mechanism of nucleophilic substitution
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
carbon halogen bond
polarised due to halogen being more electronegative than carbon…halogen draws electron pair in the bond away from carbon nearer to itself and carbon becomes positive
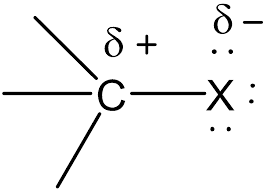
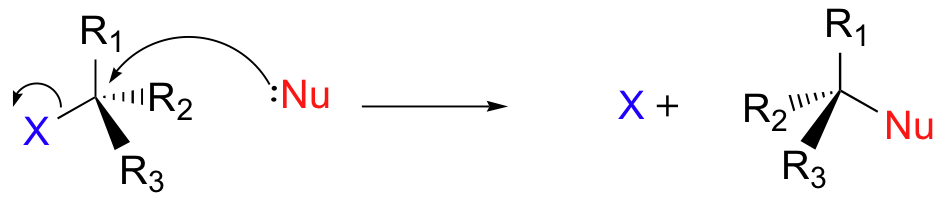
primary halogenoalkane mechanisms (SN2)
steps in a nucelophilic substitution which determines the rate of reaction involves 2 reacting species
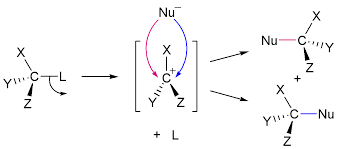
tertiary halogenoalkanes (SN1) mechanisms
steps in nucleophilic substitution which determines the rate of reaction and only involves the organic reactant
carbocation stability
primary are least stable and tertiary is most stable
secondary carbocations
make use of both SN1 and SN2 mechanisms
elimination reactions using NaOH in ethanol
alkene is formed
using aqueous NaOH in a nucleophilic substitution
an alcohol is formed