A2 Units 3.6, 3.7 & 3.5 Enthalpy, Entropy and Chemical Kinetics
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
Rate of reaction & Activation energy

Measuring the rate of reaction - Changes in colour

Measuring the rate of reaction - Change in volume of gas

Measuring the rate of reaction - Inverted measuring cylinder

Measuring the rate of reaction - Changes in pressure

Measuring the rate of reaction - Changes in pH

Measuring the rate of reaction - Formation of a precipitate

Measuring the rate of reaction - Loss in mass of a reactant

Measuring the rate of reaction - Quenching
Quick but requires multiple stages

Iodine Clock

CALCULATING THE RATE OF REACTIONS USING NUMERICAL OR GRAPHICAL DATA

Example Question

Orders of reactions

Units of the rate constants

Calculating orders & rate constants

Examples

Graphs representing orders of reactions

Uses of kinetic data

Predicting mechanisms from orders of reaction

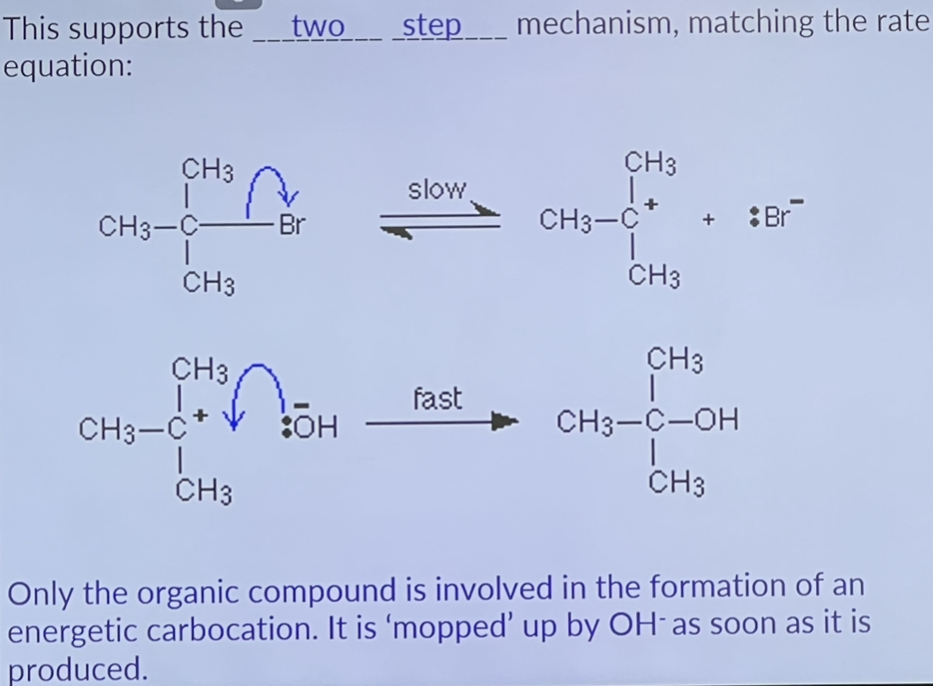
Tertiary halogenoalkanes

Two step mechanism
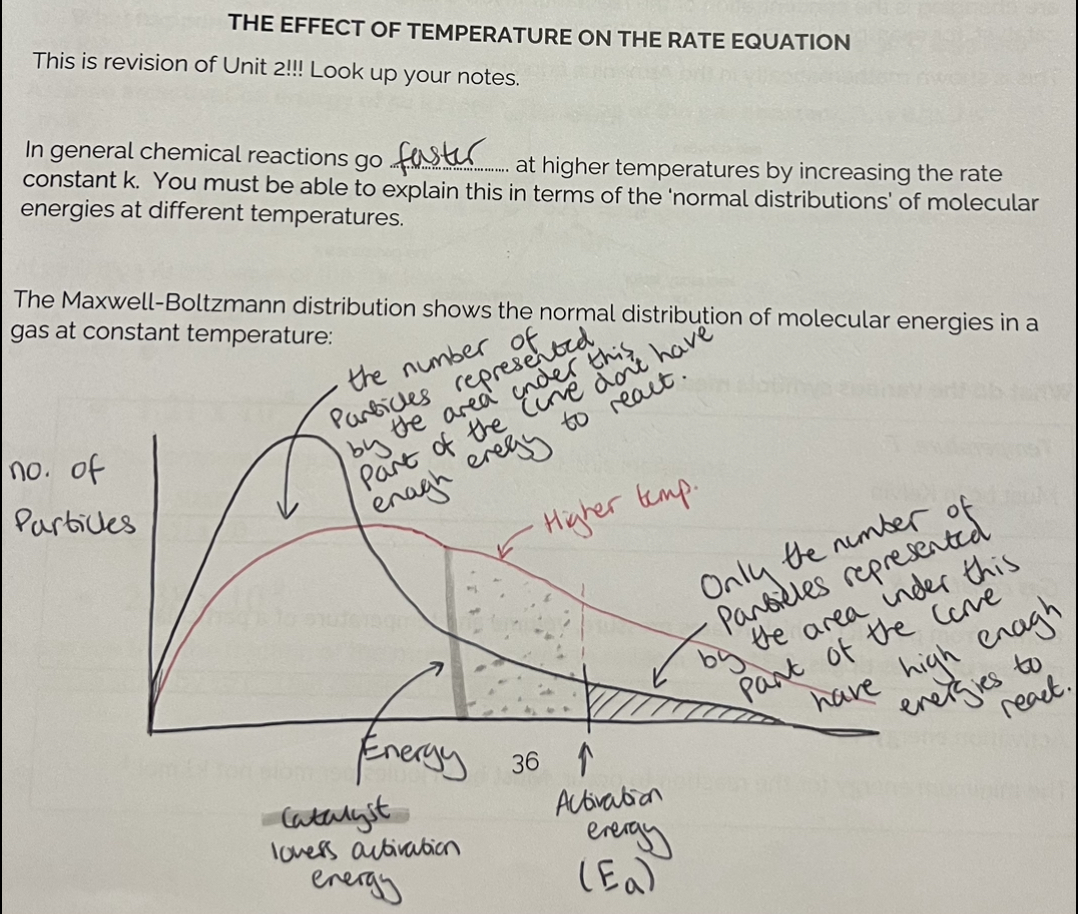
The effect of temperature on the rate equation
Generally, with every 10*C rise, the rate doubles.
The Arrhenius Equation

Using the Arrhenius Equation

The effect of a catalyst & Examples

Catalyst

Using the equation to find activation energy

Questions

Questions

Enthalpy Changes for solids and solutions
Chemical reactions occur because the products of a reaction represent a more stable system than the reactants; the system will tend to move to a position of lower energy.

What are standard conditions & Enthalpy?
Enthalpy = /\H0

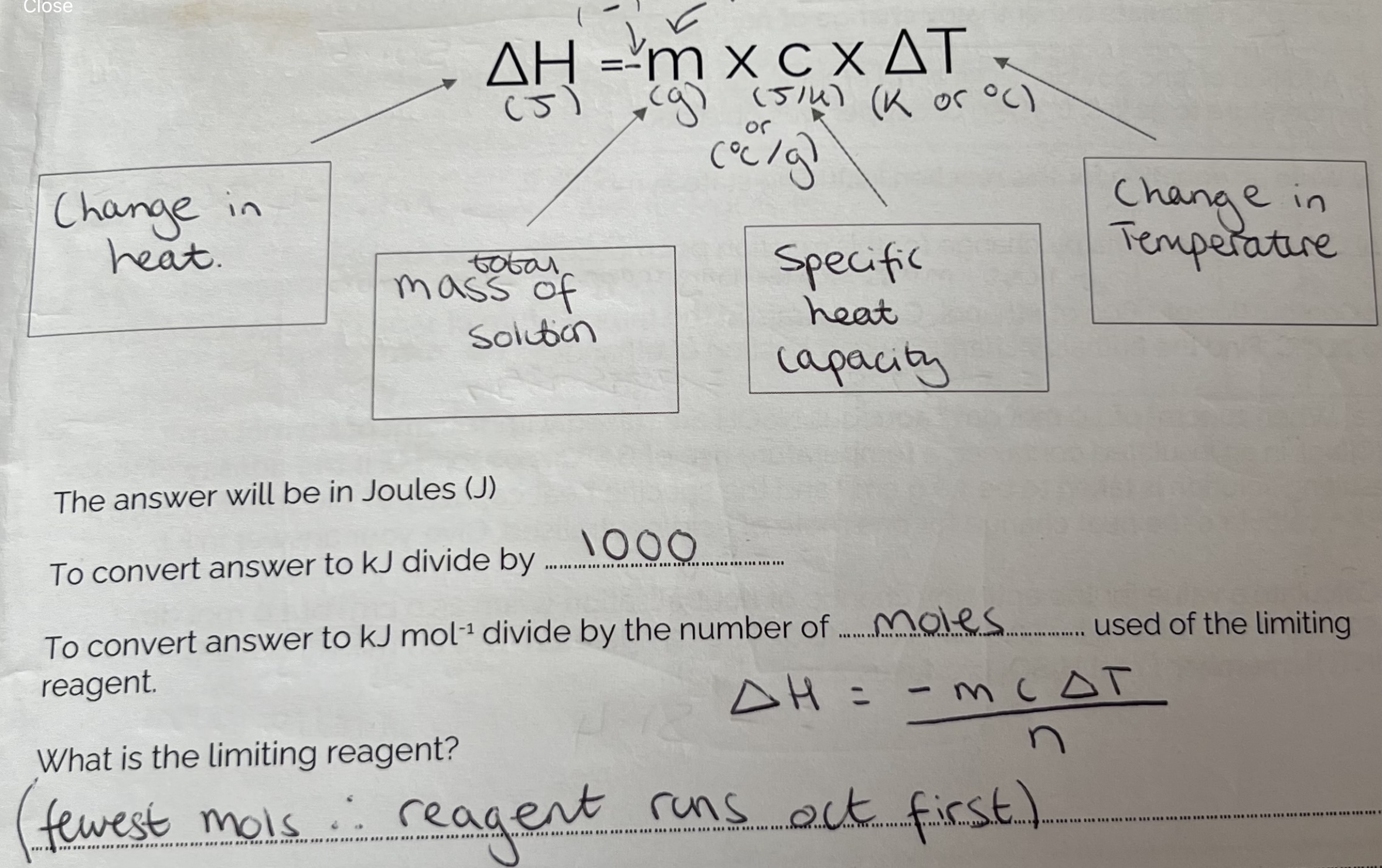
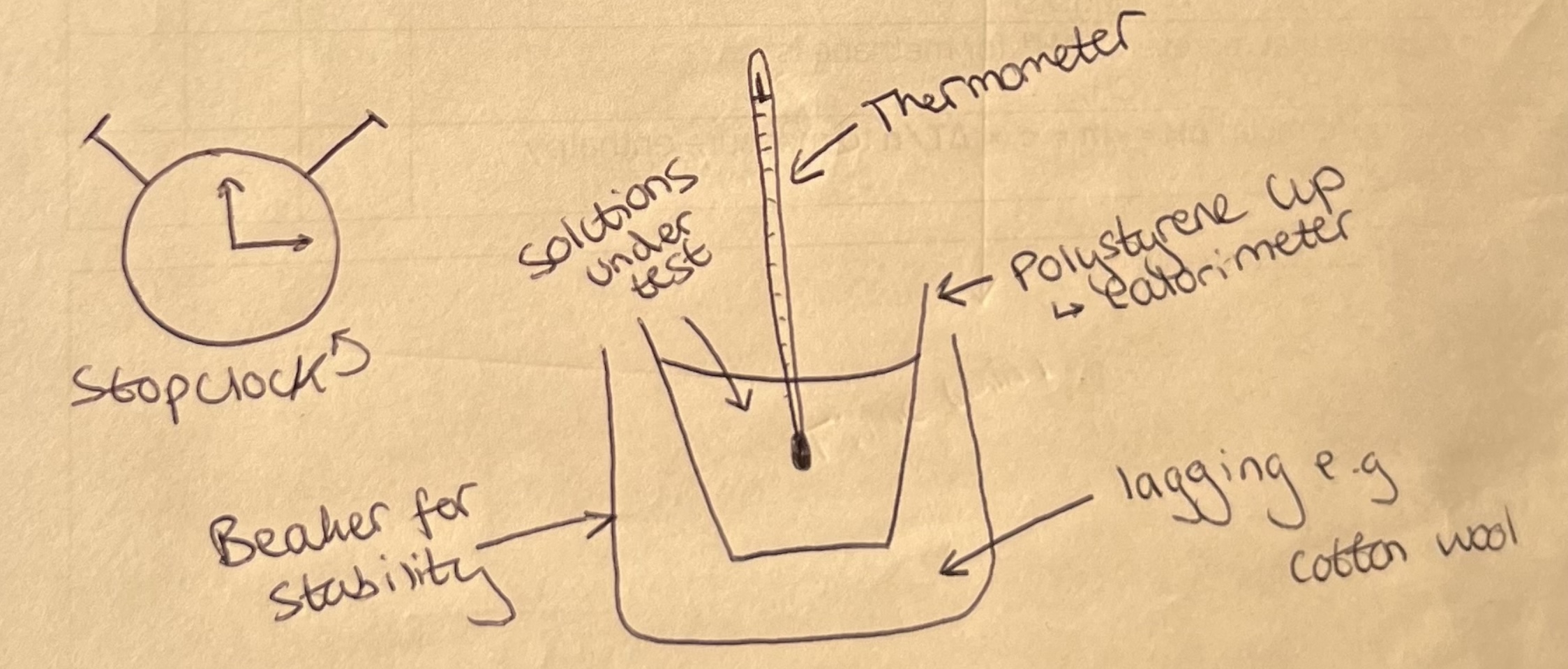
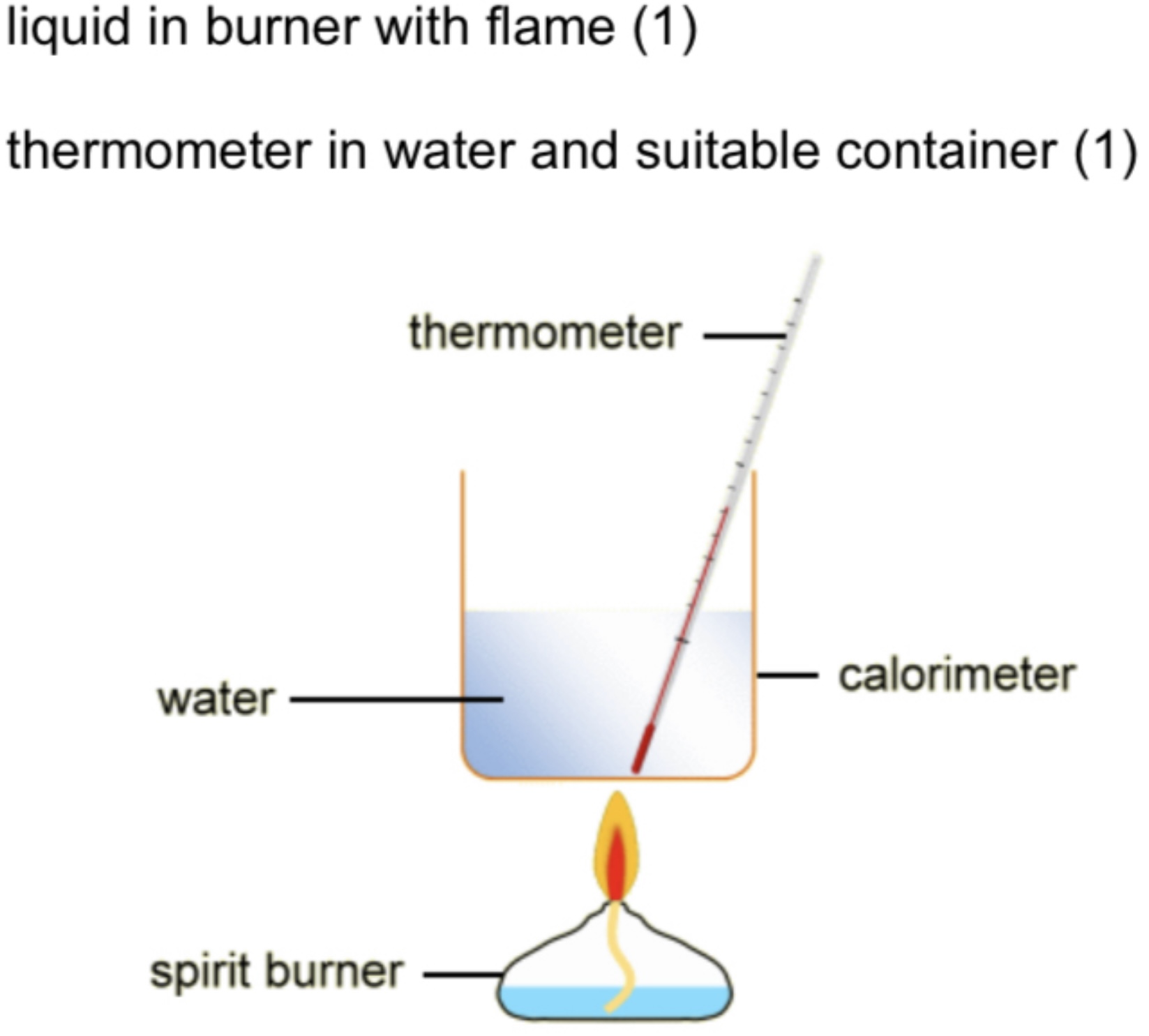
Measuring Enthalpy Changes
Enthalpy changes are measured in k J mol-1 but experiments on measuring enthalpy changes always measure a temperature change. The temperature change is then converted to enthalpy change using the following formula.
Labelled apparatus to measure enthalpy change
Spirit burner
Example
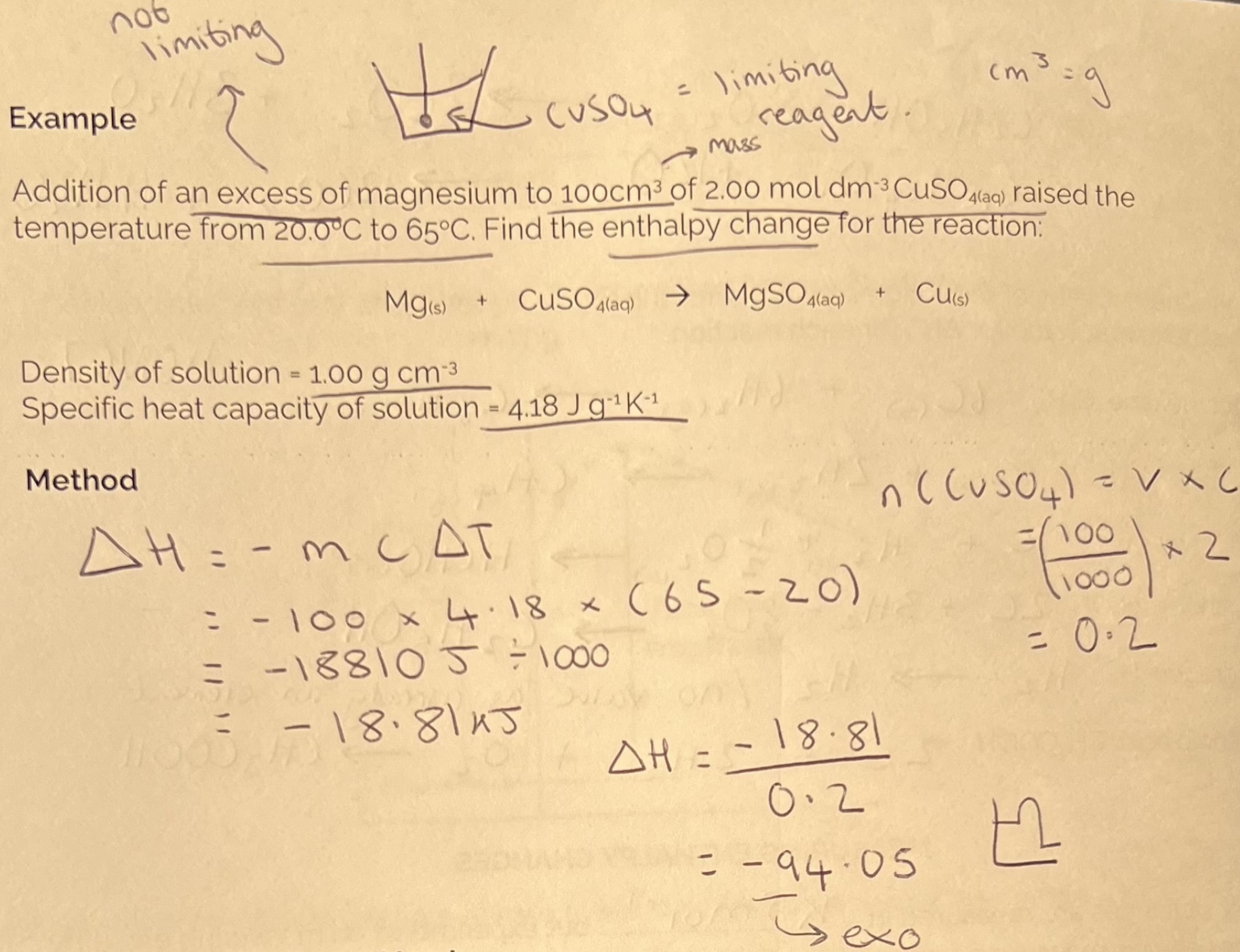
Hess’s Law & Energy Cycle
The overall enthalpy change in a chemical reaction is independent of the route taken or the number of intermediate steps.

Example

Enthalpy changes of combustion data

Enthalpy change of formation

Using bond enthalpies to determine Enthalpy changes

Calculating enthalpy change from a graph
If we used large lumps of zinc instead of zinc powder in this experiment, the reaction would be slower because lower surface area, so, less chance of collisions / fewer collisions per unit time.
If large lumps were used, it would take more time for the graph to reach its maximum after mixing, so graph would not rise as steeply.

Enthalpy changes in chemical reactions - Standard Enthalpy of Reaction & Combustion

Standard Enthalpy of formation

Standard Enthalpy of atomisation

Enthalpy change of hydration, lattice formation, lattice breaking & standard Enthalpy of solution

Ionisation Energy & Electron Affinity
Ionisation Energy - The Enthalpy (energy) required to remove 1 mol of electrons from 1 mol of gaseous atoms to from 1 mol of gaseous ions with a single positive charge (+1)
e.g M(g) —> M+(g) + e-
Electron Affinity - The Enthalpy change to make 1 mol of gaseous negative ions from 1 mol of gaseous atoms by the addition of 1 electron
e.g Cl(g) + e- —> Cl-(g)
/\H is negative
Examples

Born-Haber Cycles

Example - NaCl(s)

Examples CaCl & CaCl2

Calculating /\Hf
You can see that /\H, for the theoretical compound CaCl(s) is favourable for its formation. Why is CaCl2(s) the compound that exists?
The enthalpy of formation for CaCl2(s) is far more exothermic so that this compound is known. The very large, negative value of the formation lattice energy in the latter case is the most significant factor.

Finding the lattice energy of calcium oxide

Using Enthalpy to explain solubility

Entropy

Order of increasing entropy
Order of decreasing entropy:
4 molecules —> 2 molecules (-ve entropy)

Gibbs’s Free energy
(Gibbs free energy is negative) as entropy increase of solution overcomes endothermic enthalpy change.

Feasibility of reactions
N.B. Remember thermodynamics only deals with the feasibility of a reaction and gives no indication of the kinetics (how fast). Many reactions that are energetically feasible do not take place under ordinary conditions since the activation energy is too high for the reaction to occur.
T = /\H - /\G / /\S

Calculating entropy change of a reaction

Predicting feasibility of reactions at different temperatures

Factors affecting lattice energies

Relationship between enthalpy changes of solution, hydration and lattice breaking

Dissolving

Example
(practice questions from pg. 12)

Exothermic & Endothermic compounds Stability
