Chapter 21 and 22 Reagents
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
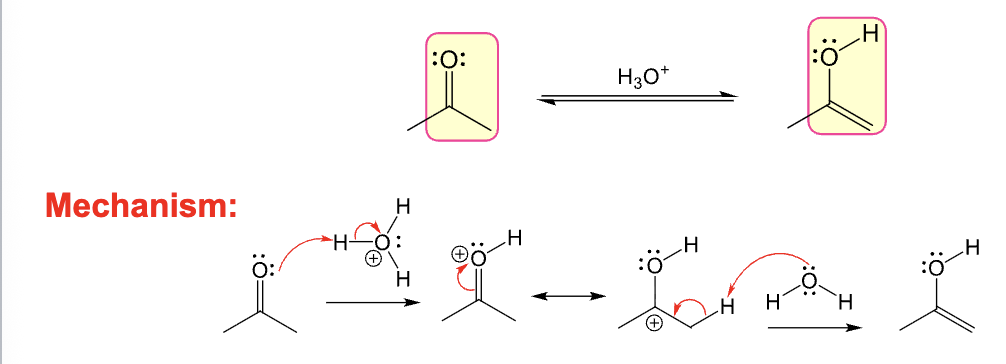
Provide the function for the Reagent: H3O+
Used to acid-catalyzed keto-enol tautomerization
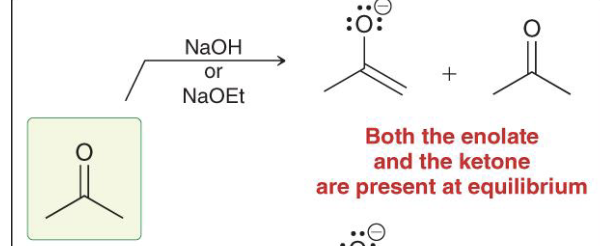
Provide the function for the Reagent: NaOH or NaOR
Used to form a reversible enolate (carbonyl and enolate are at equilibrium)
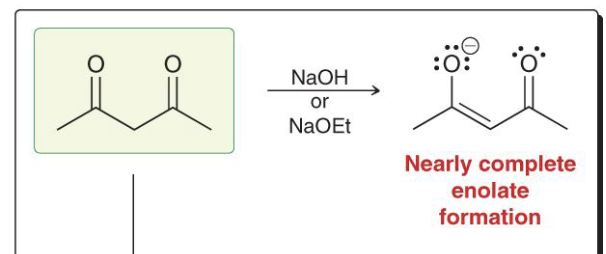
-Nearly complete enolate formed when protons are alpha to two carbonyl groups
Provide the function for the Reagent: LDA or NaH
Forms irreversible enolate
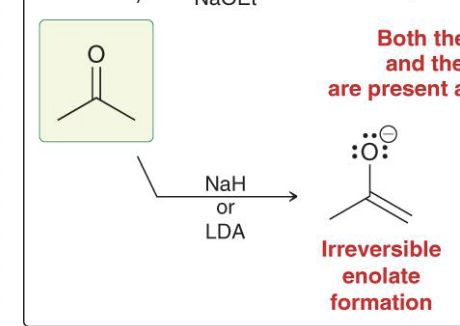
Provide the function for the Reagent: [H3O+], X2
Alpha halogenation in acidic conditions
Adds to more substituted alpha position of ketone or aldehyde
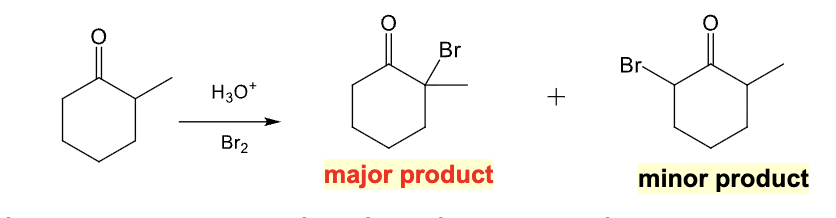
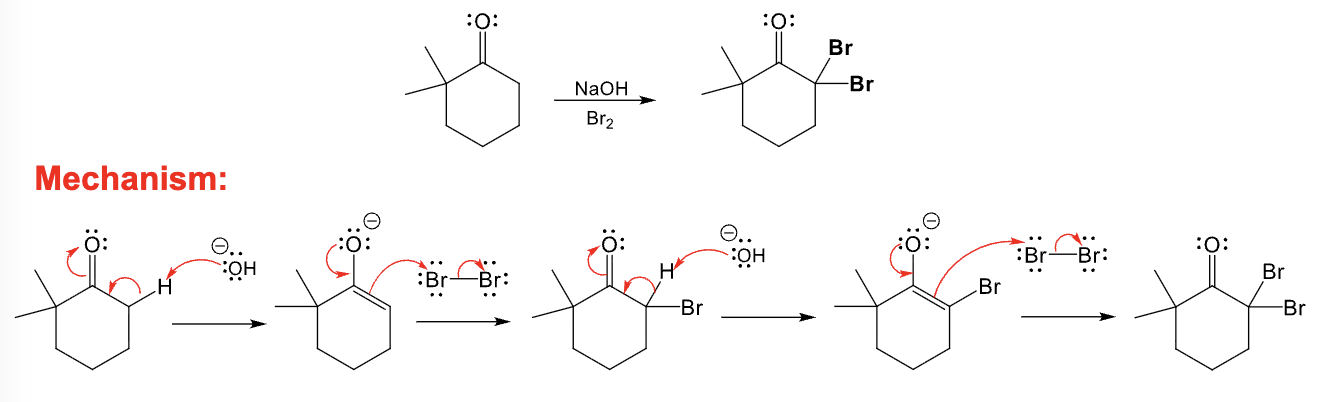
Provide the function for the Reagent:
NaOH, Br2
Alpha halogenation in basic conditions
Multiple halogenations occur
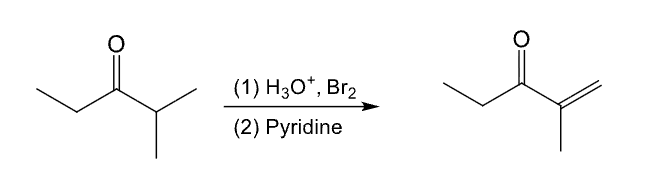
Provide the function for the Reagent:
[H3O+], X2
Pyridine
Used to convert ketone or aldehyde to a ⍺,β-unsaturated ketone or aldehyde
Provide the function for the Reagent:
xs NaOH, xs X2
H3O+
Multiple halogenations occur
Used to convert a methyl ketone into a carboxylic acid
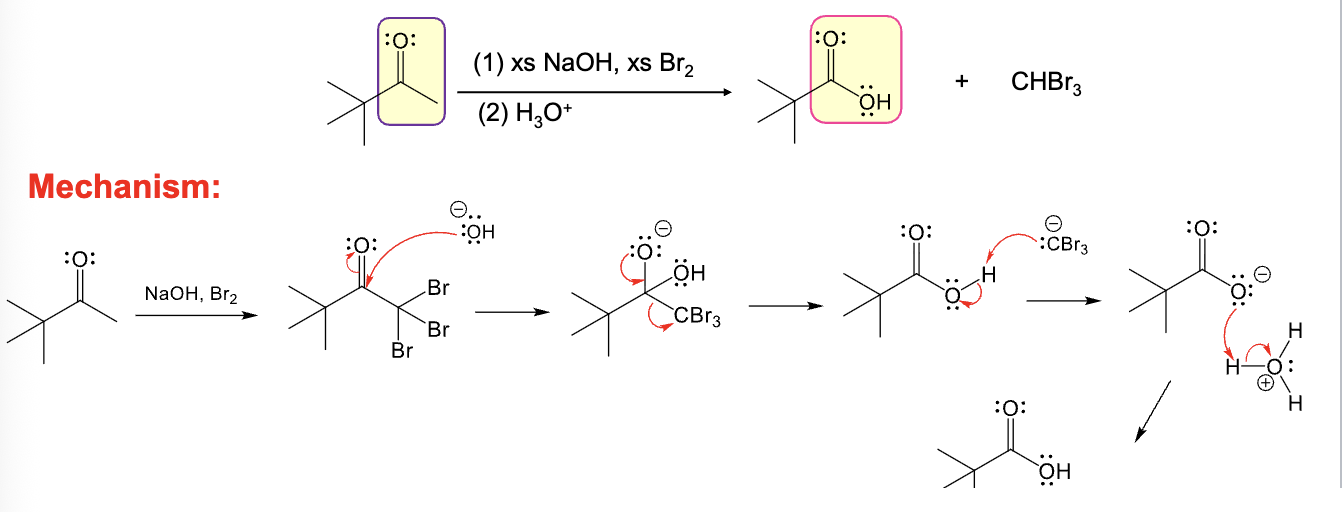
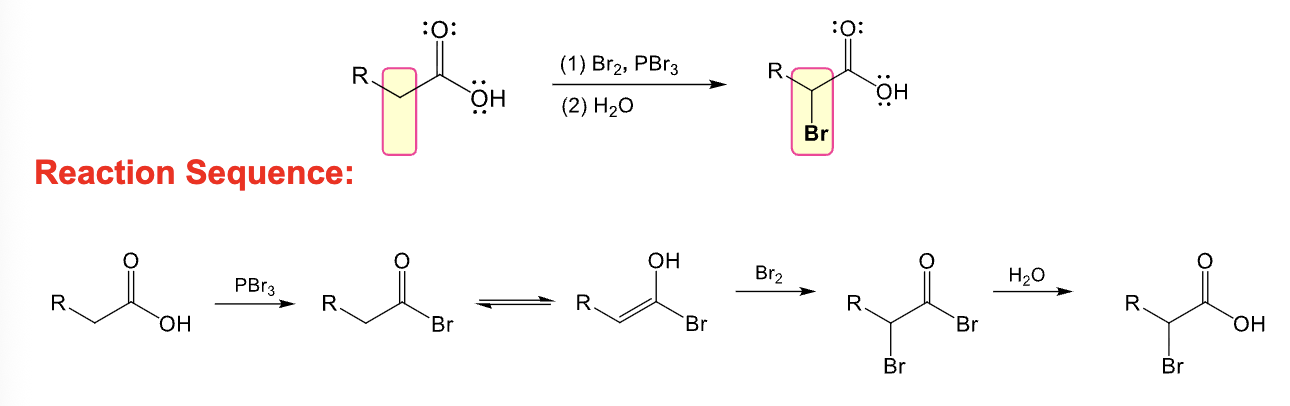
Provide the function for the Reagent:
Br2, PBr3
H2O
Hell-Volhard-Zelinsky reaction occurs
Adds a bromine atom at alpha position of a carboxylic acid
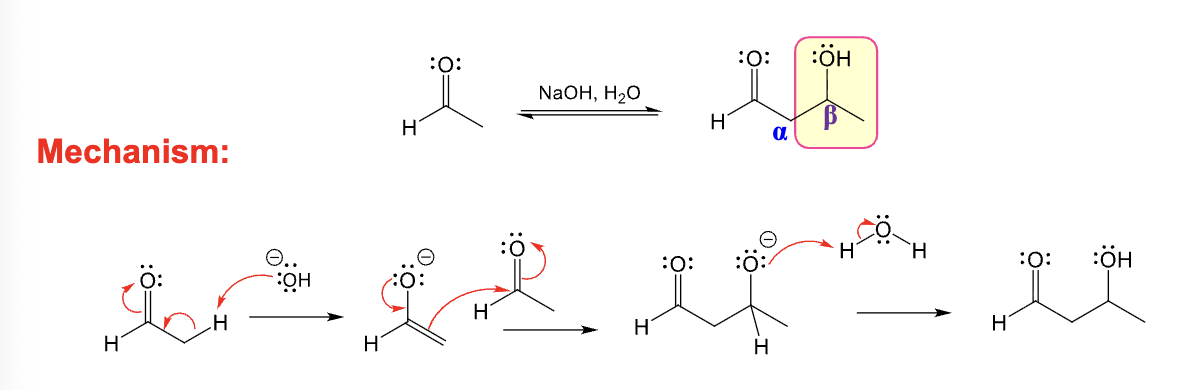
Provide the function for the Reagent: NaOH, H2O
Adol addition occurs:
Two of either the same ketone or aldehyde react together
or
Two different aldehydes and/or ketones are crossed
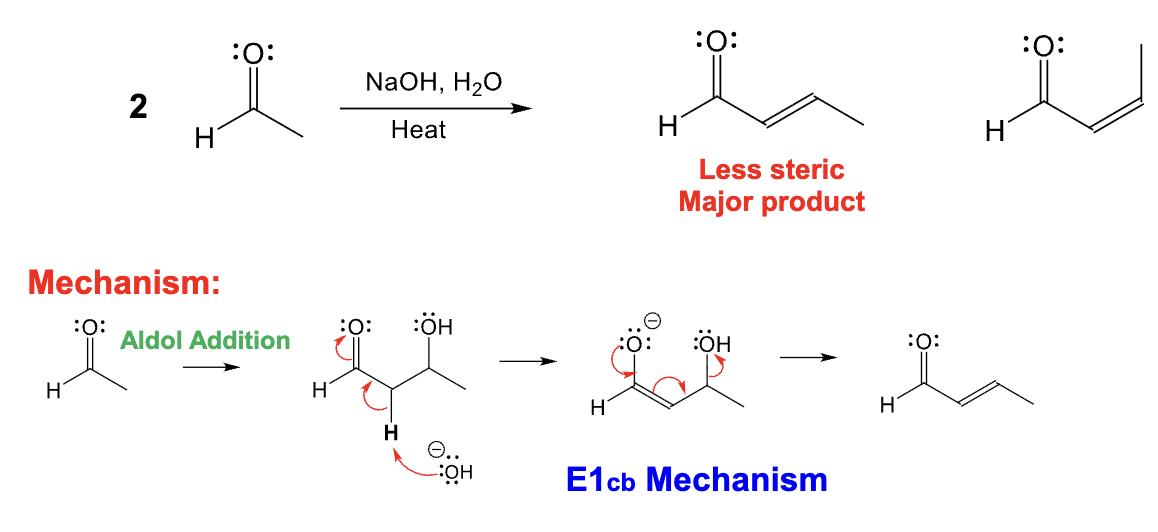
Provide the function for the Reagent: NaOH, H2O, Heat
Adol condensation reaction occurs
Provide the function for the Reagent:
NaOR
H3O+
Used in Claisen Condensation
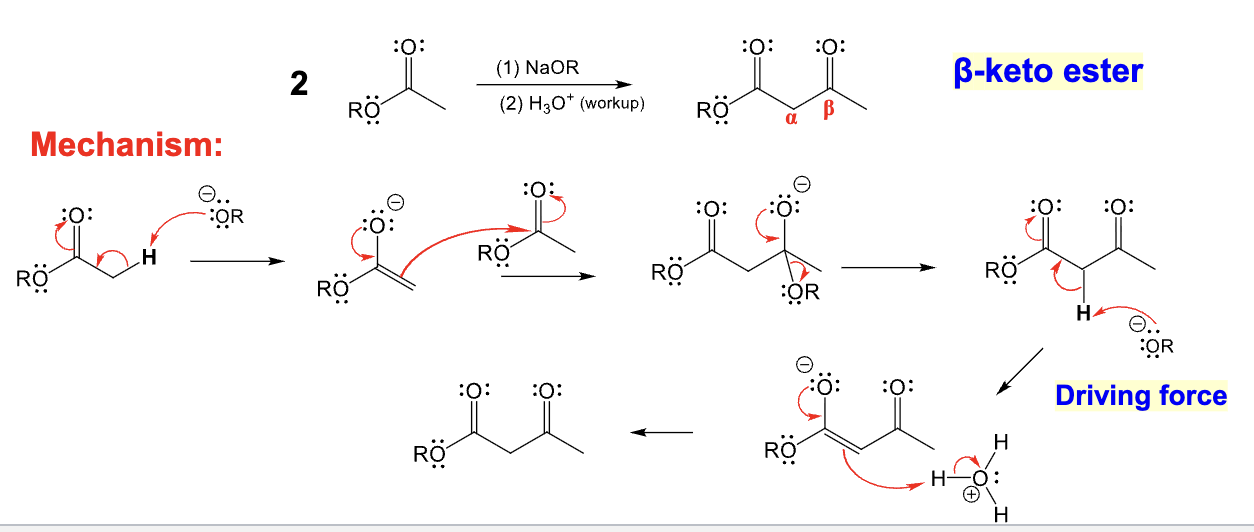
Provide the function for the Reagent:
LDA
Aldehyde or Ketone
H3O+
Used for directed adol reaction between two different aldehydes and/or ketones
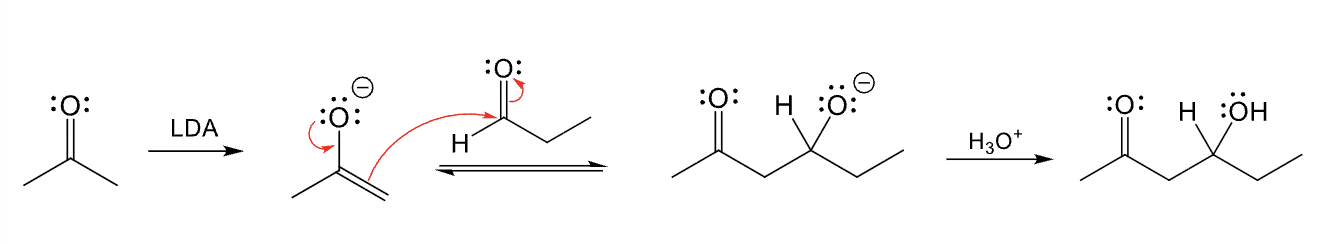
Provide the function for the Reagent:
LDA
Ester
H3O+ (workup)
Used for directed adol reaction between two different esters
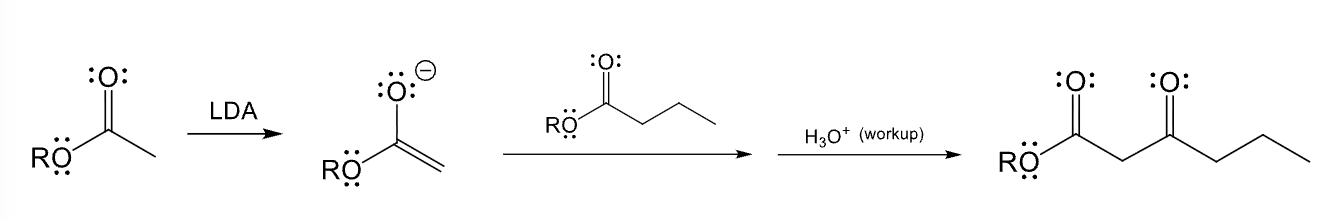
Provide the function for the Reagent:
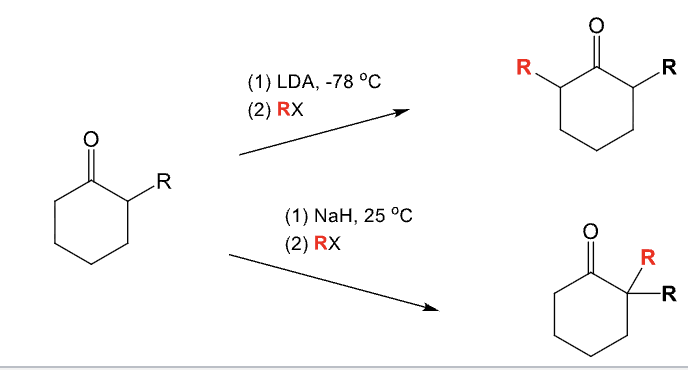
LDA, -78ºC
RX
Alkylation of the alpha position
Used to add an alkyl group (R) to the alpha position of an aldehyde or ketone
With an unsymmetrical ketone:
R group adds to less substituted side
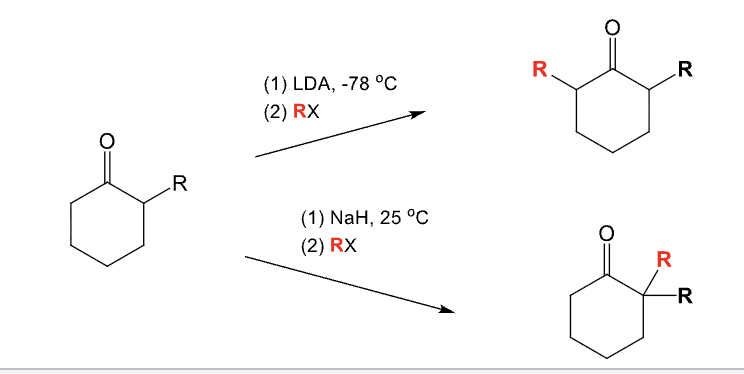
Provide the function for the Reagent:
NaH, 25ºC
RX
Alkylation of the alpha position of an aldehyde or ketone
With unsymmetrical ketone:
Add R group to the more substituted side
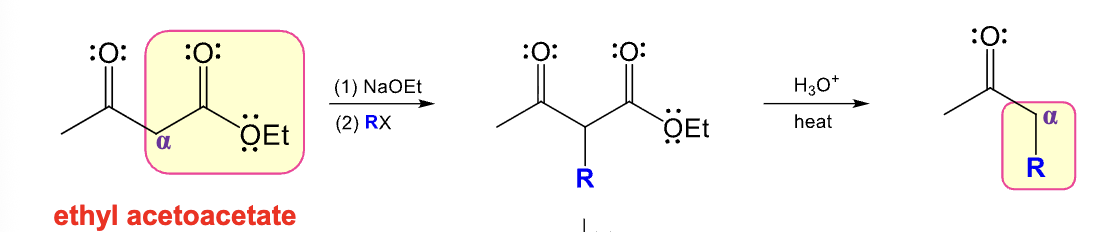
What are the structures for Ethyl acetoacetate and diethyl malonate?

Provide the function for the Reagent:
NaOEt
RX
H3O+,Heat
Acetosynthetic ester synthesis
Ethyl Acetoacetate can be converted into a monosubstituted derivative of acetone
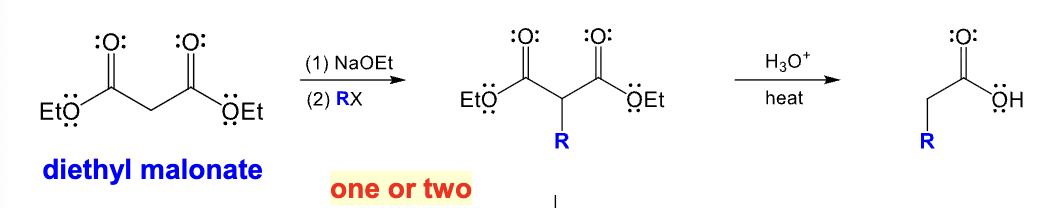
Malonic ester synthesis
Diethyl malonate can be converted into a monosubsituted derivative of acetone
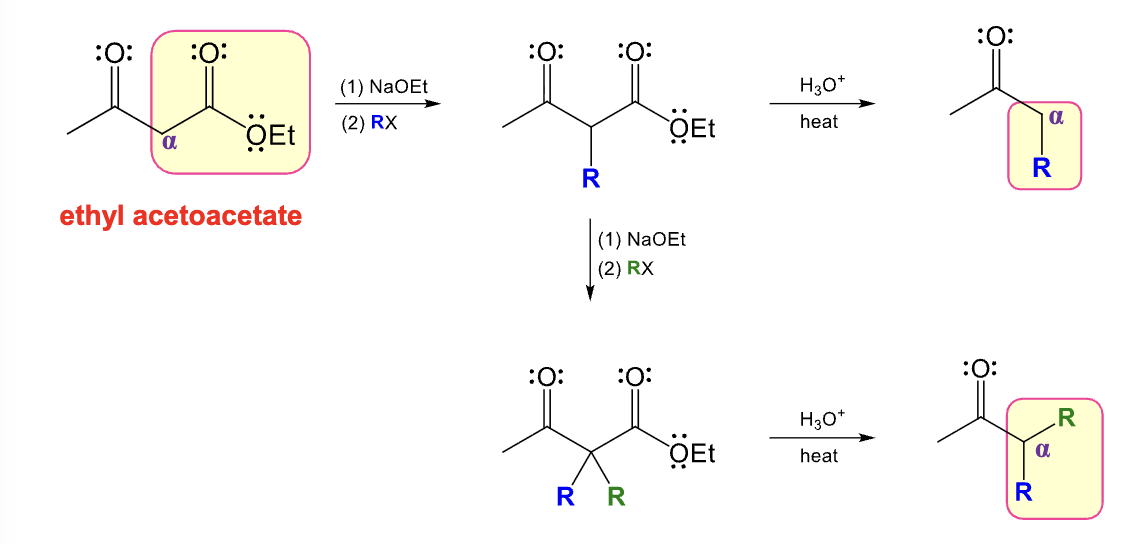
Provide the function for the Reagent:
NaOEt
RX
NaOEt
R’X
H3O+, heat
Acetosynthetic ester synthesis
Ethyl Acetoacetate can be converted into a di substituted derivative of acetone
Malonic ester synthesis
Diethyl malonate can be converted into a di subsituted derivative of acetone
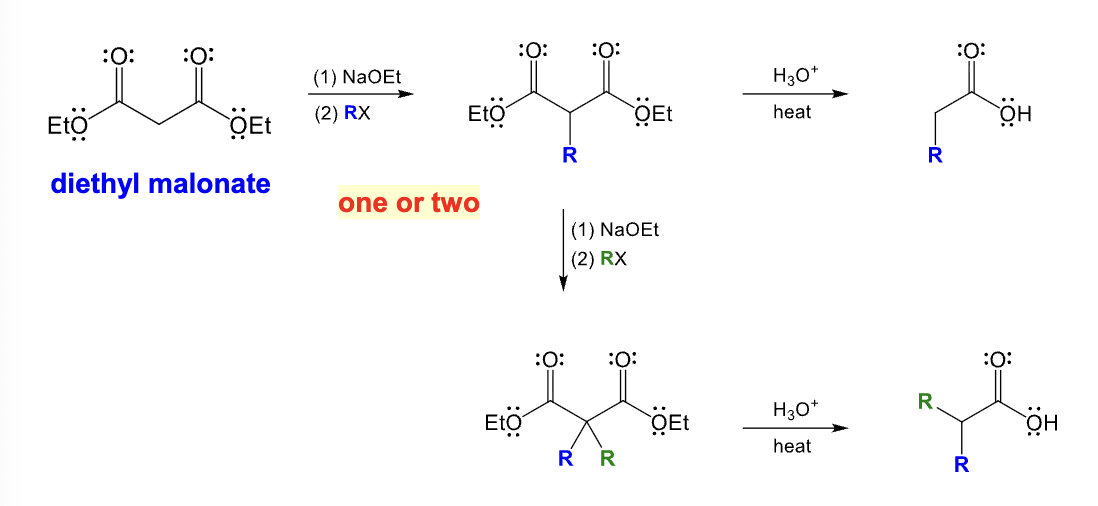
Provide the function for the Reagent: (Gilman Reagents)
R2CuLi
H3O+
Micheal reaction
Add one R group to the beta position and a hydrogen to the alpha position
1,4 addition product
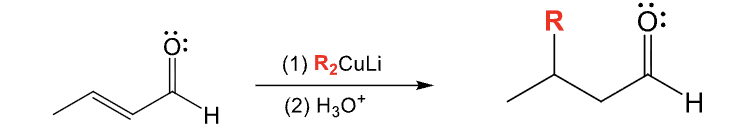
Provide the function for the Reagent: (Grignard Reagents)
RMgBr
H3O+
Makes a 1,2 addition product
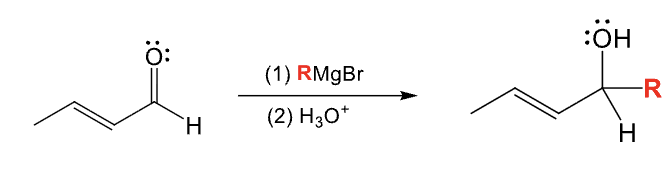
Provide the function for the Reagent:
R2CuLi
R’X
H3O+
Micheal reaction
Add one R group to the beta position and the R’ group from step 2 to the alpha position
Provide the function for the Reagent:
R2CuLi
R’X
or
H3O+
R group is added to the beta position
With RX as the second step:
Add second R group to alpha position
With H3O+ as the second step:
Add a hydrogen to the alpha position
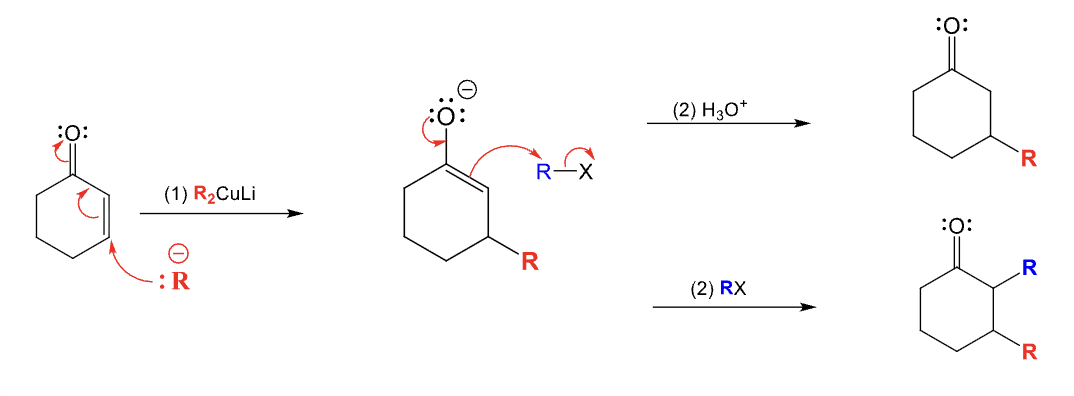
Provide the function for the Reagent:
NaCN (or KCN)
xs LiAlH4
H2O
Used to convert an alkyl halide into an amine
Adds one more carbon to starting material
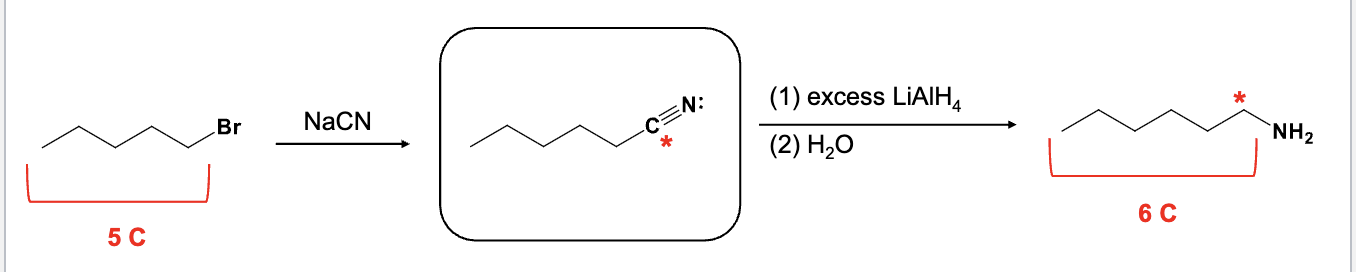
Provide the function for the Reagent:
SOCl2
NH3
xs LiAlH4
H2O
Used to convert carboxlyic acid into an amine
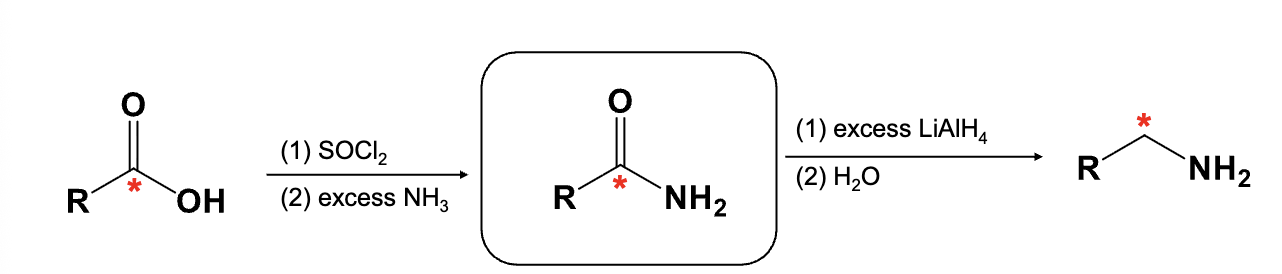
Provide the function for the Reagent:
Creation of nitration of aromatic ring using HNO3, H2SO4, and then
Fe or Zn, H3O+
NaOH
Reduces aryl nitro group (NO2) to an amino group (NH2)
-NaOH is required to get neutral amino group
Provide the function for the Reagent:
SnCl2, H3O+
NaOH
Selectively reduces an aryl nitro group (NO2) to an amino group (NH2)
-NaOH is required to get a neutral amino group
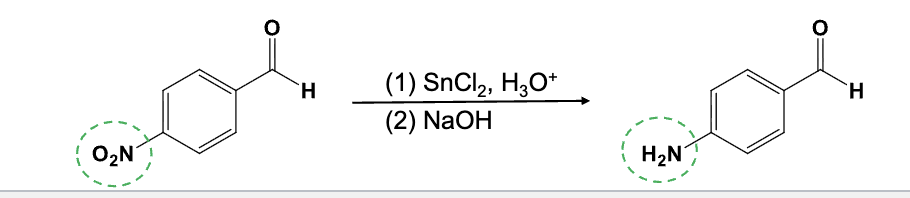
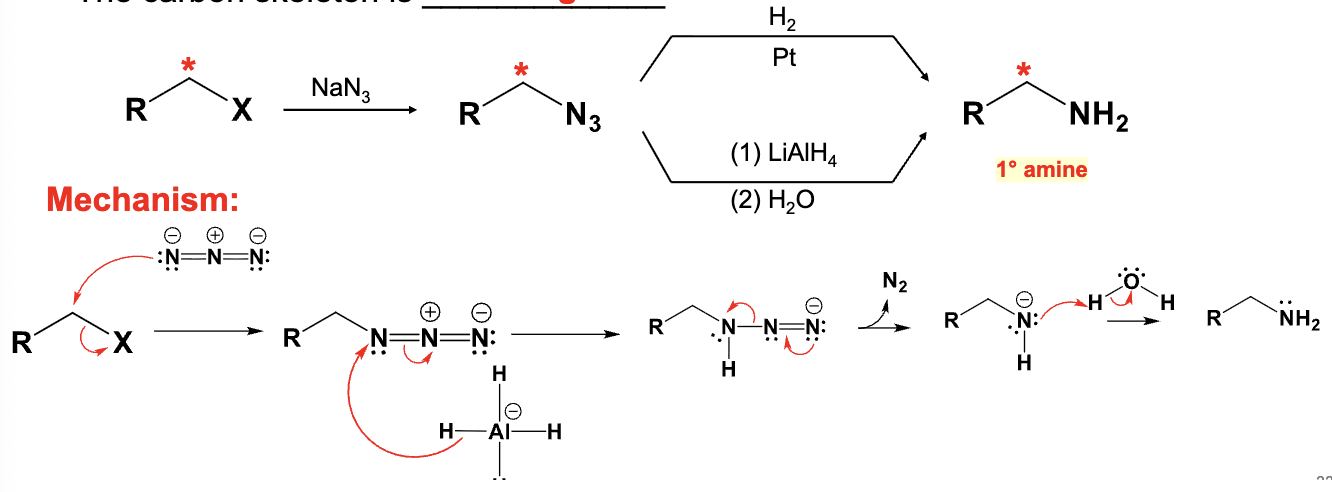
Provide the function for the Reagent:
NaN3
LiAlH4
H2O
Converts alkyl halide into an amine
-Adds one more carbon to starting material
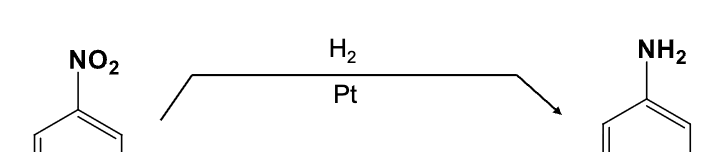
Provide the function for the Reagent:
H2, Pt
Reduces NO2 group into an NH2 group
Reduces (Azide) N3 into an NH2 group
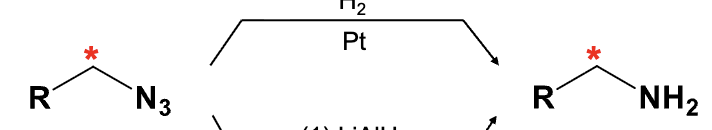
Provide the function for the Reagent:
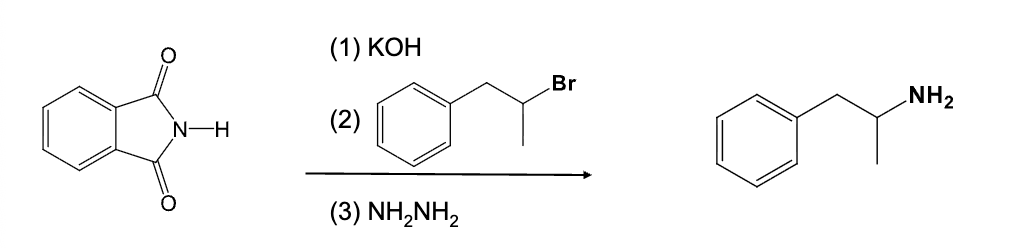
KOH
RX
NH2NH2
Gabriel synthesis
Reaction starts with pthalimide
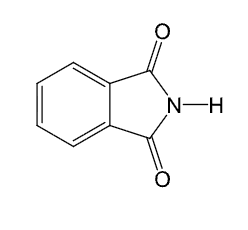
Makes primary amines
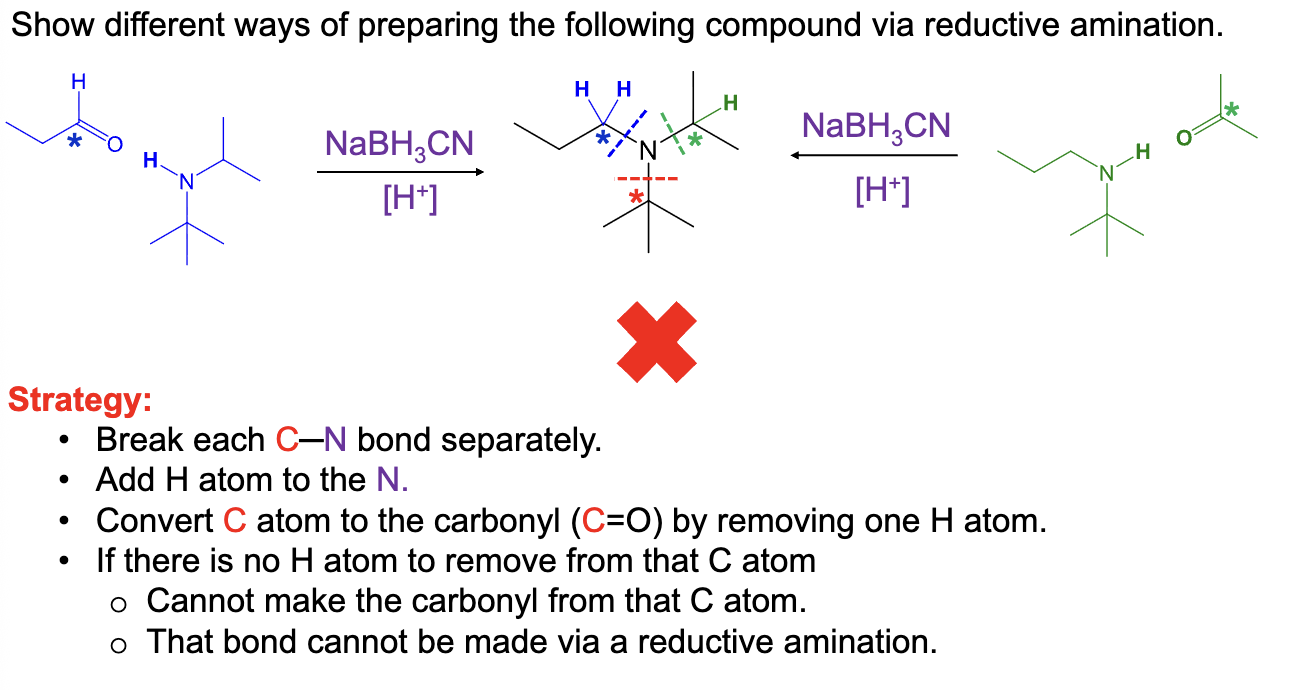
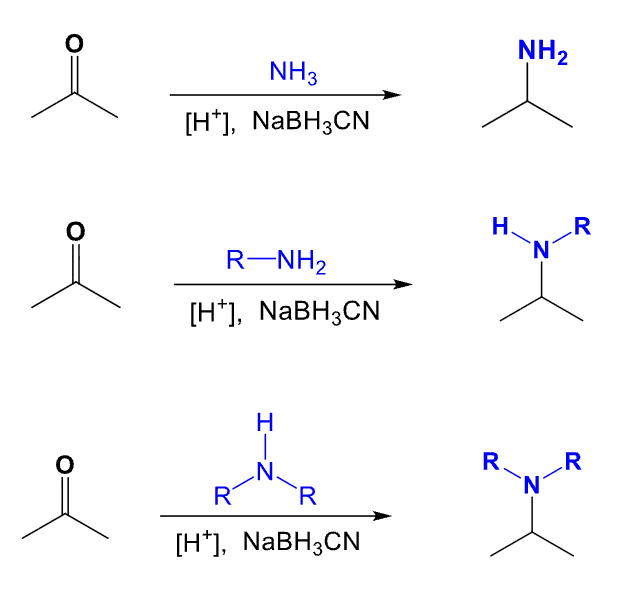
Provide the function for the Reagent:
H+, NaBH3CN
Used to recieve a reductive amination
Reaction occurs between a ketone/aldehyde and an amine/ammonia
To get the product:
Remove the carbonyl carbon oxygen atom with a double bond and connect the N atom in the amine/ammonia to the former carbonyl compound by a single bond
Provide the function for the Reagent:
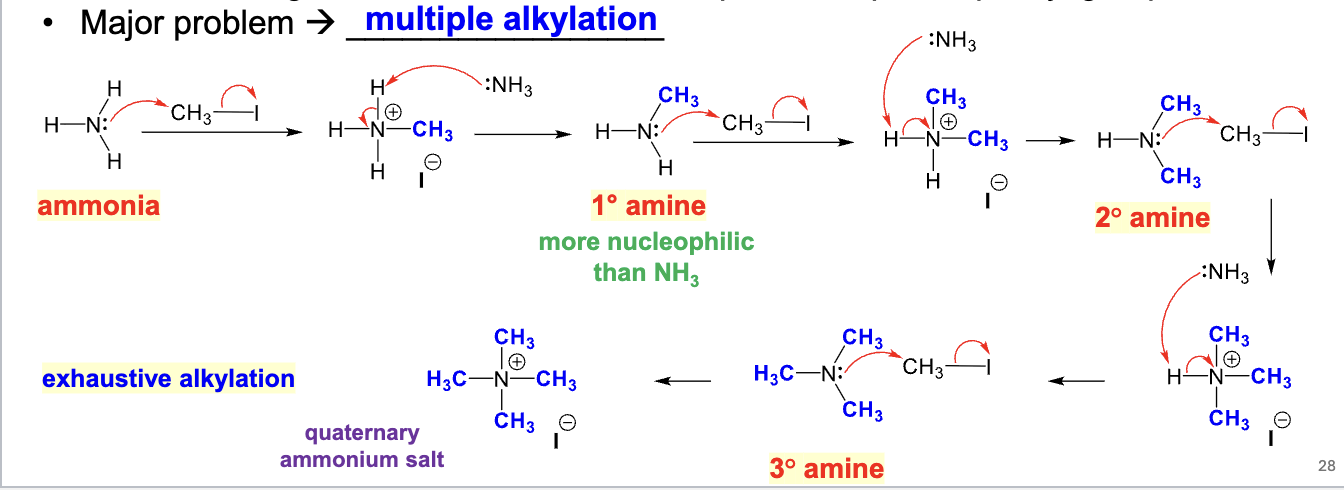
xs CH3I
Used to convert amine/ammonia to a quaternary ammonium salt
Provide the function for the Reagent:
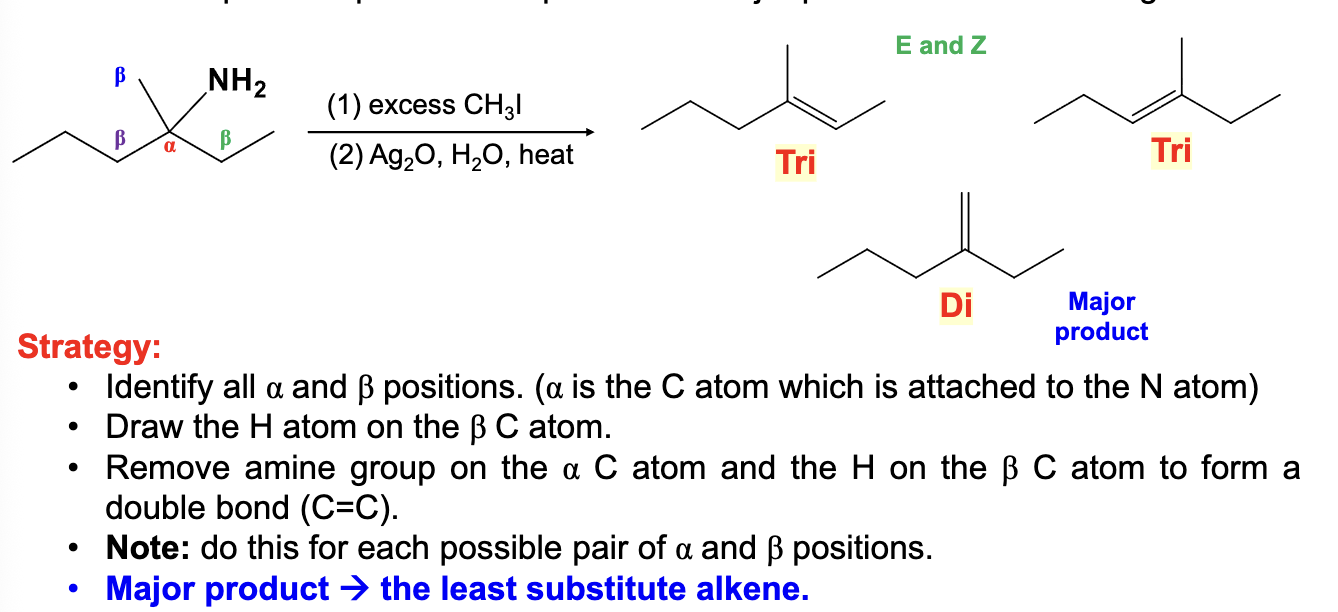
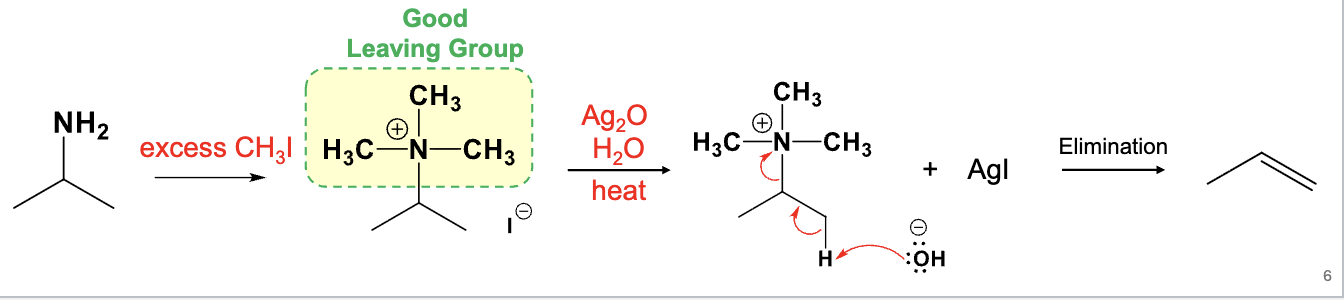
Excess CH3I
Ag2O, H2O, heat
Hoffman elimination
Elimination of beta H and NH2 (or NHR’, NR’R’’) to make an alkene
A less substituted product is the major product
Provide the function for the Reagent:
ROCl
An amine will undergo acetylation, giving an amide
Provide the function for the Reagent:
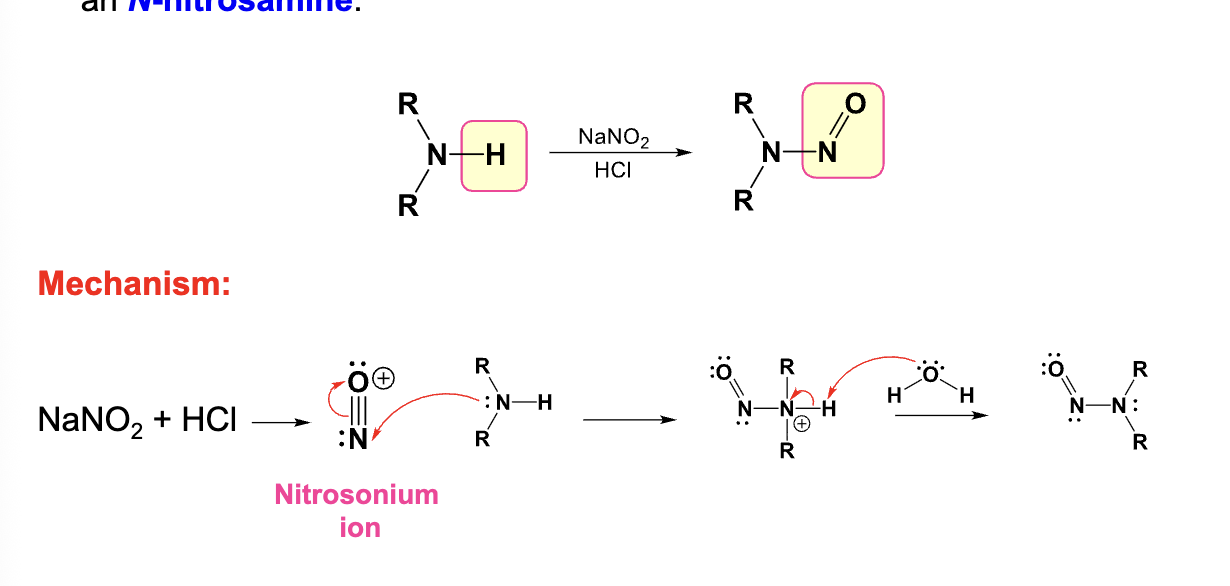
NaNO2, HCl
Converts primary amine into a diazonium salt
Converts a secondary amine into an N-nitrosamine
Provide the function for the Reagent:
CuCl
The diazonium group is replaced with a chlorine atom
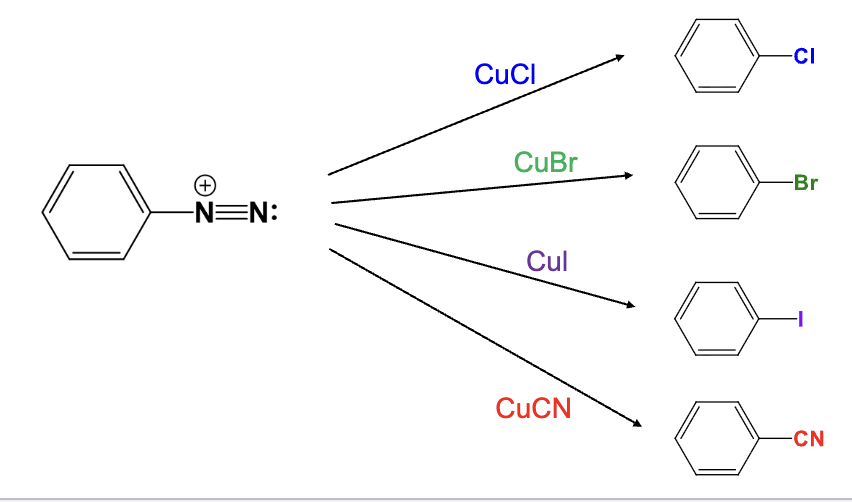
Provide the function for the Reagent:
CuBr
The diazonium group is replaced with a bromine atom
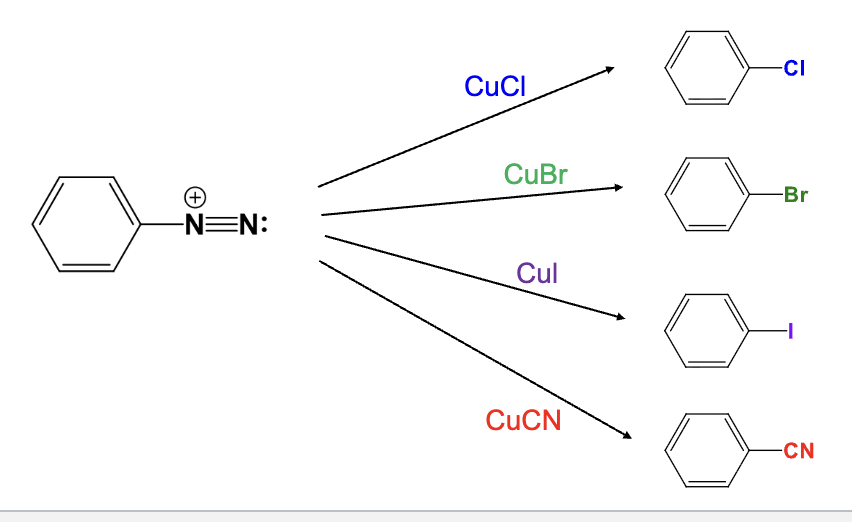
Provide the function for the Reagent:
CuI
The diazonium group is replaced with a Iodine atom
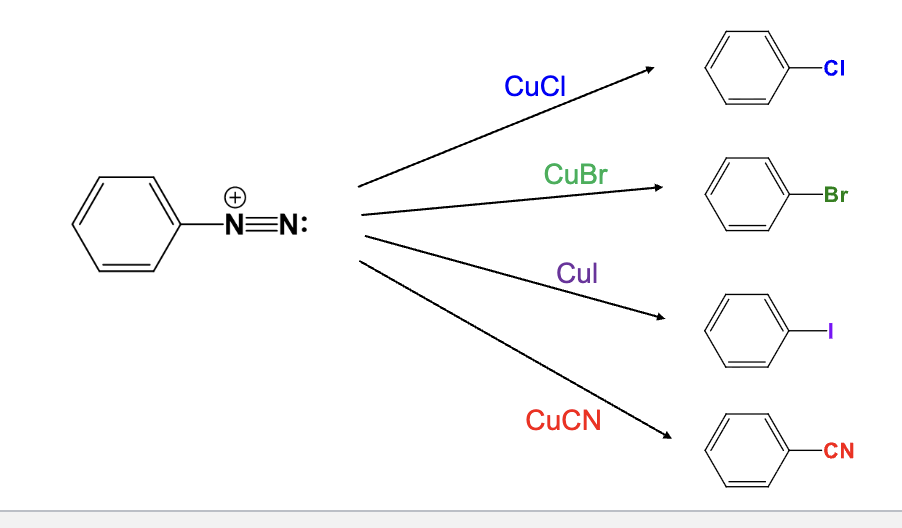
Provide the function for the Reagent:
CuCN
The diazonium group is replaced with a Cyano group
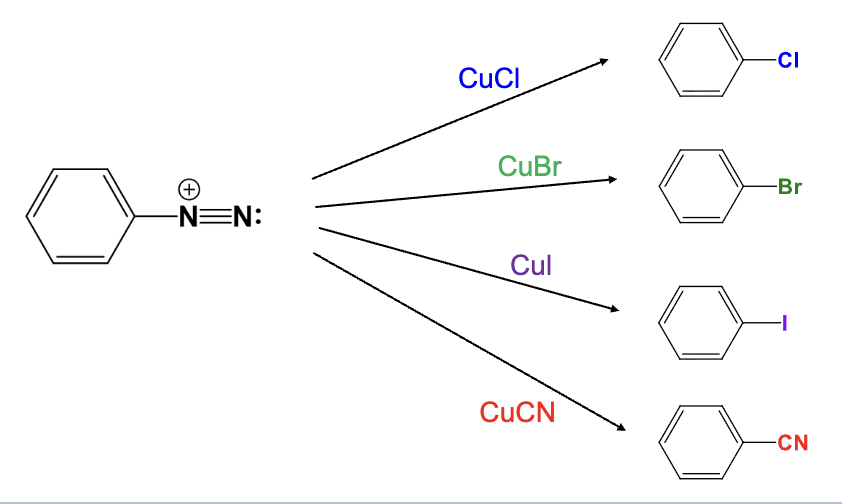
Provide the function for the Reagent:
HBF4
The diazonium group is replaced with a fluorine atom
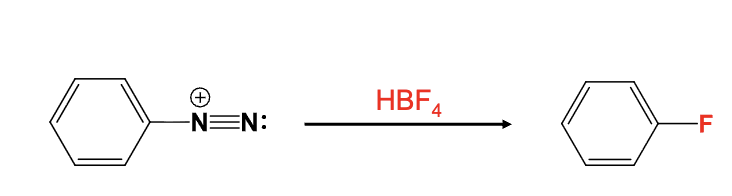
Provide the function for the Reagent:
H3PO2
The diazonium group is replaced with a hydrogen atom
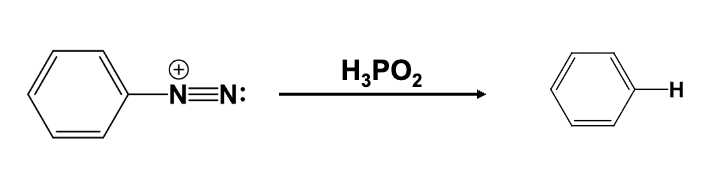
Provide the function for the Reagent:
H2O, heat
The diazonium group is replaced with a OH group
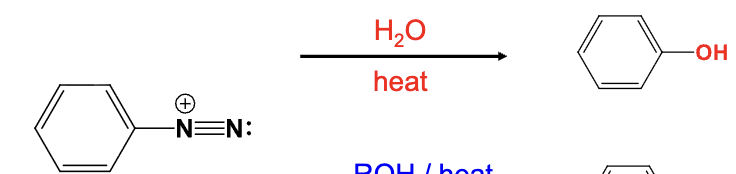
Provide the function for the Reagent:
ROH, heat
The diazonium group is replaced with a OR group
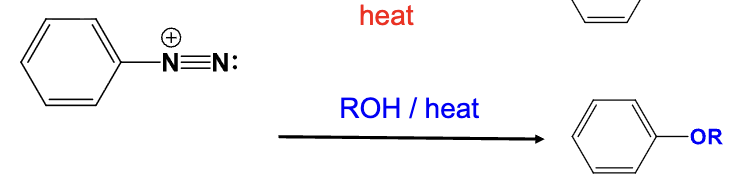
Provide the function for the Reagent:
Br2, 0ºC
Add a bromine atome to the C2 position on a pyrrole ring by replacing an H atom
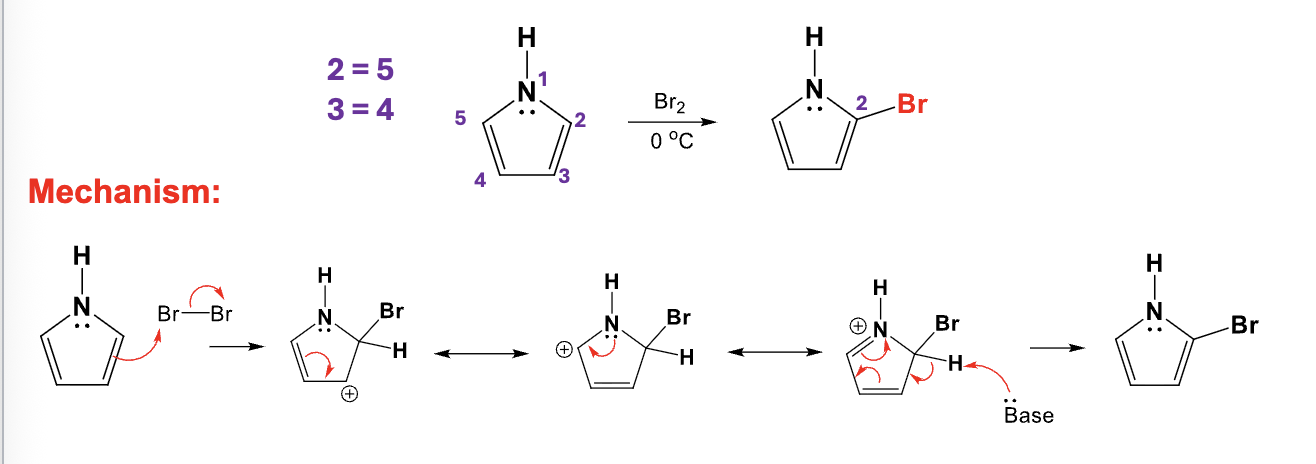
Provide the function for the Reagent:
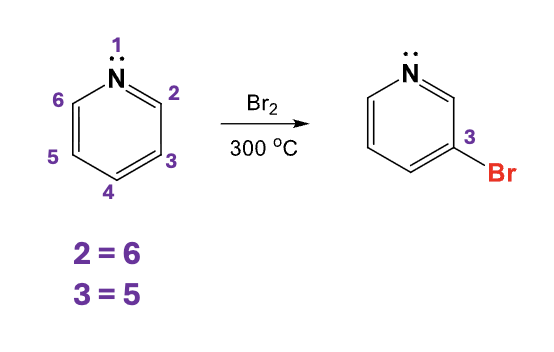
Br2, 300ºC
Add a bromine atome to the C3 position on a pyridine ring by replacing an H atom
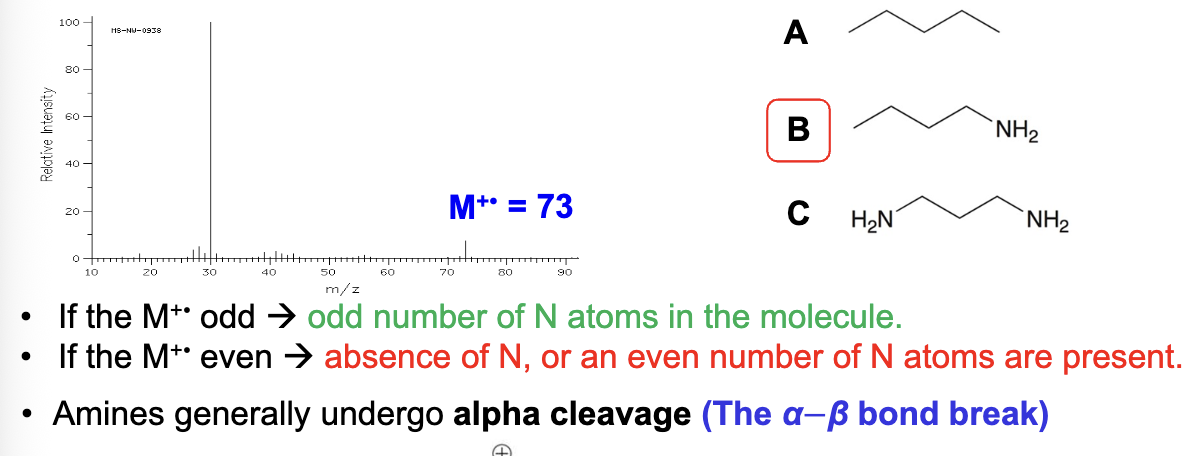
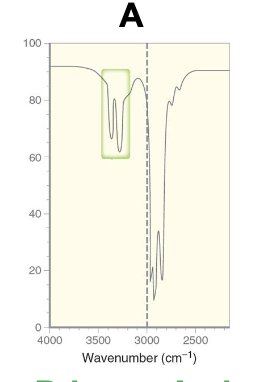
How can you identify amines from Spectroscopy?
Ir spectroscopy:
Primary amine has two sharp peaks around 3300 - 3500
Secondary amine has 1 sharp peak around 3300 - 3500
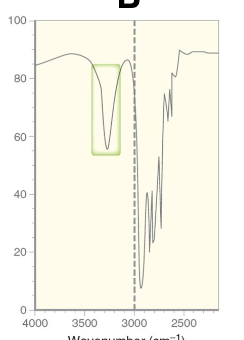
Tertiary = N/A
Mass spectrometry: