10. Proton transfer reactions: acid-base behaviour
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
The ionic theory
Electrolytes dissociate into freely moving charged particles called ions when dissolved in water or melted
How to calculate the pH of a strong base
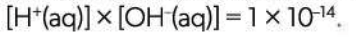
First find hydroxide ion (OH-) concentration, then H+ concentration
Experiments to distinguish between strong and weak acids and bases
pH measurement
Conductivity measurement (strong A&B solutions contain more ions, so are more conductive than weak A&B solutions)
Concentration measurement (RoR of strong acids with metals, metal oxides, metal hydroxides, metal hydrogencarbonates, and metal carbonates is greater than that of weak acids)
Alkali
A base that is soluble in water.
rain water pH
5.65
Acid rain
pH<5.6
Wet deposition, due to the acidic oxides dissolving and reacting with water in the air
Acid deposition
Process in which acidic particles, gases, and precipitation leave the atmosphere
Sulfur trioxide
Sulfur dioxide is oxidized by sunlight
Sulfurous acid and sulfuric acid
Sulfur trioxide + water in the air
Nitrogen monoxide, NO
Produced in the internal combustion engine and in jet engines.
Acid rain environmental impact
Increased acidity in the soil leaches important nutrients, such as Ca2+, Mg2+, and K+
Causes leaching of Al3+ ions from rocks → harmful to tress and aquatic life
Animals can’t survive lower pH rates
Acid rain impact people
Stone containing calcium carbonate, e.g. marble, is eroded by acid rain.
Salts can also form within the stone, causing the stone to crack and disintegrate.
Acids formed when NOx and SOx dissolve in water irritate the mucous membranes and increase the risk of respiratory illnesses, such as asthma, bronchitis, and emphysema.
In acidic water, there is more probability of poisonous ions, such as Cu2+ and Pb2+, leaching from pipes.
Methods to lower or counteract the effects of acid rain
Reduce amounts of NOx and SOx formed
Improved engine design
Using catalytic converters
Removing sulfur before, during, and after combustion of sulfur-containing fuels.
Switch to alternative methods of energy production
Reduce the amount of fuel burned, e.g. by reducing private transport,
Liming of lakes (Adding calcium oxide or calcium hydroxide (lime) neutralizes the acidity)
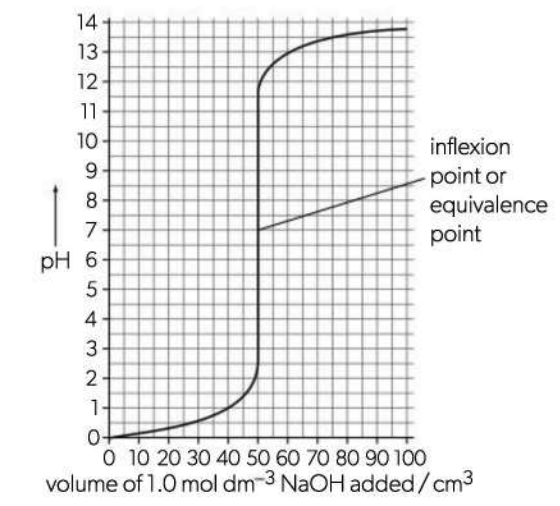
Inflexion point or equivalence point
Area on the graph where the pH changes most rapidly and the line is almost vertical
Concentration of the hydrogen ions
1 x 10-7 mol dm-3
pH
power of Hydrogen
Kw
Ionic product constant of water
Monoprotic
Acid/Base can donate/accept 1 proton
Diprotic
Acid/Base can donate/accept 2 protons
Triprotic
Acid/Base can donate/accept 3 protons
Oxidation
Loss of electrons
Reduction
Gain of electrons
Strong Acid and Base Properties
Acid completely dissociates
Irriversible (→)
Have weak conjugates
Weak Acid and Base Properties
Partially dissociates in water
Reversible (are at equilibrium, ←→)
Have Strong conjugates
Strong bases
Ba(OH)2
NaOH
KOH
LiOH
Metal hydroxides (M-OH)
Strong acids
HCl
HBr
HI
HNO3
H2SO4
Weak bases
Amines (NH(2)-R)
Ammonia (NH3)
Aminoethane (C2H5NH2)
Carbonates (contain CO32-)
weak acids
organic acids (-COOH)
CH3COOH
HCOOH
HF
H3PO4
HNO2
H2CO3
Dissociation/ ionization
The process by which ions are formed and separate when a compound dissolves in and reacts with water
pH
H+(aq) concentration
Measure of how acidic/alkaline some thing is (1-14)
(sf. start after decimal point)
pOH
the hydroxide ion concentration
Neutralization reaction
acid + base → salt + water
Dissociation reaction
Breaking into separate components (with water usually)
Incomplete dissociation
Not all compounds break apart
Hydroxide ion
OH-
Hydronuim ion
H3O+
Polyprotic
Acids/Bases that can donate/Accept more than 1 proton
Oxidation/redox reaction
transfer of electrons between two species
Amphoteric
A substance that can act as both an acid and a base
Amphiprotic
A substance that can act as both a proton acceptor and a proton donor
Bronsted-Lowry
Acids and bases are proton donors or acceptors
Arrhenious
Acid and bases increase concentration of hydrogen ion or hydroxide ion
Autoionization of water
H20(aq)<->H+(aq) + OH-(aq)
Estrification
A carboxylic acid (ROOH) reacts with an alcohol to form an ester.
Precipitation
Formation of a solid during a chemical reaction
Dissolution
A solute in a gaseous, liquid, or solid phase dissolves in a solvent to form a solution
Saturated solution
Has highest possible concentration of solute under given conditions
Determination of pH
Acid-base indicators
pH meter
"Universal" indicator
Corrosive
Chemically reactive
Concentrated
High number of moles of solute per dm3 of solution
Dilute
Low number of moles of solute per litre of solution
Synthesis
Production of chemical compounds by reaction from simpler materials
Decomposition
Breaking down a chemical compound into simpler substances
Reagents
Compound or mixture used to detect the presence or absence of another substance
e.g. by a color change
Anhydrous
A substance containing no water