Hematology Final Review
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
Preanalytical phase testing
Any factors affecting the sample prior to testing
Proper identification
Collection procedure
Transportation requirements
Analytical phase testing
encompasses all steps involved in the actual testing of a specimen, starting with preparation and ending with the verification of results
Postanalytical phase testing
Anything that occurs after the test has been ran
Proper documentation
Critical reporting
Calculations post dilution
Reference values
a set of numerical limits—usually the central 95% of results—derived from testing a large group of healthy individuals
Delta checks
Comparison of current patient result and the patient’s previous result
Difference between abnormal and critical values
Abnormal values are test results outside the established normal reference range requiring follow-up, while critical values (or panic values) indicate a life-threatening, dangerous condition requiring immediate medical intervention
Calculating RBC indices
MCV=Hematocrit/RBC*10
MCH= Hemoglobin/RBC*10
MCHC=Hemoglobin/hematocrit*100
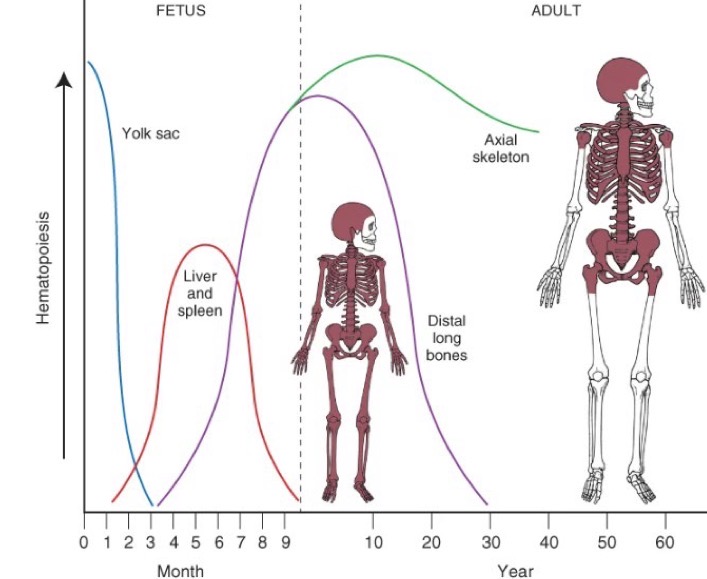
Hematopoiesis sources throughout life cycle
Yolk sac: mesoblastic period—> Liver: hepatic period—>Spleen: hepatic period—> Bone marrow
4 main functions of the spleen
Hematopoiesis
Reservoir
Filtration
Immunologic
Bone marrow’s role in hematopoiesis
responsible for producing over 500 billion blood cells daily
Pluripotent stem cells—what they can differentiate to?
Embryonic Stem Cells
Induced Pluripotent Stem Cells
Role of EPO
Functions as an erythroid growth factor
stimulates RBC production
Primary bone marrow source for collection
Iliac crest
Understanding CBC results
High RBC/Hb/Hct: Potential dehydration, smoking, or heart/lung disease.
Low RBC/Hb/Hct (Anemia): Possible bleeding, iron deficiency, or chronic disease.
High WBC (Leukocytosis): Likely infection, inflammation, or leukemia.
Low WBC (Leukopenia): Potential bone marrow issues, autoimmune disorders, or medication reaction.
High Platelets: Potential infection or iron deficiency.
Low Platelets: Risk of bleeding, possible liver issues, or medication side effects.
Approximate life span of a RBC
120 days
Anisocytosis
Variation in size
Poikilocytosis
Variation in shape
Polychromasia
Gray/blue in color
Hypochromia
Larger central pallor than normal red cell, >3µm
Spherocytes
Compact RBCs
Small, dense, and dark
Spherical RBCs
Sickle cells
Reversible: more rounded, half-moon shaped
Irreversible: crescent shaped and pointed projections
Ovalocytes
Egg shaped
Thalassemia
Megaloblastic anemia
Elliptocytes
abnormally elongated, oval, or "cigar-shaped" red blood cells caused by membrane protein defects
Target cells
Bull’s eye—shaped cell
Stomatocytes
Elongated area of central pallor
cup-shaped red blood cells with a characteristic slit-like or "fish-mouth" area of central pallor
Difference between acanthocytes & echinocytes (burr cells)
Acanthocytes smaller red surrounded by uneven thorn-like spicules
3-9 spikes
Echinocytes have numerous, regularly spaced, short, sharp projections (burr cells) typically caused by uremia or storage artifacts
Howell-Jolly bodies
small, dark purple, spherical inclusions in red blood cells that consist of residual DNA remnants.
Pappenheimer bodies
abnormal, iron-containing purple/blue granular inclusions within red blood cells, representing excess iron not incorporated into hemoglobin
Basophilic stippling
RNA & mitochondrial remnants
Significance of reticulocyte count and when is it used?
measures immature red blood cells to evaluate how quickly the bone marrow produces them
It is primarily used to diagnose the cause of anemia, distinguish between marrow production issues and blood loss, and monitor treatment responses
Heme molecule structure
Four iron atoms in the ferrous state (Fe2+) in the center
Iron in the ferric state (Fe3+) cannot bind oxygen
Porphyrin ring
Globin molecule structure
Amino acids linked together
Forms a polypeptide chain
What kind of iron is required for properly functioning hemoglobin molecules
Ferrous iron (Fe2+)
Purpose and function of hemoglobin
Primarily oxygen delivery
Secondarily to pull CO2 away from the tissues
Hemoglobin loads oxygen 1:1 in oxygen rich environments
Unloading occurs in oxygen poor environments
Difference between Hgb F, Hgb A, & Hgb A2
Hgb A is the adult hemoglobin type
Hgb F is the main fetal hemoglobin
Hgb A2 is a minor hemoglobin component
Main hemoglobin type in fetal development and infancy
Hgb F
Main hemoglobin type in adults
Hgb A
Why is carboxyhemoglobin so dangerous
It prevents blood from carrying oxygen
No releasing of the oxygen to the tissues
Why is sulfhemoglobins so dangerous
Can be toxic at low levels
hemoglobin doesn’t bind to O2 instead sulfhemoglobin
Extravascular hemolysis
Accounts for 90% of hemolysis
RBCs are destroyed and phagocytized
Occurs in the:
Spleen
Liver
Lymph nodes
Bone marrow
Intravascular hemolysis
Accounts for 10% of hemolysis
Lysed directly in the blood vessel
Types of microcytic anemias
IDA
Sideroblastic
Thalassemia
PBS characteristics of IDA
Microcytic and hypochromic
Small and deficient in hemoglobin
PBS characteristics of Sideroblastic anemia
Presence of pappenheimer bodies
Low reticulocyte count
PBS characteristics of Thalassemias
marked microcytosis
hypochromia
target cells
basophilic stippling
What classifies as macrocytic anemia
low hemoglobin
High MCH
Normal MCHC
What does asynchrony in the BM mean
a maturation defect where the cell nucleus develops slower than the cytoplasm
Differentiation between megaloblastic and non-megaloblastic
Megaloblastic MCV is extremely high whereas Non-megaloblastic MCV is slightly elevated
Megaloblastic anemia is caused by impaired DNA synthesis via folic acid and Vitamin B12 deficiency
Nonmegaloblastic anemia is caused by thyroid issues and liver disease
What organ causes spherocytes, how is this done
The spleen
An antibody is attached to RBCs and is sheared off as it passes through the spleen, taking some of the membrane with it
Lifespan of spherocytes
10-30 days
What is a xerocyte
dehydrated, rigid RBCs that appear concentrated on one end
What is aplastic anemia
Hypoproliferative disorder
Pancytopenia
Stressed BM with decreased cellularity
How to combat CBC irregularities from CAD
strict temperature management and specialized laboratory handling to prevent artificial, false-low red blood cell counts
How do sickle cells form
due to an inherited mutation in the beta-globin gene
What are the implications of a sickle cell crisis
Viruses
dehydration
fever
stress
Bone necrosis
Which organ bears the burden of a sickle cell crisis
The spleen
Hemoglobin C crystals
Bars of gold
melt in splenic environments
Hgb C crystals—average lifespan of RBCs with this disease
40-60 days
Maturation sequence of the myelocytic lineage
Myeloblast—>Promyeloblast—>Myelocyte—>Metamyelocyte—>Band—>Segmented neutrophil
Relative vs. absolute WBC values
Relative is the % of each WBC identified during 100 cell count
Absolute takes WBC count into consideration by multiplying each WBC percentage by the patient’s total WBC
Mature lymphocytes
specialized white blood cells (B, T, or NK cells) that have completed maturation in primary lymphoid organs (bone marrow or thymus) and now circulate in the blood and peripheral tissues to defend the body.
Mature Eosinophils
Can appear at the myelocytic stage
Cytoplasm has large distinct red-orange specific granules with orange-pink cytoplasm
Eccentric nucleus that is bilobed
Allergy response
Mature basophils
coarse, clumped, bilobed nucleus
Cytoplasm is large with specific purple-black granules
Histamine response
Mature monocytes
Loose and lacy chromatin
Abundant grey-blue cytoplasm
Antigen presentation
Mature neutrophils
the most abundant type of white blood cell (50–70% of leukocytes) and act as the innate immune system's primary, short-lived defense, patrolling the blood for bacterial and fungal infections
neutrophilic precursor—Blast
Round, oval nucleus
light purple chromatin
small blue cytoplasm
2-4 nucleoli present
neutrophilic precursors—promyelocytes
Oval, round, flattened nucleus
light purple chromatin
Moderate blue cytoplasm with large blue-red granules
Large, prominent nucleoli
neutrophilic precursors—Myelocytes
Oval-indented nucleus
Denser, red-purple chromatin with a coarser appearance
specific or secondary granules present in cytoplasm
last stage capable of dividing
neutrophilic precursors—metamyelocytes
kidney bean shaped nucleus
pale blue to pinkish cytoplasm
No nucleoli
condensed chromatin
neutrophilic precursors—bands
C or S shaped chromatin
Brown-pink cytoplasm
Filament represents a meta but indentation is more than half of the nuclear margin
Left shift
Bone marrow is sending out younger and less mature cells
What causes increased neutrophils
Infections/inflammatory responses
Pregnancy
Stress response
Surgery
What causes increased eosinophils
Skin disease
Parasitic disease
Transplant rejections
Asthma
Allergies
What causes increased basophils
Myeloproliferative disorders
Renal disease
Ulcerative colitis
Hypersensitivity reactions
What causes increased monocytes
Malignancies
Bone marrow failure
Chronic infections
What causes increased lymphocytes
when the body is fighting an infection, reacting to inflammation, or experiencing significant stress
Toxic granulation in neutrophils
Direct response to lysosome enzyme production
Sometimes resembles Basophils
Vacuoles
Appear in cytoplasm
Prolonged drug exposure may lead to phagocytosis of granules
Possible sepsis
Dohle bodies
Cytoplasmic inclusions
Ribosomal RNA
Appear due to rapid or stressful production
rod-shaped, pale blue structures
Hypersegmentation
>5 lobes
Seen in megaloblastic processes
Accompanied by oval macrocytes
Pelger-Huet Anomaly
Peanut shaped or dumbbell shaped nucleus
Spherical with no lobes
May initially appear as bands or meta
Fairly common inherited disorder
Main differences in acute leukemias and chronic leukemias
Acute leukemia is more severe with quick and aggressive onset usually in blasts AND predominantly mature cells
Chronic leukemia is less severe with insidious onset usually in predominantly mature cells
Myelodysplastic syndrome
a group of blood cancers occurring when immature blood cells in the bone marrow do not mature properly, resulting in low blood cell counts
Which leukemia has the highest cure rate
Hairy Cell Leukemia
Collagen role in hemostasis
A potent stimulator for platelet activation
Acting as the primary initiator of blood clotting when vascular injury exposes subendothelial collagen to blood flow
Endothelial’s role in hemostasis
The endothelium acts as a dynamic interface between blood and tissues, playing a crucial role in maintaining hemostasis by balancing anticoagulation and procoagulation
How many platelets come from ONE megakaryocyte
2000 platelets
Difference between platelet plug and fibrin clot
A platelet plug is a rapid, temporary, and fragile seal formed during primary hemostasis.
A fibrin clot is a slower, stable, and strong meshwork of fibrin protein that reinforces the plug during secondary hemostasis, ensuring permanent repairs
Primary vs. secondary hemostasis
Primary hemostasis is the initial, rapid response to vascular injury, forming a temporary, weak platelet plug to stop bleeding.
Secondary hemostasis follows immediately, using the coagulation cascade to create a stable, insoluble fibrin mesh that reinforces the plug into a solid clot
Intrinsic vs. extrinsic pathways
Intrinsic pathway is a slower process and is quantitatively significant process that involves factors XII, XI, IX, and VII. aPTT measures this pathway
Extrinsic pathway is a rapid process that involves Factors VII and III. PT measures this pathway.
Why does dysfunctional vWF cause impaired platelet adhesion
it fails to act as the essential molecular bridge between exposed subendothelial collagen and platelets, preventing their capture and anchoring at injury sites
How do platelets “call in the troops?”
to stop bleeding and initiate healing primarily through a rapid, complex chemical signaling process
Afibrinogenemia vs. hypofibrinogenemia vs dysfibrinogenemia
Afibrinogenemia is homozygous autosomal recessive with <10 mg/dL in plasma and poor wound healing
Hypofibrinogenemia is the heterozygous form of afibrinogenemia with 20-100 mg/dL in plasma and severe postoperative bleeding characterizes it.
Dysfibrinogenemia is autosomal dominant and is inherited homo and heterozygously.
Why is thrombin a “jack of all trades”
It acts as the central hub of coagulation, switching between procoagulant (clotting) and anticoagulant (anti-clotting) roles, while also influencing inflammation and cell repair
What is the key component of clot dissolution
Plasminogen
What is the main fibrinolytic product measured in routine laboratories
D-dimer
Why is DIC so dangerous?
Destroys clotting factors and platelets as soon as they are activated
systemic excessive disposition of thrombi and hemorrhage
Why is heparin Xa superior to PTT
It directly measures heparin's anticoagulant activity, offering greater accuracy, faster time to therapeutic range, and fewer dose adjustments
What is measured for Warfarin and Coumadin therapy
the INR (International Normalized Ratio) and the PT (Prothrombin Time)
What is measured for heparin therapy
activated partial thromboplastin time (aPTT) to measure the blood's clotting time