Reactions EXAM 3
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
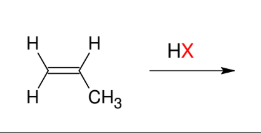
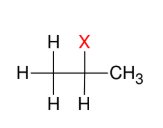
Addition of HX (Mark)
Add H and X
Adds a halide
to more subst.
carbon.
Note: Look for hydride/methyl shift
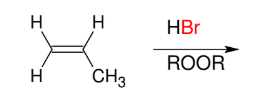
Addition of HBr (Anti-M)
Adds H and Br
Br on less subst. carbon
Can only use Br as halide
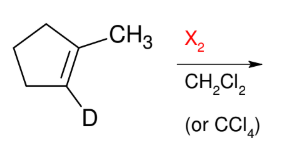
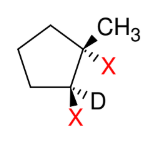
Dihalide Addition
Add X and X
Anti and co-planar
Bottom reagent can be ROOR (peroxide)
Trans
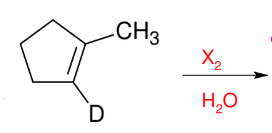
Halohydrin Reaction (Mark w/ OH)
Add X and OH
Anti and co-planar
OH on more subst.
KNOW MECHANISM
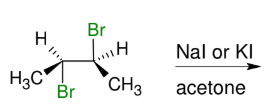
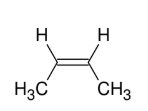
Forming alkene from vicinal dihalide
Wedges with wedges
and dashes with dashes
E2 Like!
Take away Br and forms DB
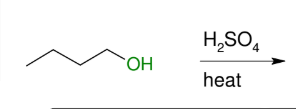
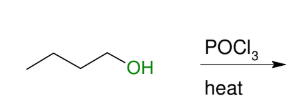
Dehydration to alkene (non-terminal)
E1 like and it cannot give terminal alkene
forms DB
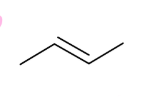
Dehydrates to form terminal alkene
forms DB
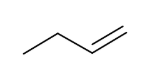
Additon of Oh (Mark and direct)
Racemic mixture
add OH only (Mark)
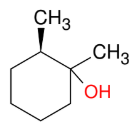
Oxymercuration/demercuration (OH mark w/o h2so4)
add H and OH (mark)
Anti
trans
Oxymercuration/demercuration (OH mark w/ h2so4)
1.) H3O+ / H+ / H2SO4
2.) H2O
or
H2SO4 (dilute)
add H and OH (mark)
Anti
If conc. H2SO4 —→ E1 rxn
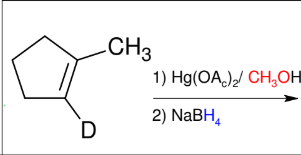
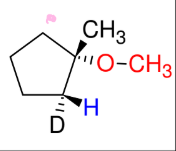
Oxymercuration/demercuration (forming ethers Mark)
add H and -OCH3 (mark)
Anti
SPECIAL: Adds alcohol
instead to form ethers!
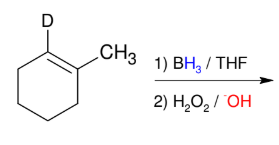
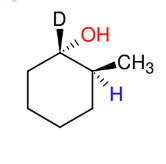
Hydroboration Oxidation
Add H and OH
Anti-M
syn
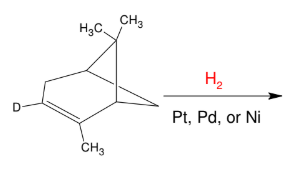
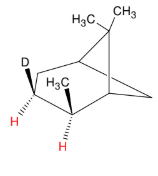
Catalytic Hydrogenation
Add H and H
syn
Pay attention to steric factors
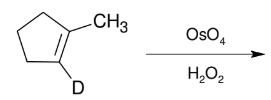
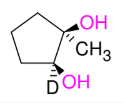
Glycol Synthesis from Alkene Oxidation (OsO4)
Adding OH and OH
syn
can use NMO instead of H2O2
treatment w/ HIO4 does ring opening “ozonolysis”
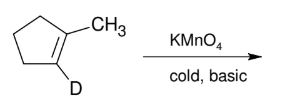
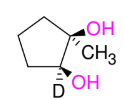
Glycol Synthesis from Alkene Oxidation (KMnO4 cold)
Adding OH and OH
syn
treatment w/ HIO4 does ring opening “ozonolysis”
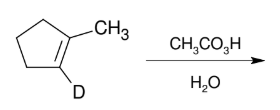
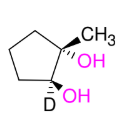
Glycol Synthesis from Alkene Oxidation (RCO3H or MCPBA)
Adding OH and OH
anti
KNOW MECHANISM
Can also use 1.) MCPBA, CH2Cl2 2.) H3O+
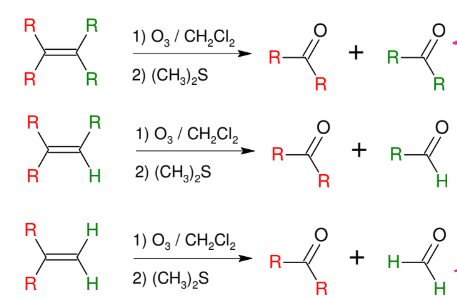
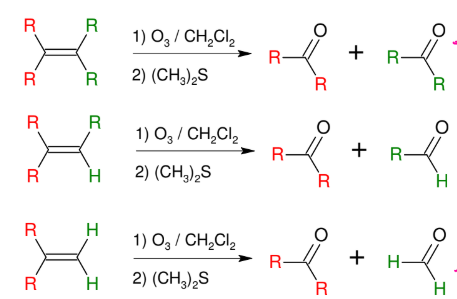
Ozonolysis
Cleaves the DB to turn C=C into C=O
creates aldehyde or ketone or isolate molecule
USE O3 / DMS or (CH3)2S
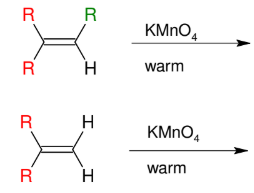
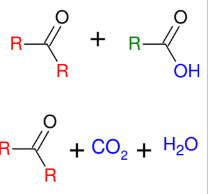
Warm KMnO4 Cleavage
Cleaves the DB to turn C=C into C=O
creates carboxylic acid or ketone
*further oxidizes to form
carboxylic acids
*cannot isolate the
formaldehyde
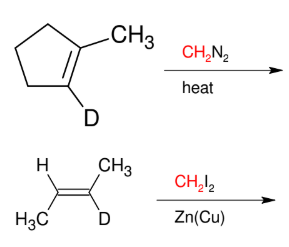
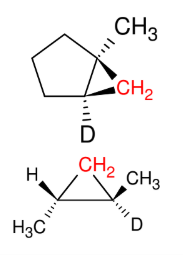
Carbene / Carbenoid addition (formation of cyclopropane)
add CH2 only
syn
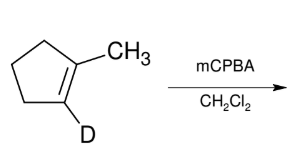
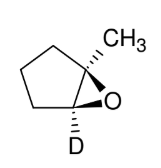
Oxidation of Alkenes: oxirane synthesis
adds O to form epoxide
syn
mCPBA with nonpolar
solvent can isolate
oxirane
treatment with H3O+ forms OH and OH anti
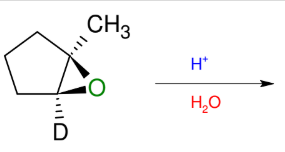
Opening of Epoxides
Adds OH and OH to epoxide
Can simply use H3O+ instead
*acidic conditions opens
from more substituted
side.
KNOW MECHANISM
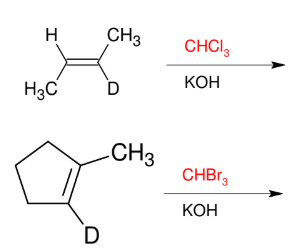
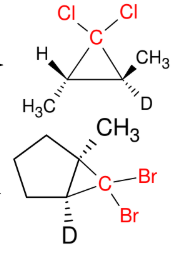
Formation of Dibromocarbenes and Dichlorocarbenes
Add CX2
Syn
*please look up the
mechanism so you can
see how the carbene
is formed
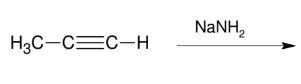
Formation of the acetylide anion
*forms the nucleophile that is handy when connecting carbons!
Can use NaH as well
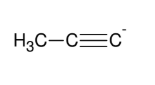
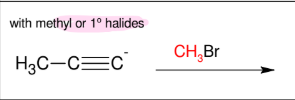
Using acetylide anion to form internal alkyne
Forms internal alkyne R-≡-R
Can introduce a long chain (i.e. CH3CH2CH2Br)
SN2
1° halides
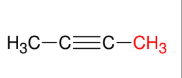
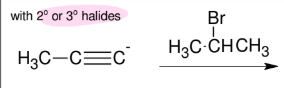
E2 rxn
w/ 2° or 3° halides
forms DB
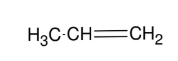
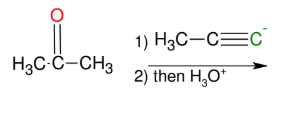
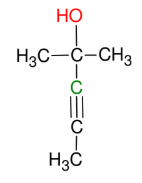
Uses of the acetylide anion with carbonyl groups (ketones, aldehydes, and formaldehydes)
adds OH group and R to carbonyl carbon
*acetylide anion attacks
partially positive carbon
*DO NOT FORGET
then H3O+
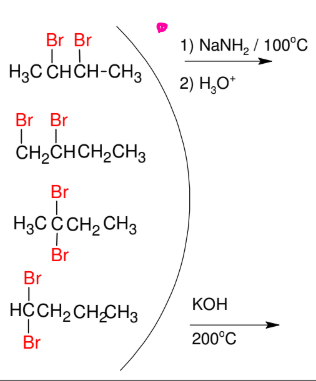
Synthesis of terminal Alkynes
*Need either geminal or
vicinal dihalides
*Look up mechanism
*NaNH2 FAVORS
terminal alkyne formation
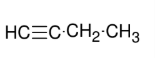
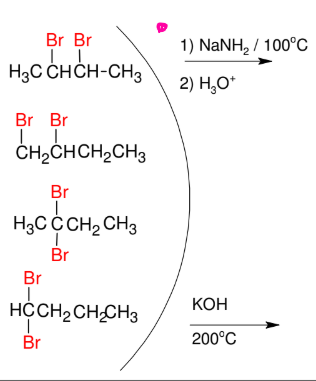
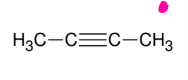
Synthesis of internal Alkynes
*Need either geminal or
vicinal dihalides
*Look up mechanism
*KOH FAVORS
internal alkyne formation
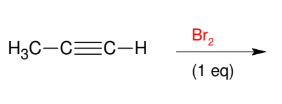
Halogenation of alkynes (Br2 and alkyne)
Adds Br and Br to alkyne to make alkene
Stereochemistry cannot be controlled
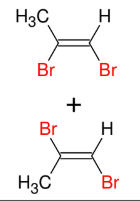
Halogenation of alkynes (HBr and alkyne - Mark)
Adds Br and H to alkyne
1 eq makes alkene, 2 eq makes alkane
Mark
Syn
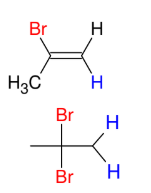
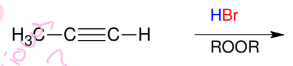
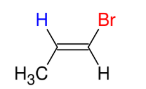
Halogenation of alkynes (HBr and alkyne - Anti M)
Adds Br and H to alkyne
Anti M
Syn
ROOR = peroxide
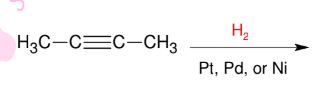
Catalytic reduction of alkyne with reactive catalyst
*Takes it all the way back
to alkane
*generally bad yield
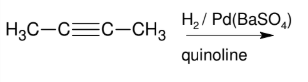
Alkyne to Alkene: TRIPLE to DOUBLE BOND (cis alkene formation)
Adds H and H to form cis alkene
Syn (cis alkene formation)
Dont worry about other reagents - just mention H2 / Lindlar
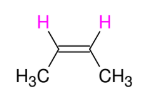
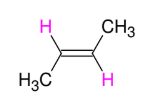
Alkyne to Alkene: TRIPLE to DOUBLE BOND (trans alkene formation)
Adds H and H to form trans alkene
Anti (trans alkene formation)
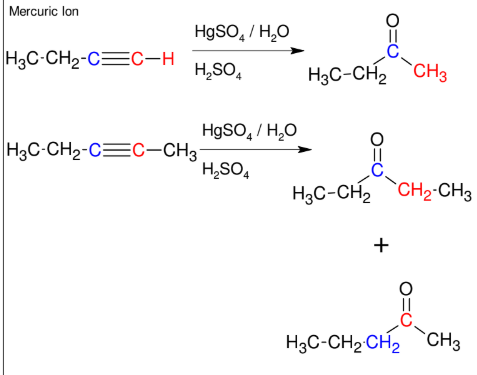
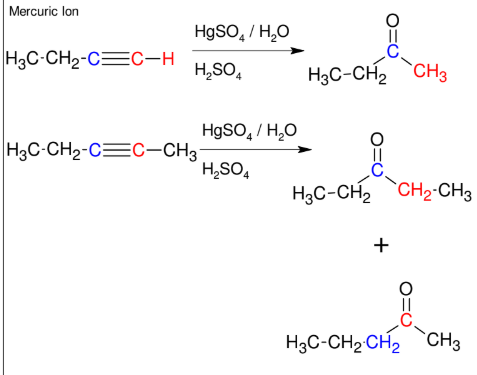
Addition of H-OH to alkynes (forming ketone)
Adds H-OH to form ketone
Mark
*If not terminal, you will
get a mixture.
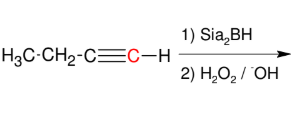
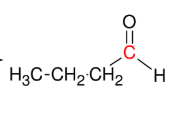
Addition of H-OH to alkynes (forming aldehyde)
Adds H-OH to form aldehyde
Anti M
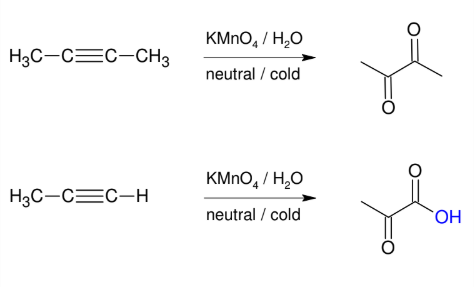
Oxidation of alkynes (mild conditions)
Adds O and O to make two carbonyls (ketones usually)
*Forms vicinal
carbonyls
*further oxidizes terminal
alkynes to form
carboxylic acid.
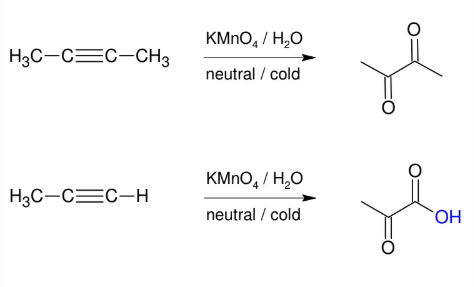
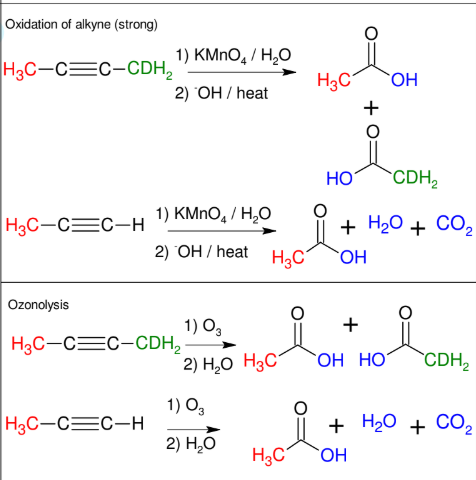
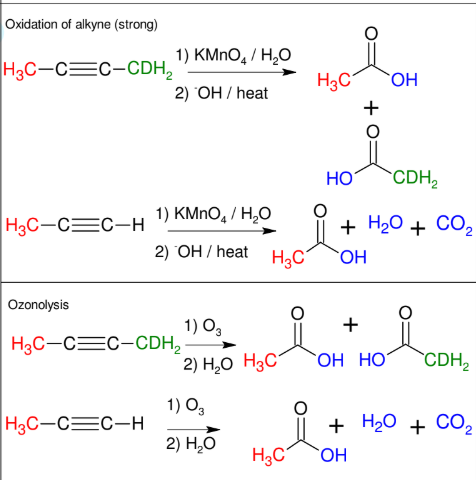
Cleavage of Alkynes
C≡C gets split into two COOH groups
forms H2O and CO2 if terminal
Can proceed via oxidation of alkyne (strong) or ozonolysis
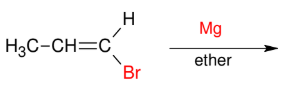
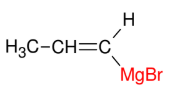
The Grignard Reagent
Add Mg to make R-MGBr
adds to 1°, 2°, or 3° allyl, vinyl, or aryl carbons
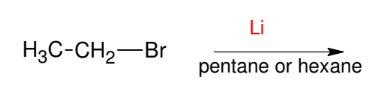
The Organolithium Reagent
Replace X with Li
Acts like grignard but stronger
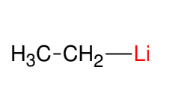
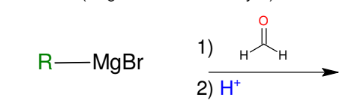
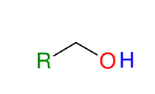
Formation of alcohols from Grignard (forming 1° alcohols)
Adds R group to formaldehyde
Acid workup step to make OH group
*Know this mechanism!
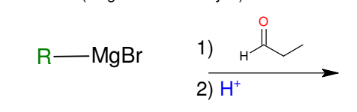
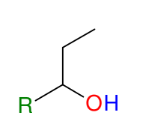
Formation of alcohols from Grignard (forming 2° alcohols)
Adds R group to aldehyde
Acid workup step to make OH group
*Know this mechanism!
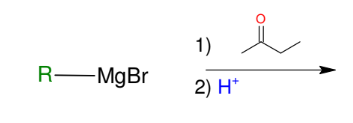
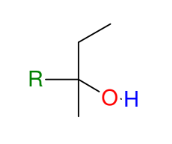
Formation of alcohols from Grignard (forming 3° alcohols)
Adds R group to ketone
Acid workup step to make OH group
*Know this mechanism!
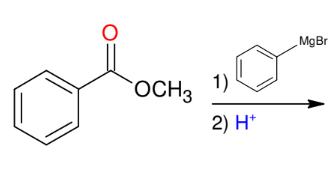
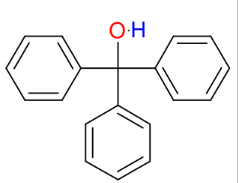
Grignard and esters or acid halides
Know from OCHEM LAB (synthesis of triphenylmethanol) !
adding phenyl groups
*Reaction goes until
completion
*Know this mechanism !
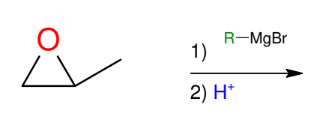
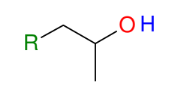
Grignard and Epoxides (opening of epoxides)
*SN2 like (attacks least
substituted side)
*Know this mechanism!
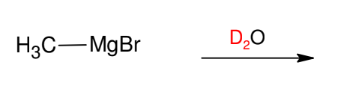
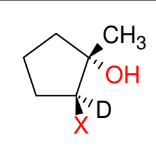
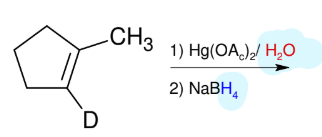
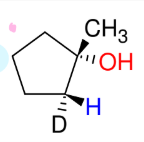
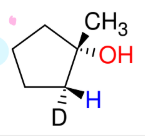
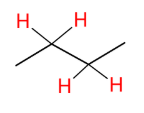
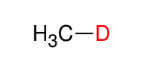
Attaching Deuterium to Carbons
*This is just good to
know.
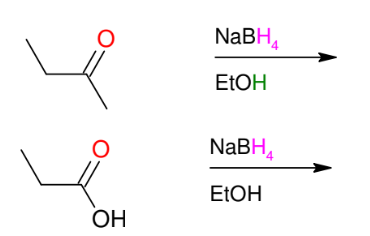
Hydride reduction of carbonyls (mild conditions - NaBH4 as reagent)
*reduces ONLY
aldehydes and
ketones
*use alcohols as a
solvent.
*KNOW MECHANISM
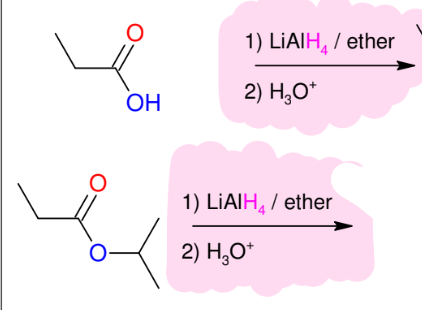
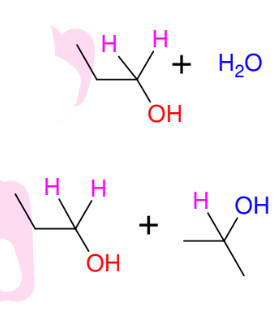
Hydride reduction of carbonyls (strong conditions - LiAlH4 as reagent)
*reduces aldehydes,
ketones, esters, acid
halides, carboxyllic
acids (ALL Carbonyls)
*Use ethers solvents
*Acid 2nd step
*KNOW MECHANISM
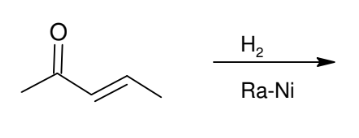
Raney Nickel
Reduces both carbonyl and alkene
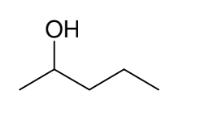
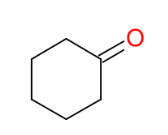
Oxidation of alcohols 2° alcohols into ketones
Makes ketone from 2° alcohols
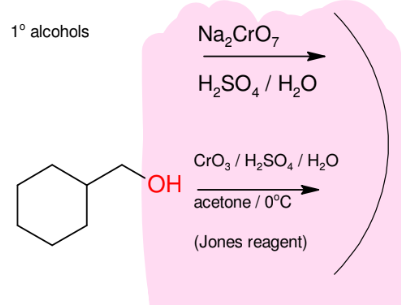
Oxidation of alcohols 1° alcohols into carboxylic acid group
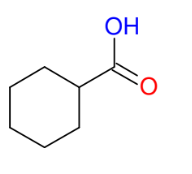
Makes carboxylic acid from 1° alcohols
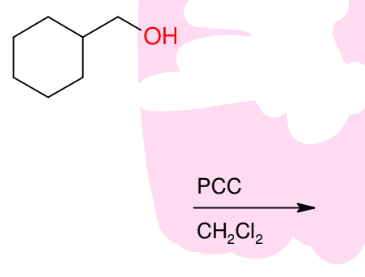
Oxidation of alcohols 1° alcohols into aldehyde/formaldehyde group
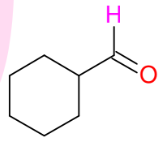
Makes aldehyde from 1° alcohols
*PCC is the only one
that can isolate
the formaldehyde.

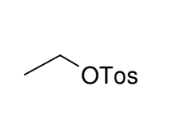
Formation of the Tosylate Ester
OH group replaced by OTos
*RETENTION from
where alcohol was
originally (SN2 purposes)
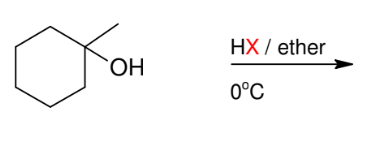
Formation of alkyl halide from 3° alcohols
OH group replaced by X
For 3° only
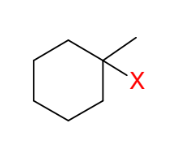
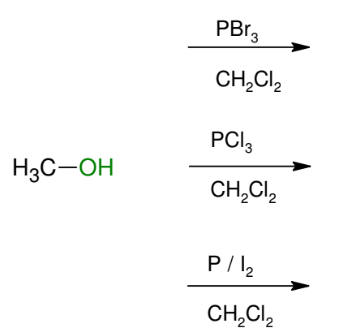
Formation of 1°/2° alkyl halides from 1°/2° alcohols
OH group replaced by X
For 2° and 1° only
*Basically an SN2
reaction. (Inversion
from original alcohol)
*Can also use SOCl2
for Cl, but it undergoes
a special mechanism!
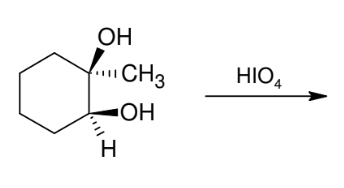
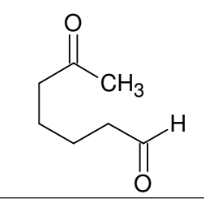
Unique cleavage with HIO4 of syn vicinal diols
*Vicinal diols MUST
be syn
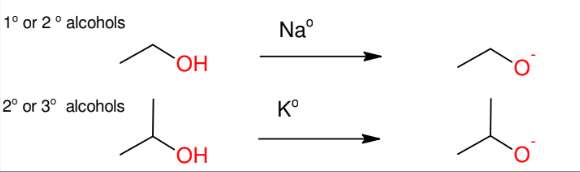
Formation of Alkoxide Anion
Will be used for williamson ether synthesis
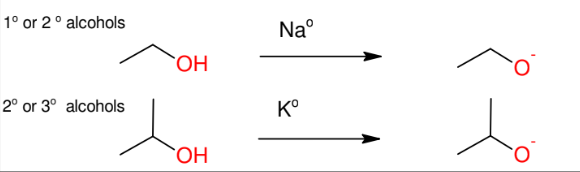
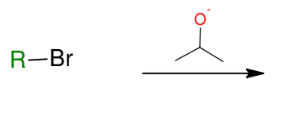
Williamson ether synthesis
Using alkoxide anion, create ether
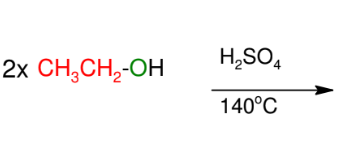
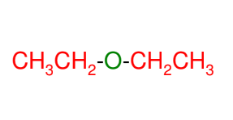
Ethers from intermolecular dehydration
Another method of forming ether
*MUST be identical
alcohols or else you
will get a mixture!!!
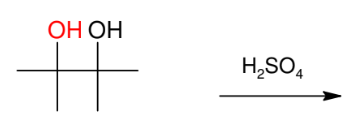
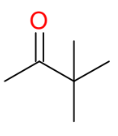
Pinacol - Pinacolone Rearrangement
*Need vicinal diols
*Know mechanism
(methyl shift!)
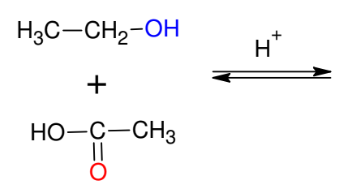
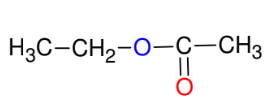
Fischer Estherification
*CAN USE ACID
HALIDE (i.e. CH3COCl) instead of
carboxyllic acid!!!