therapeutic ultrasound and safety
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
what are the effects of ultrasound
thermal effect
bubble and cavitation effects
radiation effects
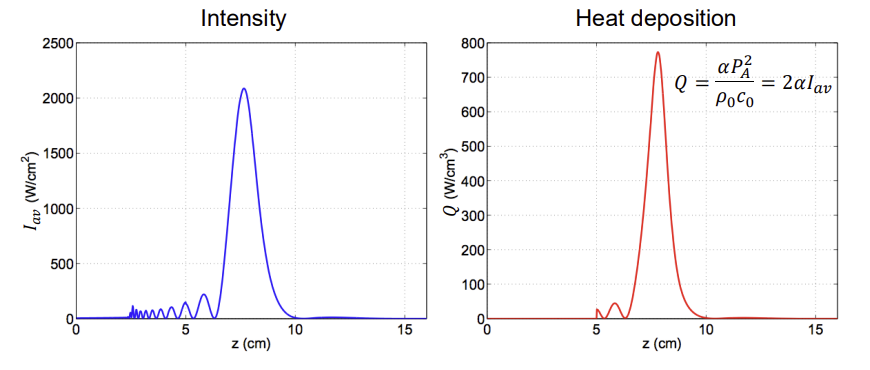
what is the volume rate of heat deposition? units?
Q=ρoCoaPA2(x) W.m−3 see notes for derivation
how does rate of heat deposition behave
it is proportional to the absorption coefficient and the square of the local pressure amplitude PA=Poe−ax
after the probe is turned off:
heating decreases due to diffusion of heat via conduction and tissue perfusion
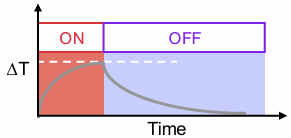
what happens if temperature deposition is larger than it’s decrease?
Δ=ρoCoCaPA2(x)t where C is the specific heat capacity(J/Kg/C)
factor affecting heating
absorption coefficient of the tissue
frequency: increased absorption at higher frequencies → more heating
non-linear effects: harmonics moves energy to a higher frequency → more heating
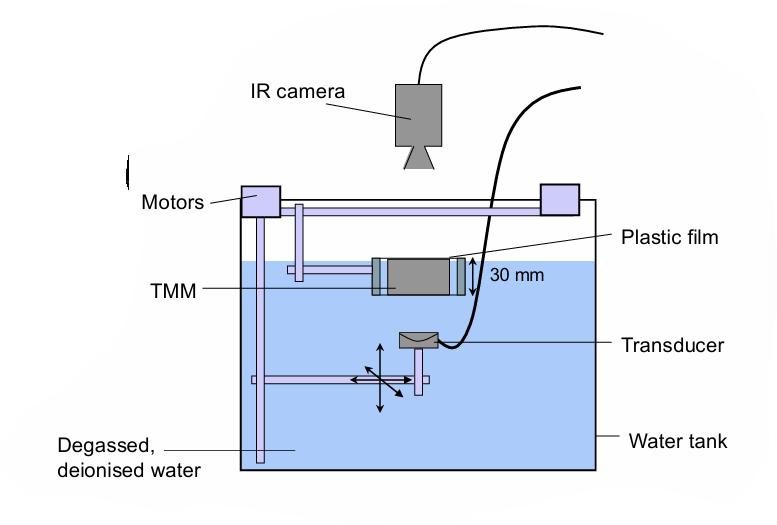
how is ultrasound heating mapped?
using infrared imaging in a water tank
what does heat damage to cells depend on
temperature
exposure time
what are the heat thresholds for tissue damage?
C < 40 → no irreversible damage
C > 40 → protein denaturing and loss of function
c >65 → collagen fibres shrink and tissue coagulates
what is thermal dose
measured in cumulative equivalent minutes (CEM 43C or t43 )
→ it is the number of minutes required to achieve the same effect as heating at 43C
→ allows us to compare the thermal dose delivered at different temperatures
t43=∫0tR43−TdT
where:
t: is time in minutes
T: is temperature in celsius
R = 0.5 when T > 43, R = 0.25 when T < 43
if temp is constant: t43=tR43−T
what is the cumulative equivalent minutes required for cell death
t43=240minutes
for every degree above 43C the time to effect is halved
heating is not uniform
thermal dose required to achieve cell death is dependent on tissue and cell type
muscle/fat → GI stuff → liver→ brain →kidneys
what is thermo-tolerance
→ if tissue is heated bellow 43 tissue becomes more tolerant to heat
due to upregulation of heat shock proteins → increases resistance to thermal toxicity
this effect is retained even after heating is stopped
what is thermal index
measure of potential tissue heating: TI=PdegreesPo
where:
P_o: time averaged acoustic power (Calibrated based on measurements)
P_degrees: power required to raise tissue temp by 1 degree → depends on frequency and tissue type
how are exposure limits set
thermal dose is cumulative → timing is kept as low as reasonably achievable (ALARA)
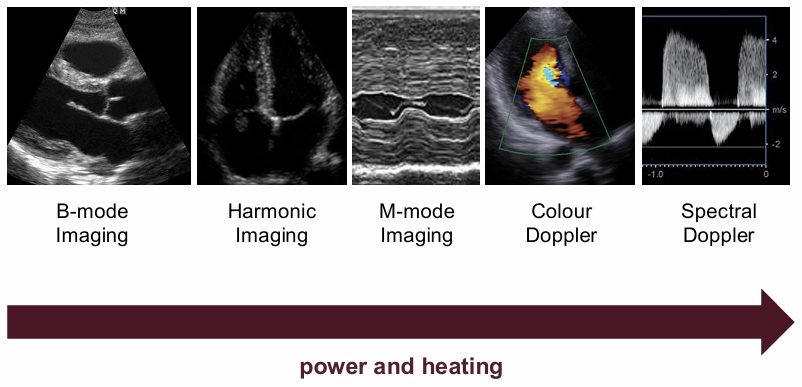
TI of different imaging modes?
B-mode: short pulses with low duty cycles and moderate power
Harmonic imaging: uses non-linear harmonic -> higher frequencies, also requires higher transmit power
M-mode imaging: the same line is fired repeatedly causing energy to concentrate in one region -> tissue doesn't get time to cool
Colour doppler: sends multiple pulses per line with longer pulse times and higher average power
Spectral doppler: continuous or near continuous transmission with high duty cycles
duty → how long a pulse is on
what is acoustic cavitation
formation and activity of gas bubble or vapour due to a medium being exposed to an acoustic wave
what are the types of cavitation
stable cavitation
inertial/transient cavitation
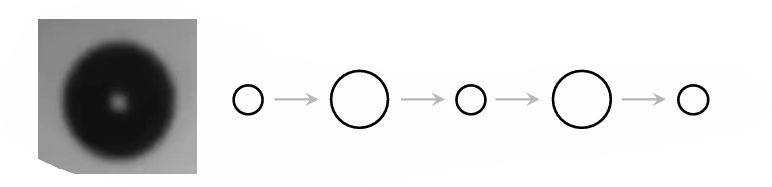
explain stable cavitation
acoustic pressure variation causes the bubble to oscillate indefinitely
at low pressure → bubble expands
at high pressure → bubble shrinks
stiffness of the bubble gas and inertia of the surrounding medium and bubble shell surface tension are balance
stiffness of gas → prevents bubble from shrinking too much
inertia of medium and surface tension from shell → prevents bubble from expanding too much and bursting
what frequency and bubble radius does stable cavitation occur
→ radius (R0 ) > 10 micrometre
→ minnaert resonance frequency: fo=Ro3 in water and atmospheric pressure
what is inertial/transient cavitation
bubble grows and collapses violently
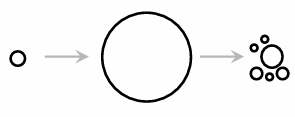
when and how does collapse occur in inertial/transient cavitation
caused by rectified diffusion:
at high pressure phase: bubble is pushed together → SA decreases → high pressure inside the bubble → less diffusion of of gas out of the bubble
at low pressure phase: gas in bubble is pulled apart → SA increases → less pressure inside the bubble → more diffusion of gas in the bubble → bubble grows
total pressure of fluid surrounding the bubble =ambient fluid pressure(PO)+acoustic pressure(P)
low pressure phase of wave → P is -ve → if P>P_O then P_T<0 → fluid is in tension → bubble collapses
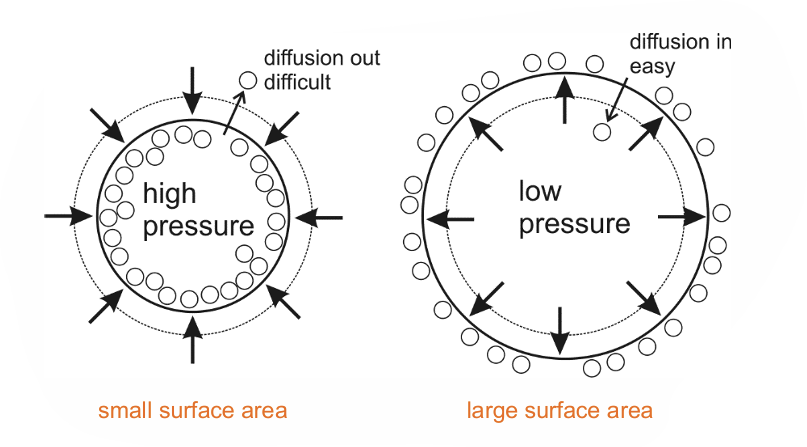
intertial/transient cavitation factors
frequency: bubble more likely to collapse at low frequency → wave spends more time in low pressure phase → bubble grows more rapidly
bubble size: larger bubble grow more slowly as there is less of a pressure difference
cavitation nuclei
acoustic pressure
cavitation outcomes (10)
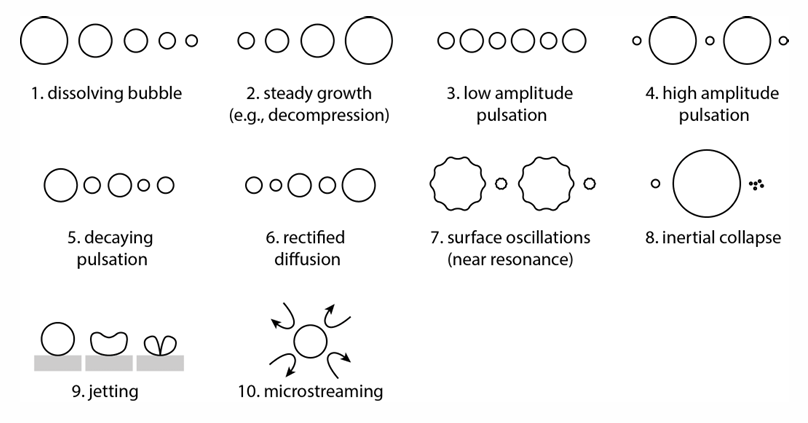
what are the tissue damaging effects of cavitation
bulk heating:
→ bubble motion causes viscous heating
→ non-linear scattering of ultrasound → increases absorption
→ bubble collapse → broadband and shockwave emission
mechanical action: bubble collapse causes jetting and microstreaming
chemical action: collapse generates high heat forming free radicals and sonochemicals
lead to cell lysis due to membrane rupturing + structural/functional change to cell and DNA
what is bubble rebounding
after collapse an unconstrained (not near a boundary) bubble might rebound producing oscillations at the resonant frequency of the bubble
what is sonoluminescence
collapse causes high heat → heats gas → producing a light pulse of ionised plasma
what damage does jetting cause?
vessel distension
vessel invagination
vessel hemorrhaging
what is the mechanical index?
measure of potential for inertial cavitation
MI=fPr
where:
P_R: is the peak -ve pressure in Mpa
f: centre frequency of the US wave
it is not time dependent as it is a threshold effect
what is the radiation effect
ultrasound hits a boundary and gives up it’s momentum to the boundary
for the wave to be reflected by the wall it must generate twice the force (to stop then to throw it back)
F^V=-<P_T\frac{DU}{Dt}>=-\frac{1}{c_0}\frac{dI_{av}}{dx}
p_t: is the total acoustic pressure
derivative of u: is particle acceleration
<>: averaged over time
how is the radiation effect related to the heating effect
−c01dxdIav=coQ=ρoCo2aPA2(x) → force is proportional to heat absorption
force required to absorb = CoPT
force required to reflect = Co2PT
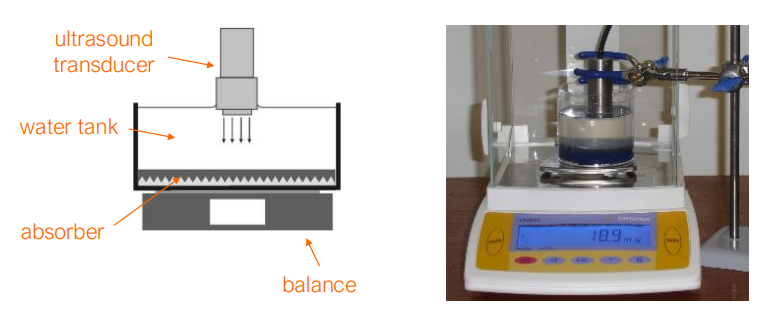
how is radiation force measured?
probe places inside a water tank with deionized and degassed water on top of a sensitive scale
weight measured before and after the transducer is turned on
F = m * g
also used to measure time averaged acoustic power for TI index
what are the uses of radiation effect
particle streaming
particle trapping: traps a particle between two high pressure areas?
what is used for mechanical therapeutic ultrasound
Low power low frequency US + addition of microbubbles
what are the uses of mechanical therapeutic US
Sonothrombolysis
bone healing
drug delivery + crossing BBB → Mechanical effects alter membrane permeability
neuromodulation → 250-500KHz low frequency and intensity → alters ion channel gating
what type of US is used for thermo-mechanical therapy
high power US waves
what are the uses of thermo-mechanical therapy
lithotripsy
HIFU
histotripsy
haemostasis
Explain HIFU
High intensity focused ultrasound → acoustic energy is absorbed causing tissue to heat up + large pressure oscillation causes inertial cavitation
what type of US trasnducer is used for HIFU and why?
uses a large area, bowl shaped single element or phased array transducer → high focusing gain + small focus
produces very high heating at the focus
regions above and below focus are unharmed → trackless therapy
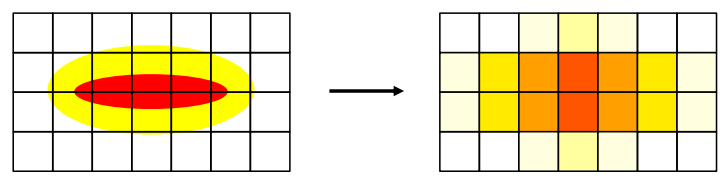
what is the area of the HIFU lesion?
how is this limitation overcome?
→ produces a small 2×10mm focal lesion
→for large areas multiple sonications or a spiral scanning pattern is used
how does cavitation enhance the heating effect
inertial cavitation → mechanical tissue damage
bubble collapse releases broadband shockwaves → energy is absorbed and converted to heat
movement of the bubble shell → viscous heating
non-linear effects → energy is scattered → increased absorption
collapse leaves microbubble → scatters and traps US
control of cavitation vs thermal effects
thermal effects are much easier to control than cavitation as bubble collapse leads to various knock-on effects that are hard to quantify
→ sound is reflected back towards transducer → tadpole shaped lesions
→ produces larger legions in the same treatment time

energy vs average power
for the same average power:
→ low energy for longer time → produces smaller well controlled lesions
→ high energy shorter time → produces a larger lesion due to cavitation effect

how is HIFU treatment monitored?
using MR or US monitoring
MR:
Gold standard
allows for mapping temperature elevations and thermal dose
treatment tries to avoid cavitation
US:
low cost and portable
high frame rate → image organ motion
allows mapping of changes in backscatter and mechanical properties
treatment tries to induce cavitation as it improves contrast
why is treatment monitoring required
target the treatment zone precisely → allows us to calibrate the focus
monitor tissue response in real time to therapy
ensure that sensitive- at risk structures are not in the beam path
explain MR thermometry
measures the shift in proton-resonance frequency due to temp changes
resonant frequency is determined by local magnetic field:
ω=2πy(1−S)Bo
where:
w: resonant frequency
s: shielding constant
B: magnetic flux
how does heating change proton-resonance frequency
heating → strength of hydrogen bonds in water weaken → electrons donated from H to O is pulled back to the hydrogen → increased nucleus screening → resonant frequency decreases
how is proton-resonant frequency measured
shift is measured using gradient-recalled echo → change in frequency causes MR signal to accumulate phase difference over time proportional to temp induced PRF
ΔT=α⋅γ⋅TE⋅BoΔϕ
where:
Δϕ : phase difference
a: thermal coefficient → depends on tissue
y: gyromagnetic ratio → conversion of magnetic field to resonance frequency
Te: echo time (time between excitation and signal readout)
Bo: magnetic field strength
how is MR monitoring callibrated
low-power sonification is done to check focal position
corrections are made for misalignment and phase aberration
phase aberration → changes in phase not due to temperature ex: motion, scanner drift, field changes
what are the drawbacks of MR monitoring (5)
need MR compliant anaesthesia and US equipment
insensitive to bone and fat due to low water content
temp is given relative to the initial phase → assumed to be homogenous and at body temp
MR has low frame rate → depends on sequencing and voxel size
voxel size causes temperature to be averaged spatially which causes underestimation of temp change
explain ultrasound cavitation monitoring
requires cavitation effect → cannot monitor thermal only lesions
coagulative necrosis and bubble formation changes US backscatter
gas has low impedance → increased backscatter → hyperechoic region
region fades a few minutes after HIFU exposure
explain US thermometry
temperature changes sound speed → apparent shift in medium position
thermal expansion of medium → produces actual shift in medium position
causes shift in speckle pattern
ΔT=kdxdD
where:
k: tissue dependent parameter → depends on thermal expansion coefficient and how sound speed changes with temp
D:displacement
drawbacks with US monitoring
B mode imaging: requires cavitation to see hyperechoic region → difficult to see tumour margin
thermometry:
very sensitive to motion artefacts
requires knowledge of tissue dependent parameters
only works for low temp elevations as changes in sound speed is only linear over a limited range
application of HIFU
prostate cancer → HIFU rectal or urethral probe at 4MHz → MR or US
uterine fibroid → HIFU 1MHz → MR or US
transcranial → HIFU 0.6MHz → MR
HIFU vs conventional surgery
pros:
non-invasive → no scarring as mechanical and thermal effects occur mostly at the focus
reduced healing time
reduced risk for infection
cons:
difficult targeting moving organs
limited access to regions shielded by bone or gas
what parameters are controlled to influence either mechanical or thermal effects
duty cycle:
continuous/high duty → favours heat accumulation
low duty/pulsed → favours mechanical effects
intensity/pressure amplitude → high amplitude favoures inertial cavitation
frequency:
low frequency → inertial cavitation
high frequency → increased absorption → increased heating
how is tissue damage avoided in diagnostic imaging
used low intensity
pulse the US beam → low duty cycles prevent heat buildup
monitor MI and TI and keep the ALARA