Chemistry Memorise Deck
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
[Cu(H2O)6]2+
Pale blue solution

Cu(OH)2 (s)
Pale blue precipitate

[Cu(NH3)4(H2O)2]2+
Dark blue solution

CuCl42-
Yellow solution

CoCl42-
Blue solution

[Cr(H2O)6]3+
Pale purple solution, turns green when heated. (Common to appear green in solution due to impurities).

Cr(OH)3 (s)
Dark green precipitate

[Cr(NH3)6]3+
Purple solution

[Cr(OH)6]3-
Dark green solution

[Mn(H2O)6]2+
Pale pink solution

Mn(OH)2 (s)
Pale brown precipitate

[Fe(H2O)6]2+
Pale green solution
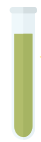
Fe(OH)2 (s)
Dark green precipitate

[Fe(H2O)6]3+
Yellow solution
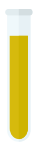
Fe(OH)3 (s)
Orange-brown precipitate

Linear
Bonding pairs: 2
Lone pairs: 0
Bond angle: 180o
Example:
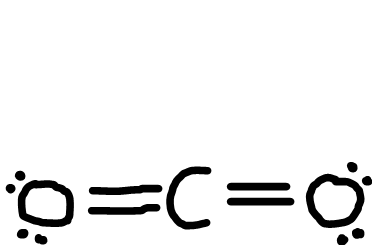
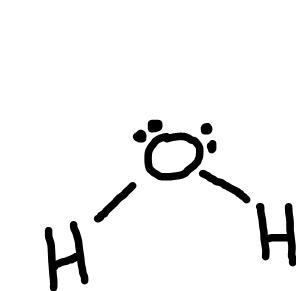
Bent
Bonding pairs: 2
Lone pairs: 2
Bond angle: 104.5o
Example:
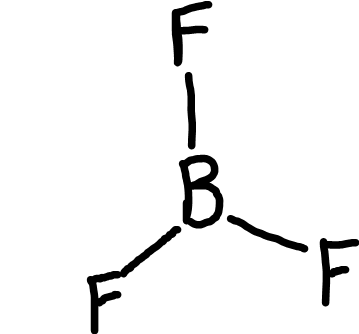
Trigonal planar
Bonding pairs: 3
Lone pairs: 0
Bond angle: 120o
Example:
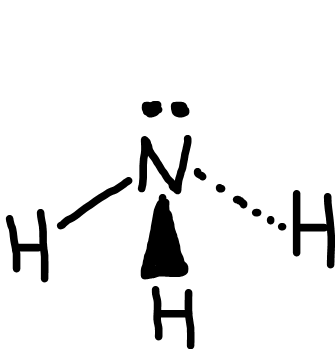
Trigonal pyramidal
Bonding pairs: 3
Lone pairs: 1
Bond angle: 107o
Example:
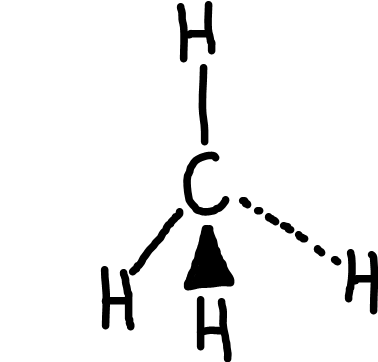
Tetrahedral
Bonding pairs: 4
Lone pairs: 0
Bond angle: 109.5o
Example:
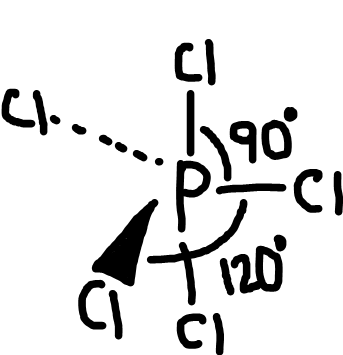
Trigonal bipyramidal
Bonding pairs: 5
Lone pairs: 0
Bond angle: 90o and 120o
Example:
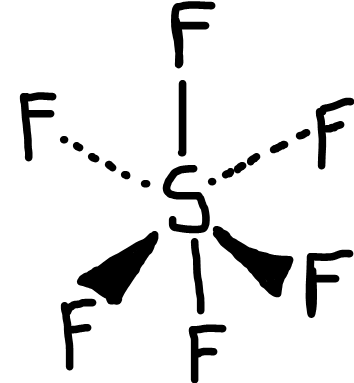
Octahedral
Bonding pairs: 6
Lone pairs: 0
Bond angle: 90o
Example:
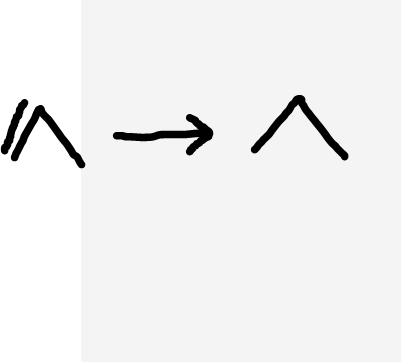
Alkene to alkane
H2(g), Nickle catalyst, approximately 150oC
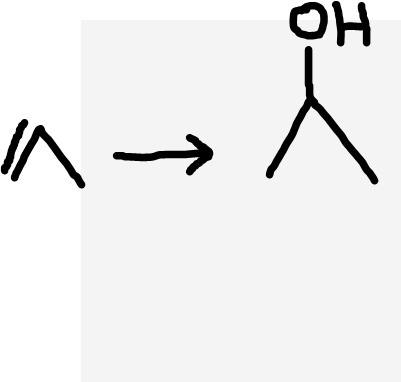
Alkene to alcohol
H2O (g), H3PO4 catalyst
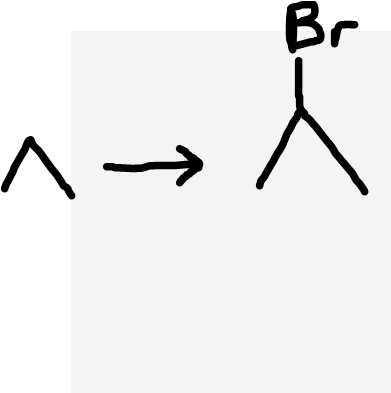
Alkane to haloalkane
Halogen (X2), UV light, heat
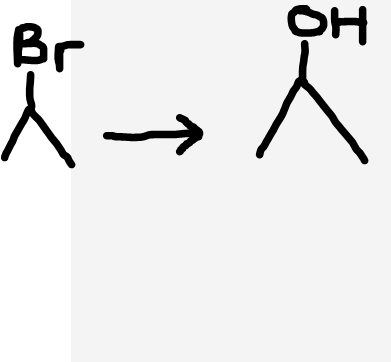
Haloalkane to alcohol
NaOH (aq), heat under reflux
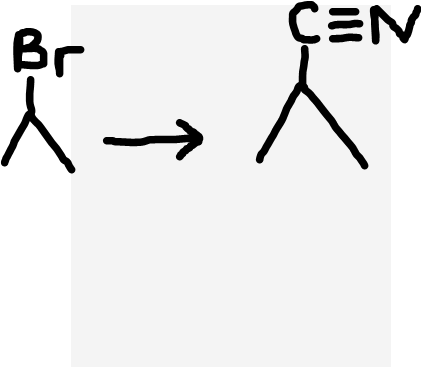
Haloalkane to nitrile
NaCN or KCN (ethanolic), heat under reflux
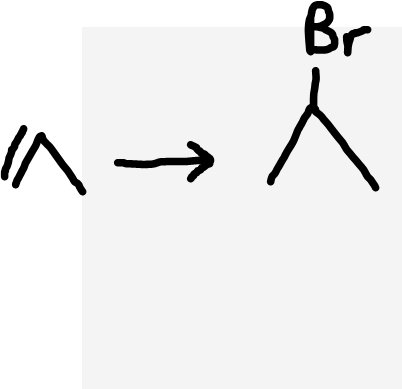
Alkene to haloalkane
Hydrogen halide (HX)
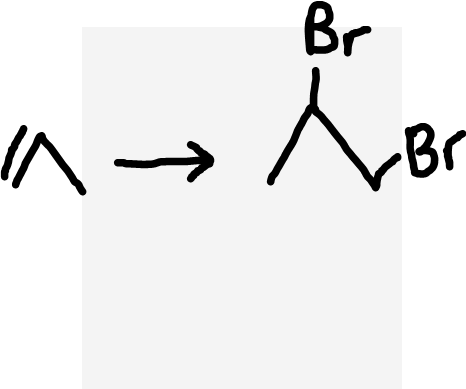
Alkene to dihaloalkane
Halogen molecule (X2)
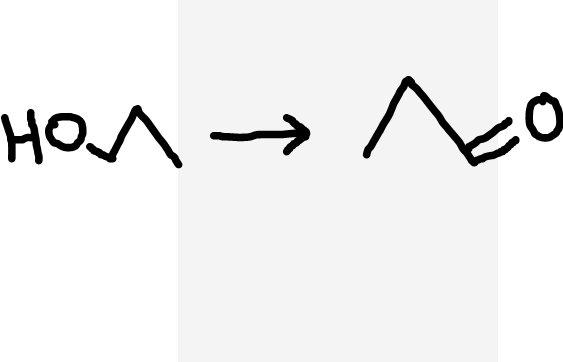
Primary alcohol to aldehyde
Acidified dichromate ions (Cr2O72-/H+), heat and distil immediately
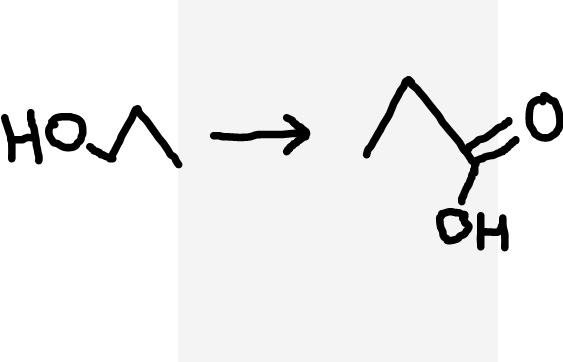
Primary alcohol to carboxylic acid
Acidified dichromate ions (Cr2O72-/H+), heat under reflux
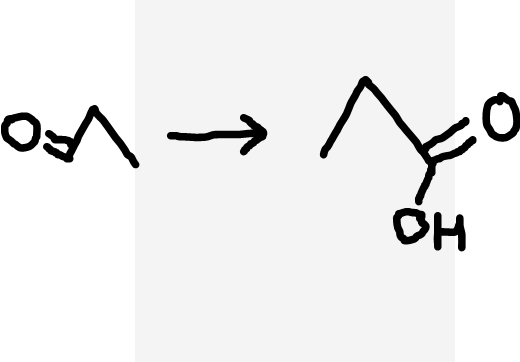
Aldehyde to carboxylic acid
Acidified dichromate ions (Cr2O72-/H+), heat under reflux
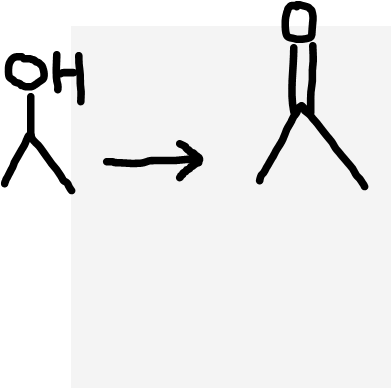
Secondary alcohol to ketone
Acidified dichromate ions (Cr2O72-/H+), heat under reflux
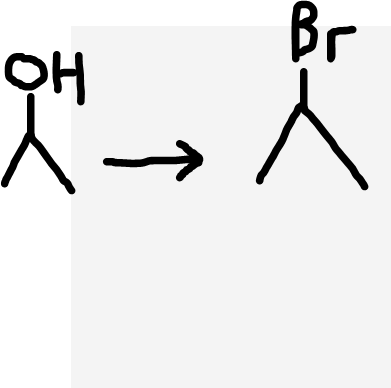
Alcohol to haloalkane
Concentrated H2SO4, sodium halide (NaX), warm temperature for distillation
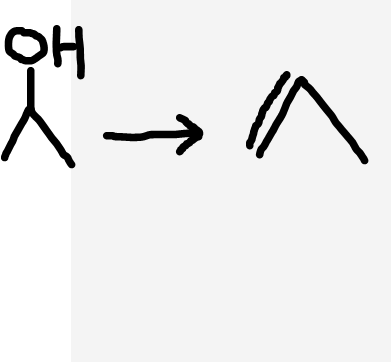
Alcohol to alkene
Heat, acid catalyst (H3PO4/H2SO4)
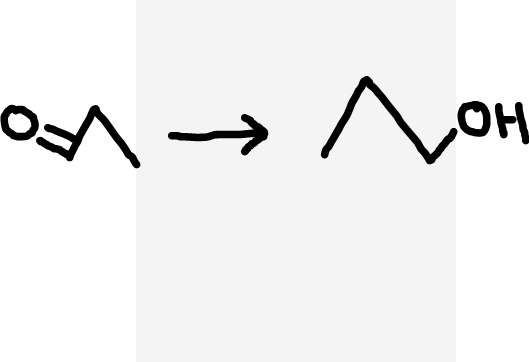
Aldehyde to primary alcohol
NaBH4
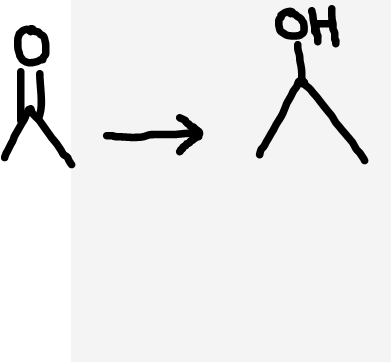
Ketone to secondary alcohol
NaBH4 (Not aldehyde to primary alcohol)
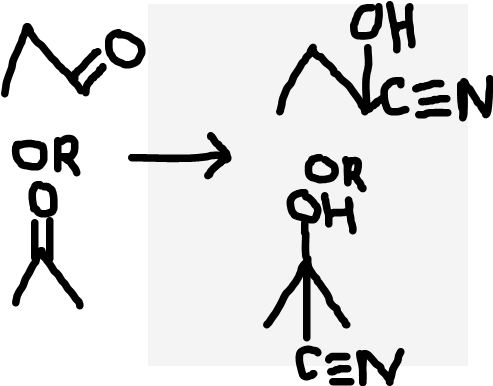
Aldehyde/Ketone to hydroxynitrile
Acidified NaCN (normally H2SO4)
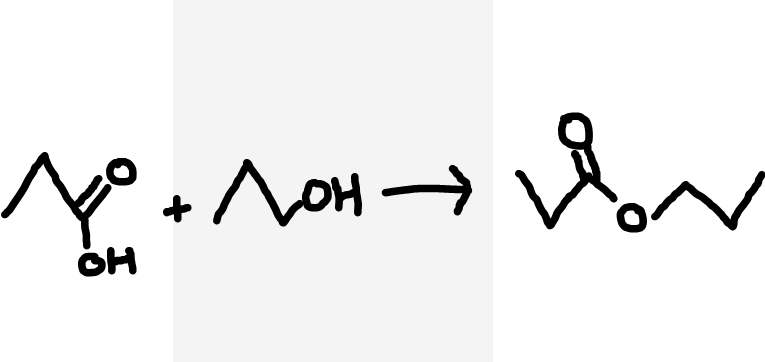
Carboxylic acid and alcohol to ester
Heat under reflux, concentrated H2SO4 catalyst
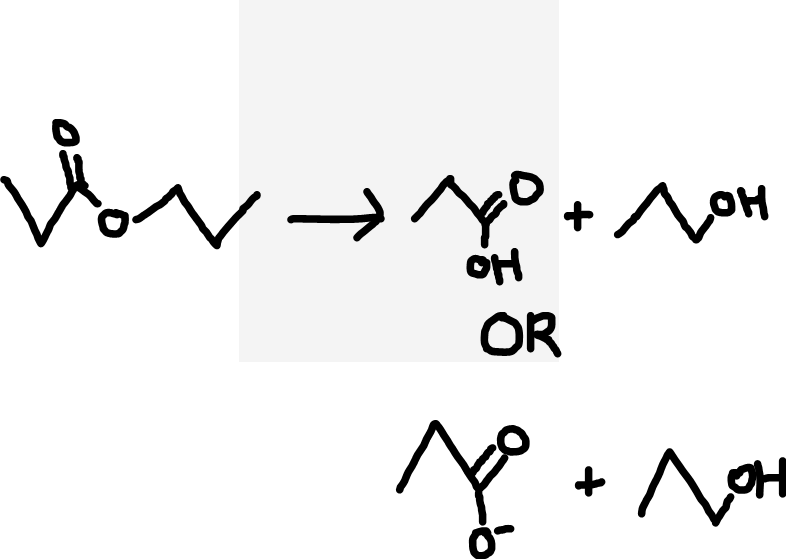
Ester to carboxylic acid and alcohol (acid hydrolysis) or carboxylate ion and alcohol (alkaline hydrolysis)
Warm, aqueous (dilute acid/alkali)
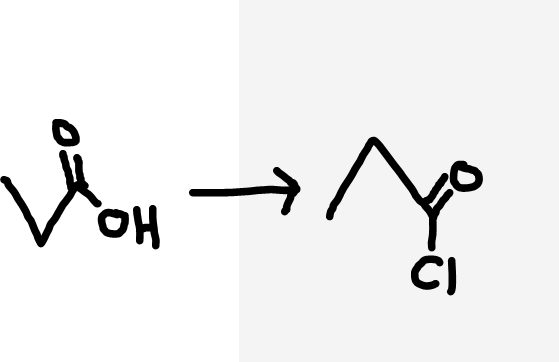
Carboxylic acid to acyl chloride
Neat (pure liquid) SOCl2
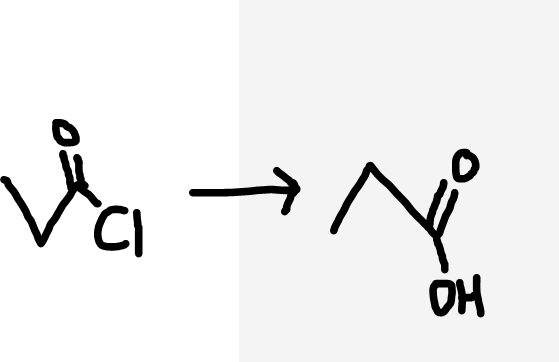
Acyl chloride to carboxylic acid
H2O
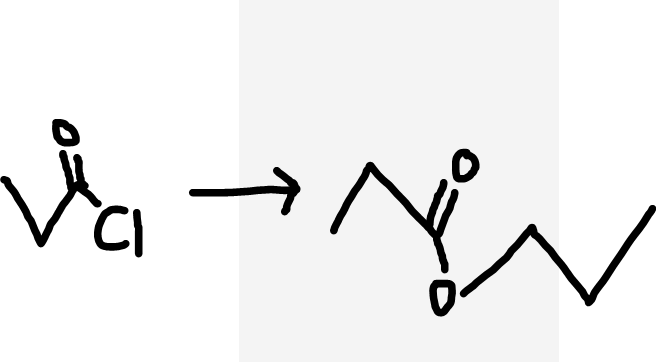
Acyl chloride to ester
Alcohol
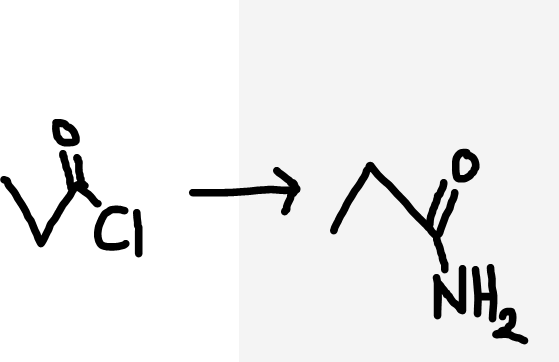
Acyl chloride to primary amide
NH3
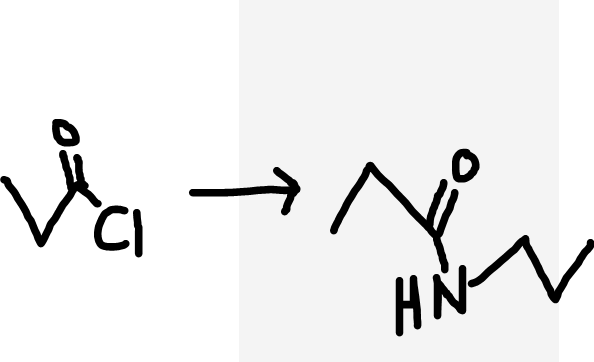
Acyl chloride to secondary amide
Primary amine (reactant)
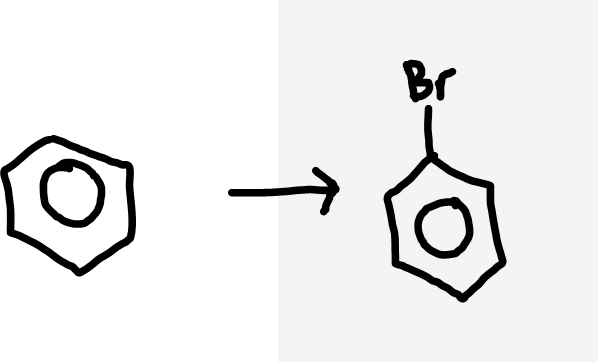
Benzene to halobenzene
Halogen molecule (X2), halogen carrier (AlX3/FeX3)
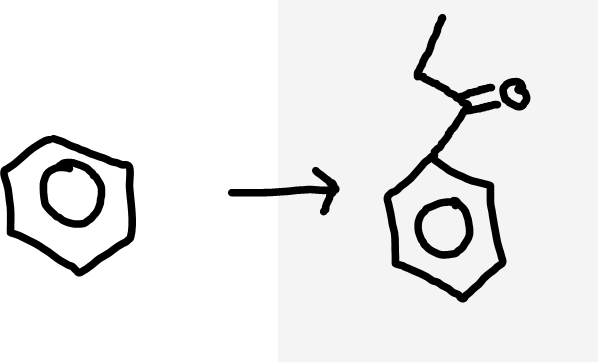
Benzene to aromatic ketone
Acyl chloride, AlCl3 catalyst, warm
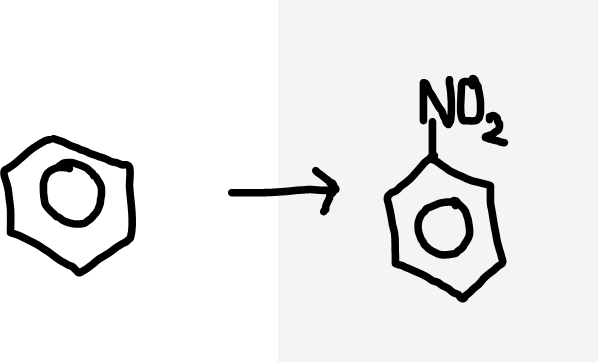
Benzene to nitrobenzene
concentrated HNO3 and concentrated H2SO4 catalyst
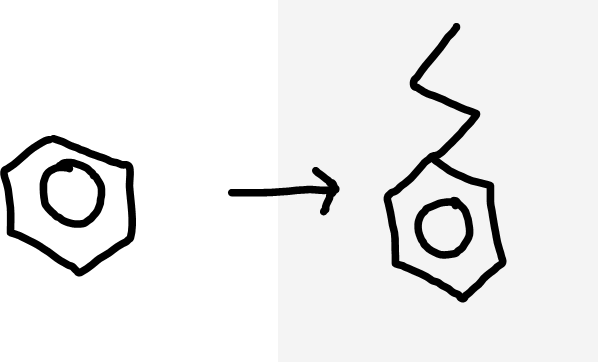
Benzene to alkylbenzene
Haloalkane, halogen carrier (AlX3) catalyst, warm
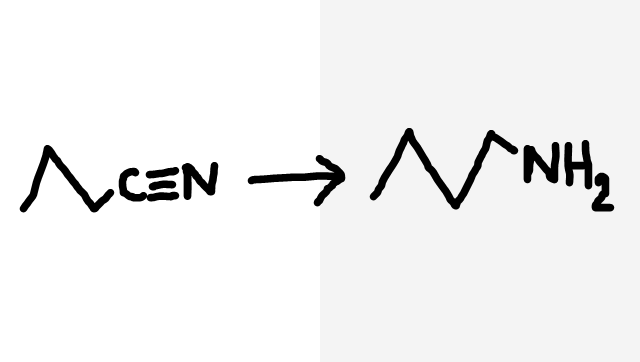
Nitrile to primary amine
H2(g), nickel catalyst (not alkene to alkane)
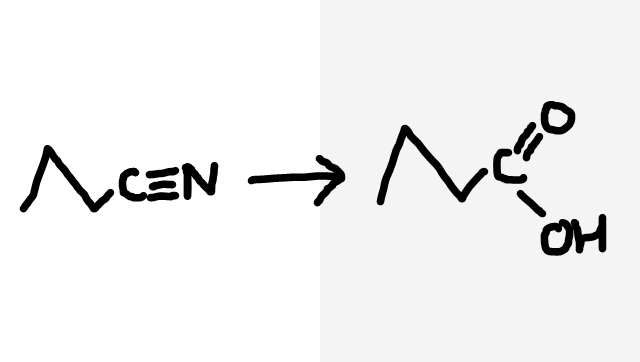
Nitrile to carboxylic acid
Dilute HCl, heat under reflux
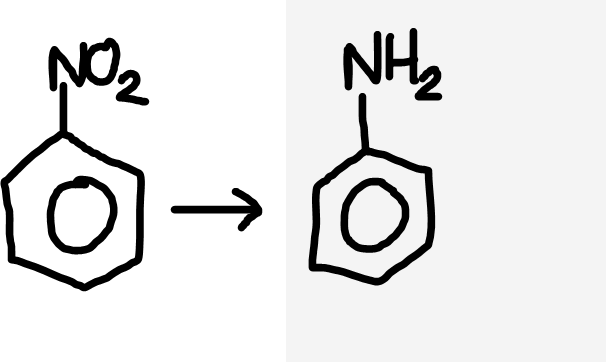
Nitrobenzene to aromatic amine
Concentrated HCl and Sn catalyst
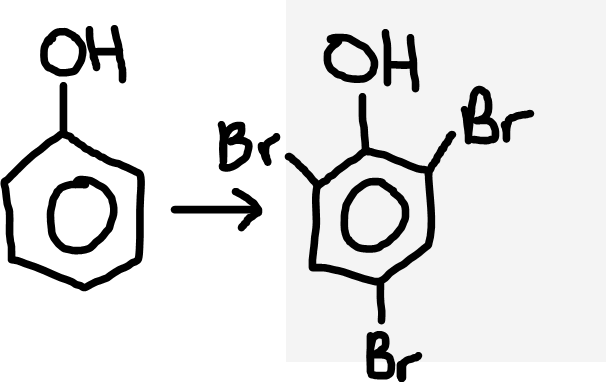
Phenol to bromophenol
Bromine water (Br2 (aq))
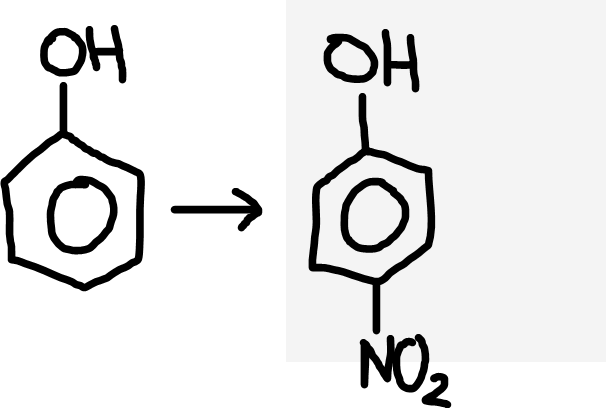
Phenol to nitrophenol
Dilute nitric acid (HNO3)