 9cal/g
Phospholipids are made up of what components? (Hint: 4 answers)
fatty acids, phosphate, a platform, alcohol
Which lipid is common in nerve cells?
sphingomyelin
What type of lipid aids in cell-to-cell interactions?
glycolipids
What is a common example of a steroid?
Cholesterol
In the cell membrane, what aids the movement of molecule across the membrane?
Proteins
Channel proteins → act like tunnels for small molecules
carrier proteins → grab molecules and carry them
How could the interactions of the cell membrane best be described? What of bonds are they?
Hydrophobic interaction and hydrogen bonds. Hydrophobic tails of the phospholipids. interact w/each other in the membrane, while the hydrophilic heads interact w/the aqueus enviroment and w/each other.
Membrane fluidity can be increased by which of the following?
Increase with (saturation/unsaturation)
Increase with (cis/trans) bonds
Increase with (short/long) chain length
(Increase/decrease) with cholesterol concentration
Increase with (saturation/unsaturation)
Increase with (cis/trans) bonds
Increase with (short/long) chain length
(Increase/decrease) with cholesterol concentration
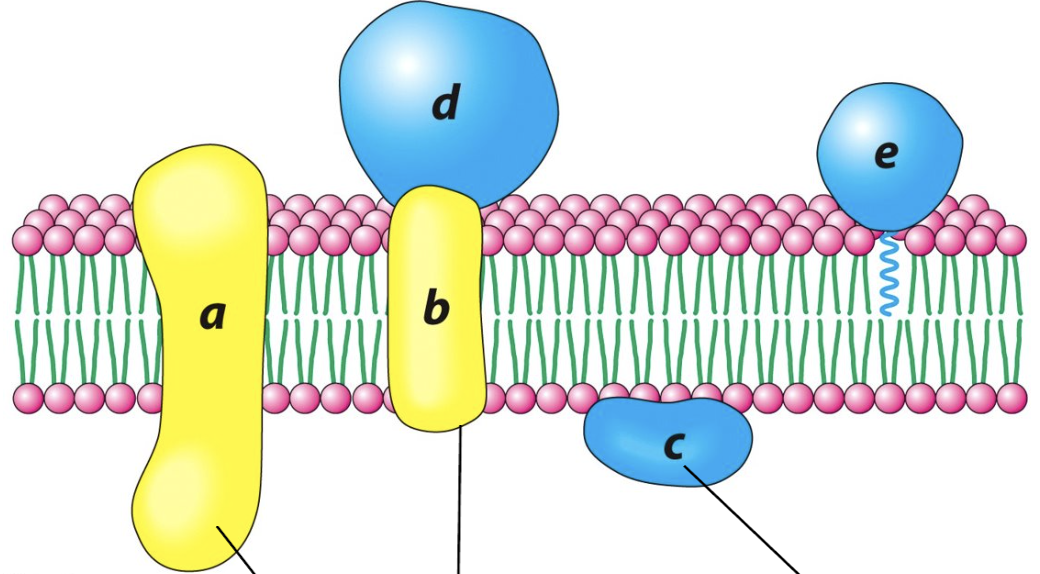
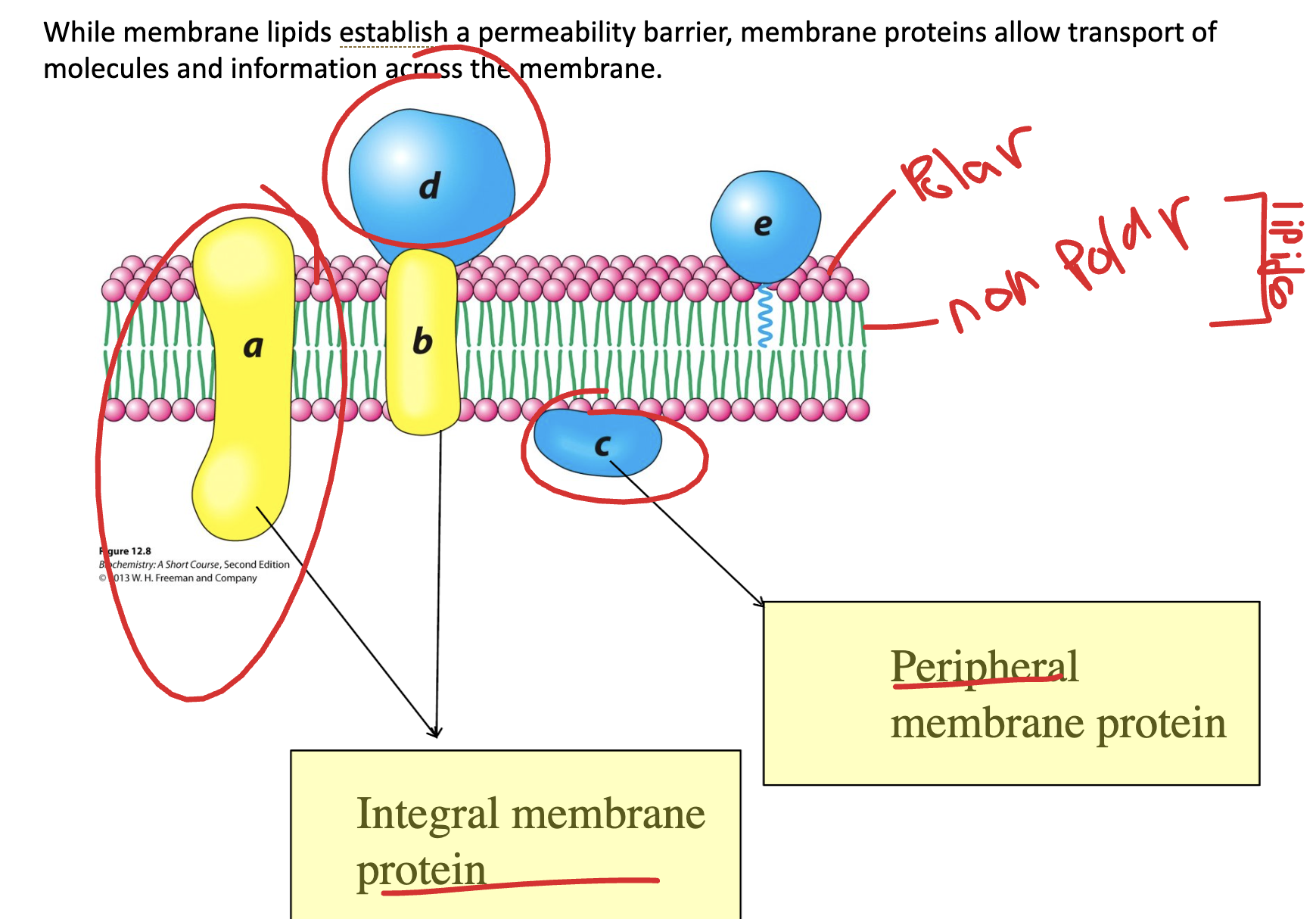
Label the following molecules and their respective function:
(a, b) transport, integral proteins
(c,d,e) perpheral proteins, cell cell communication
What does membrane fluidity directly depend upon?
(High/Low) melting point
low
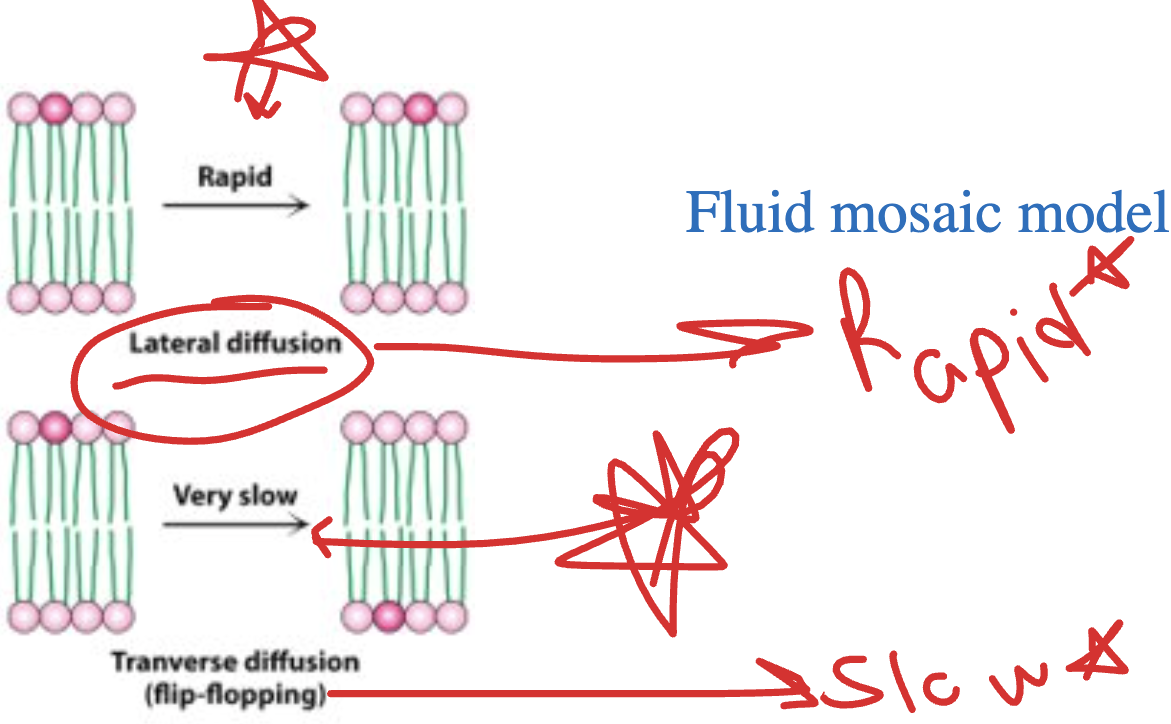
Label the following as either lateral or transverse diffusion:
*he remembers this on exam
Also known as facilitated diffusion, the movement of molecules down their concentration gradient only with the assistance of a channel. ___?___
passive transport
Movement of molecules against their concentration gradient, requiring a source of energy. ____?___
active transport
What two factors determine whether a molecule will cross a membrane?
concentration gradient and solubility
What is the most common example of active transport?
sodium potassium pump
to cross a membrane, without the assistance of a protein, should a molecule be (hydrophilic/hydrophobic)
hydrophobic
These molecules are involved in powering the transport of a molecule against its concentration gradient by grouping together with another molecule moving in the same direction down the gradient. ___?___
antiporters
*antiporter = opposite
*2 molecules in opposite direction
These molecules use one concentration gradient to power the formation of one another. ___?___
secondary transporters
These molecules use one concentration gradient to power the formation of another but move in the opposite direction. ___?___
symporters
*symporters = same both sides
*2 molecules in same direction
(True/False) Potassium channels are selective.
True
A base bound to a sugar is known as a ___?____
nucleoside
*nucleoside = base and sugar
*nucleotide = base, sugar, phosphate