information for bis M1
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
At pI, the isoelectric point, what is true about a molecule?
It will not
1- move in an electric field
2-bind to an ion exchange column
3- bind to another molecule electrostatically
pH = what? (In terms of pOH)
pH= 14 - pOH
Formula for pI
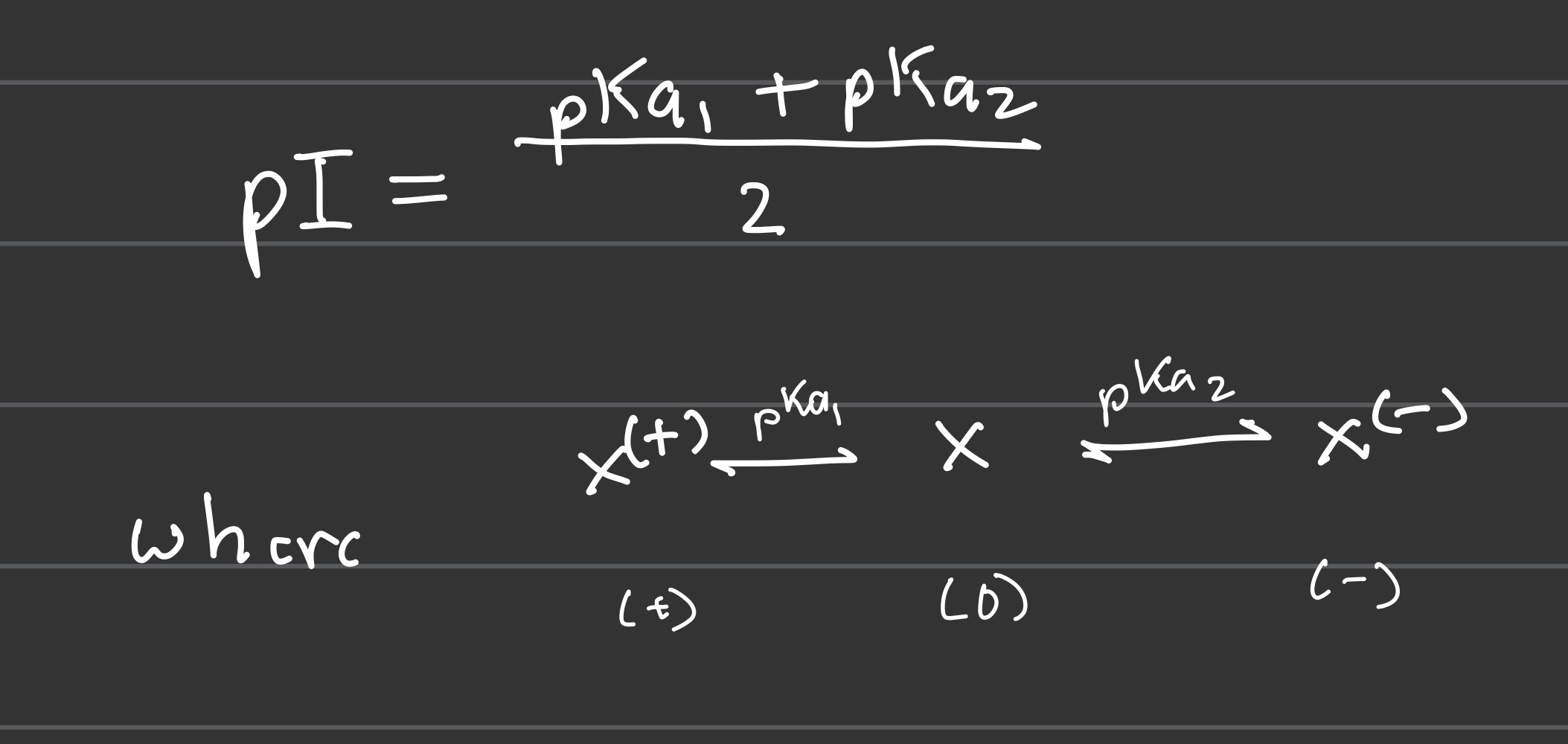
Acid and base proportion equations (a/b)

Which Ionizable AA are +/0 and which are 0/- ?
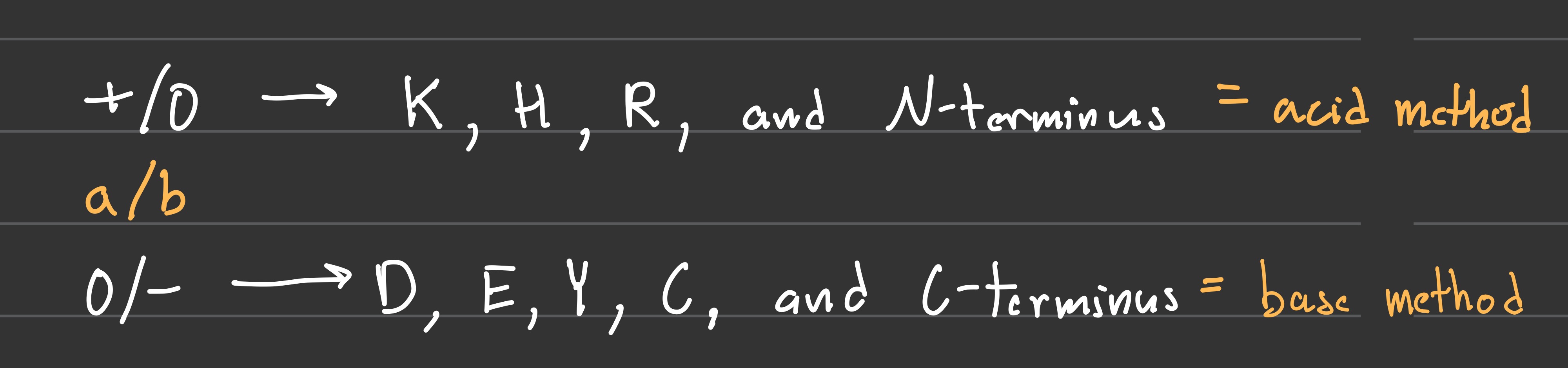
HH equation
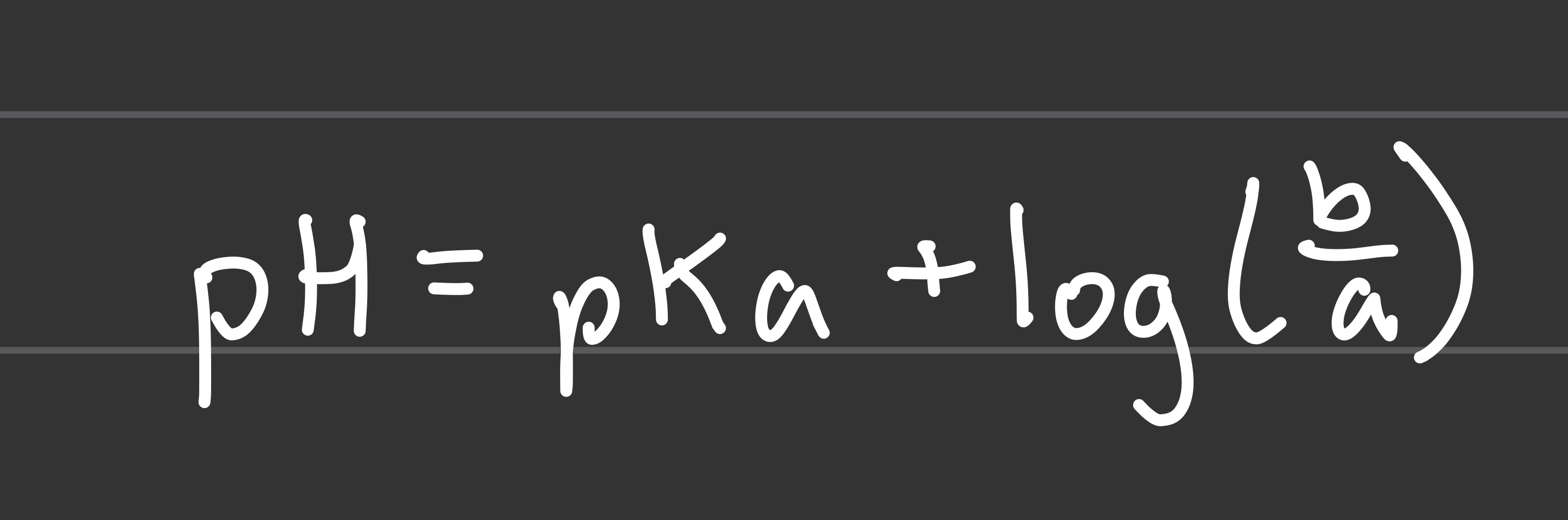
pH= and pOH= (basic concentration conversion)
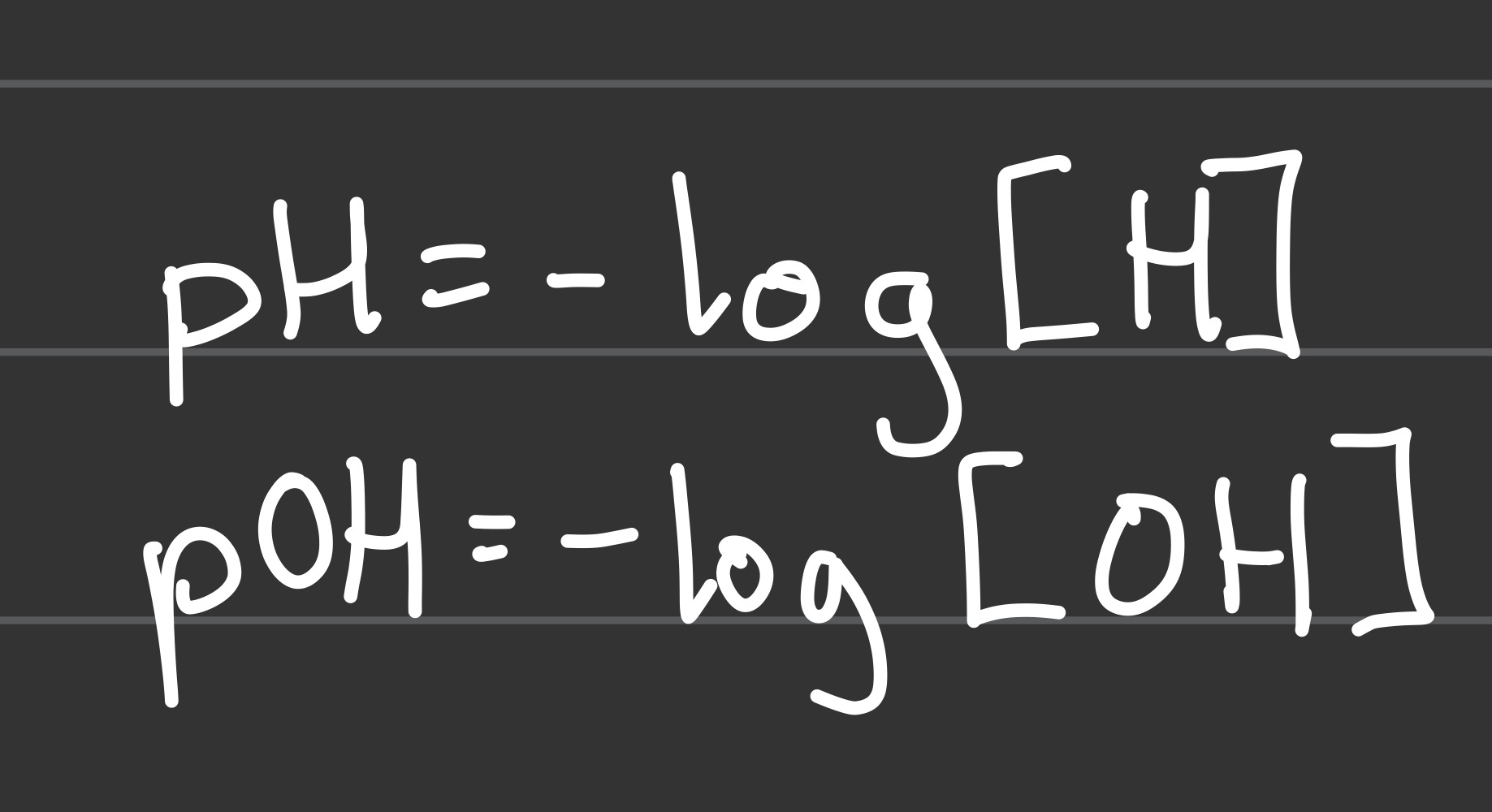
Ka and Kb in terms of pKa and pKb
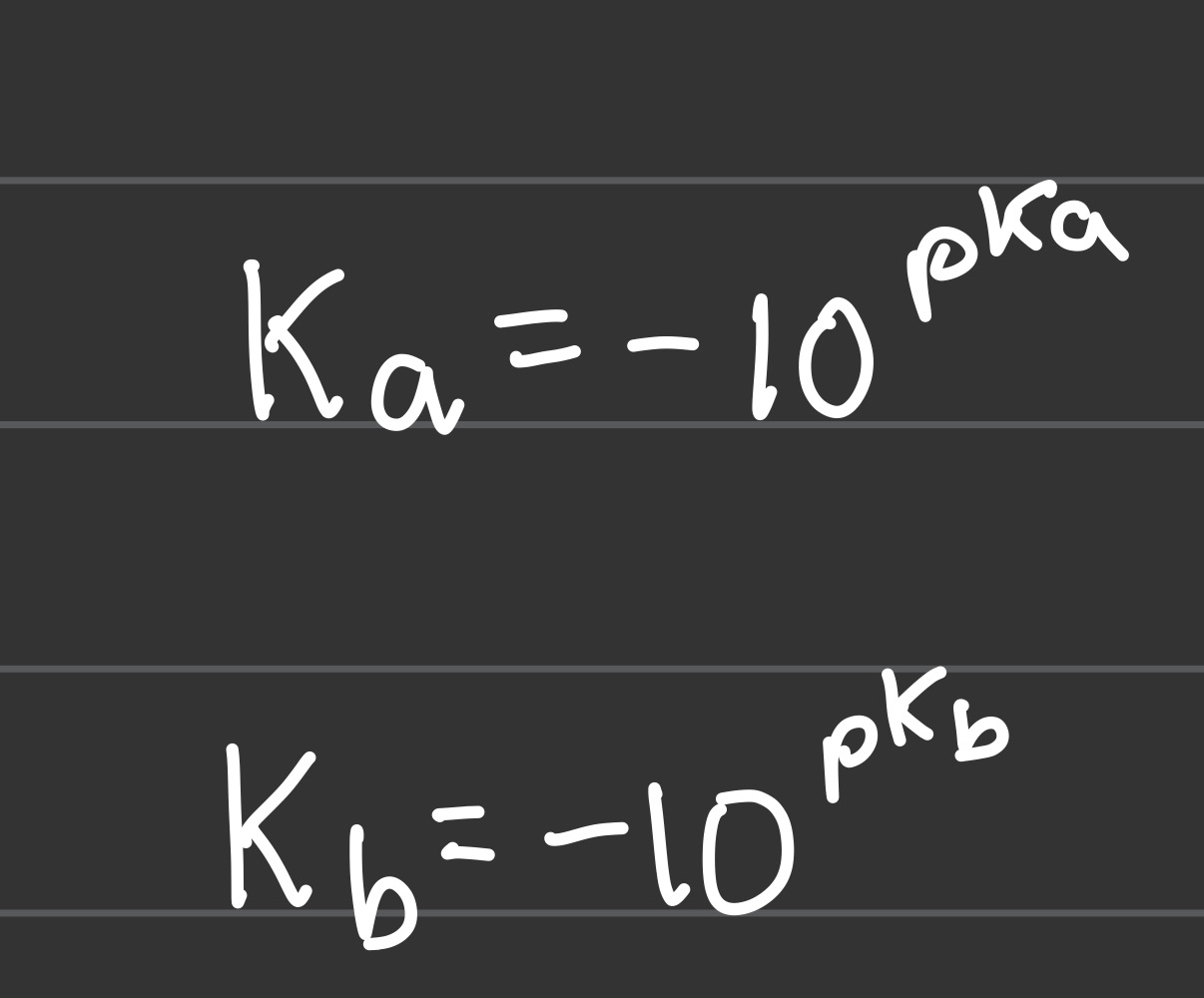
How to calculate pH for phosphate buffers b
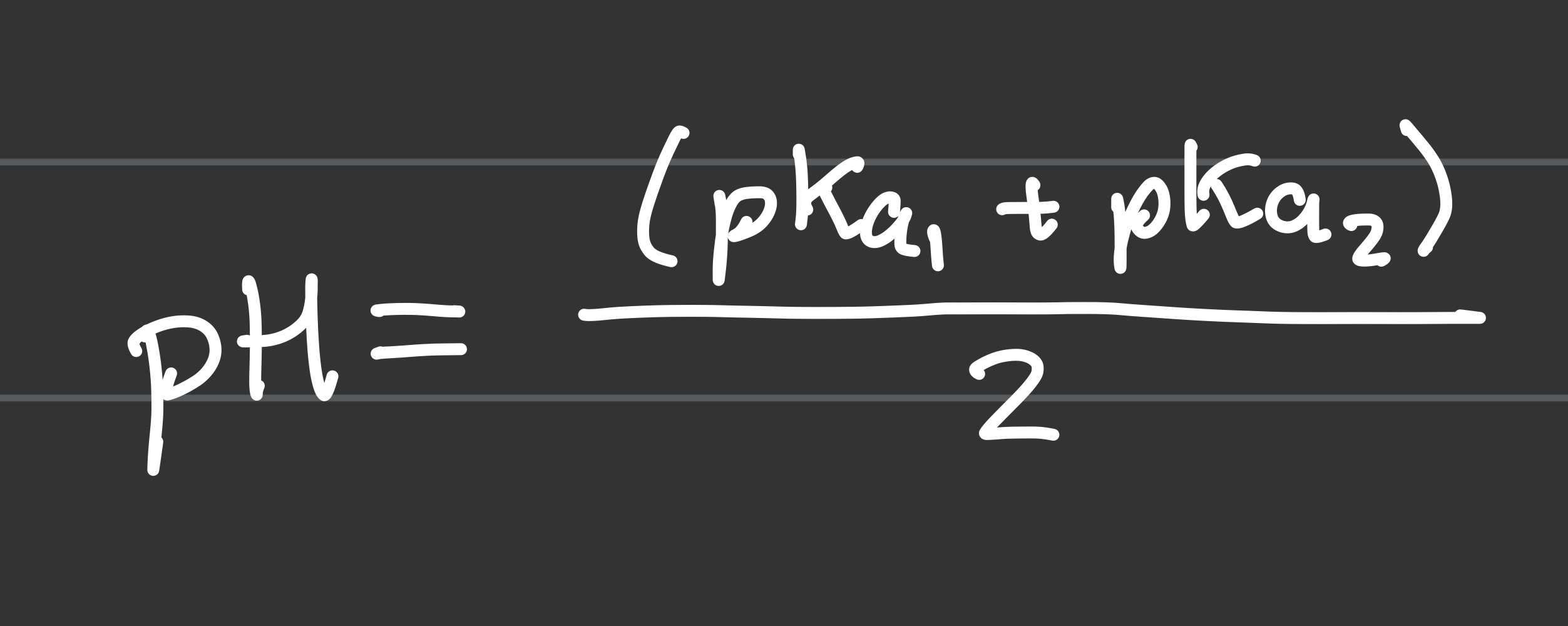
Ka and Kb (x equations)
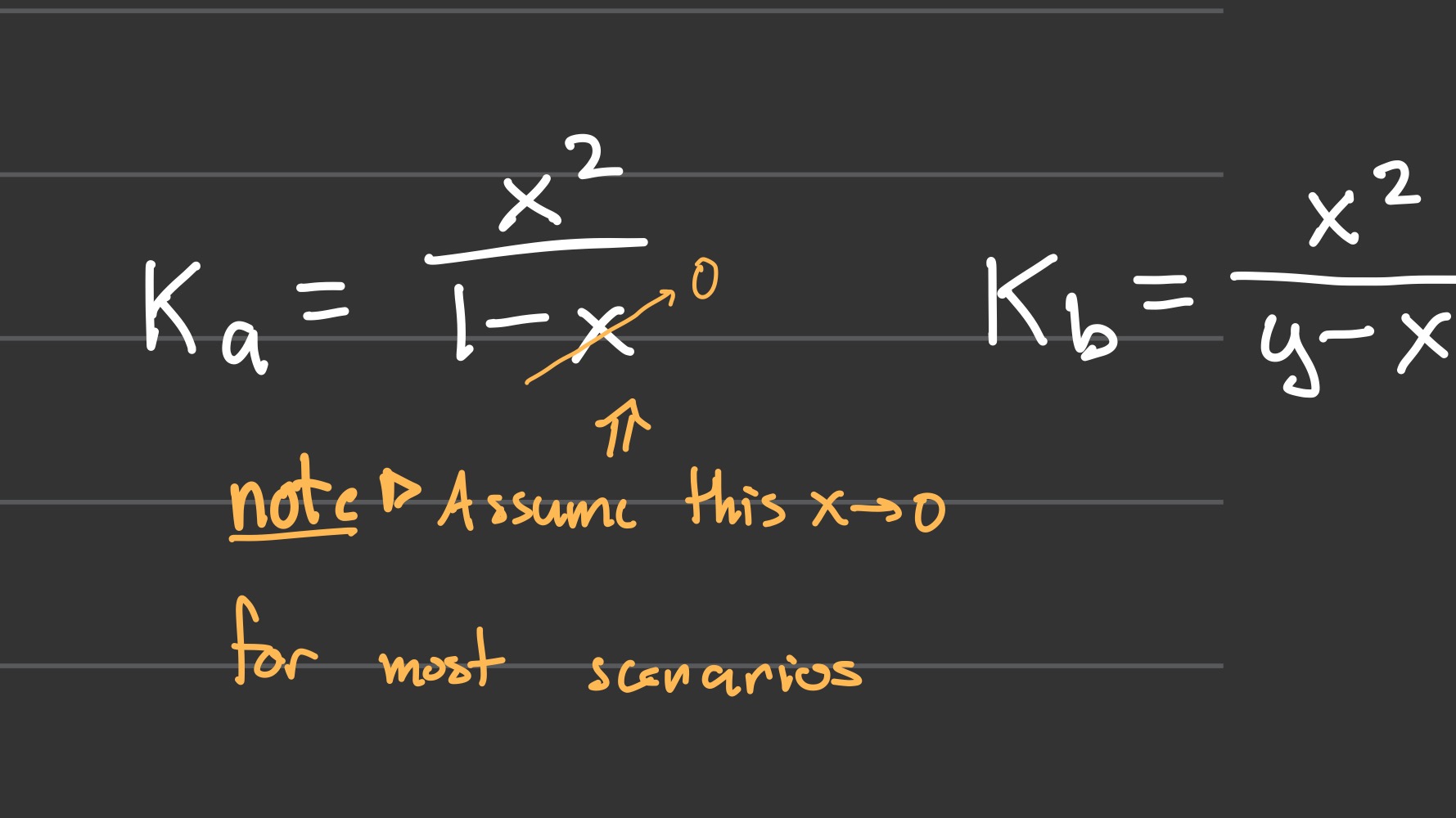
[H] or [OH] in terms of pH and pOH
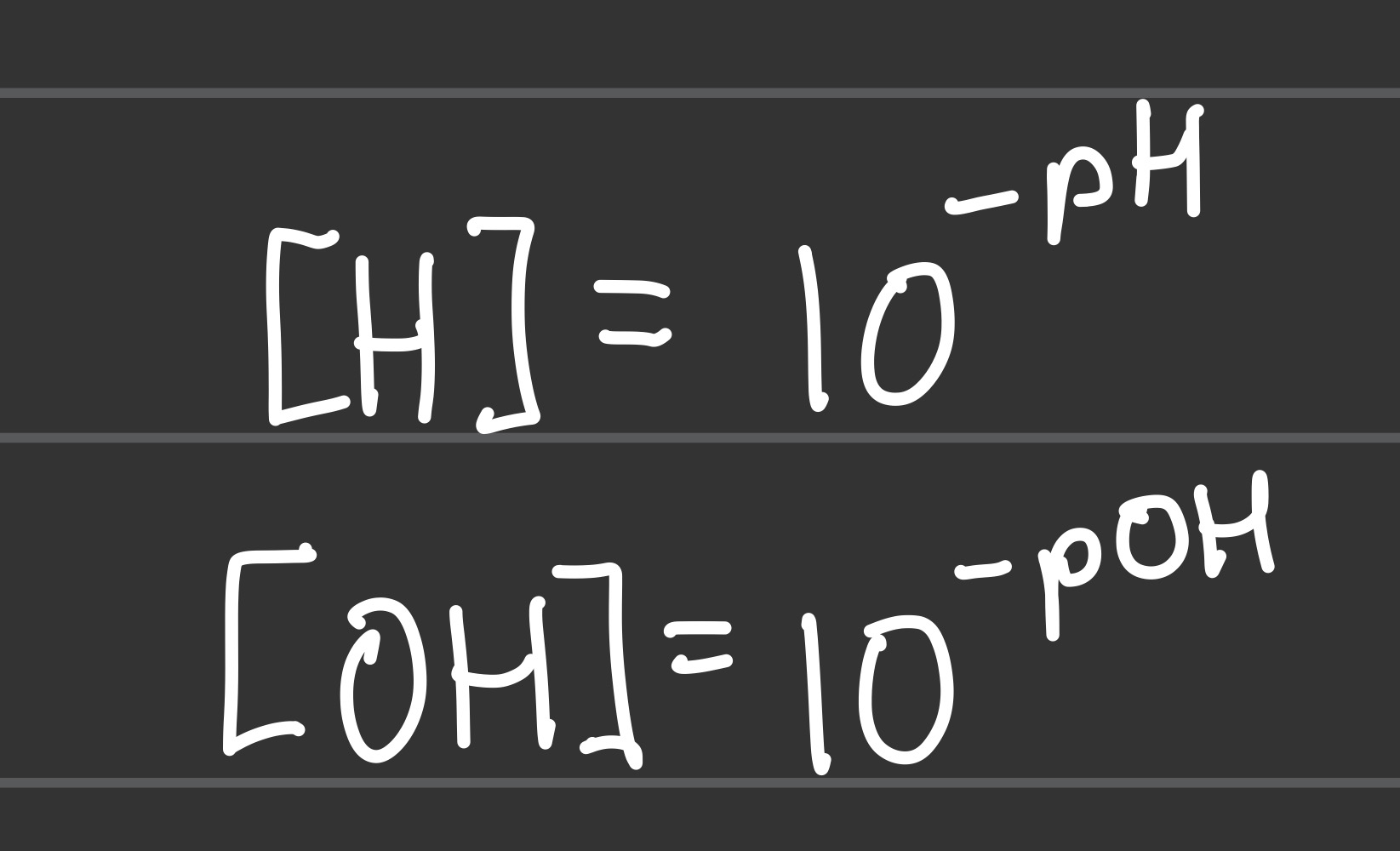
Statements about hydrophobic effect
-water forms clathrates around a hydrophobic compound
-decreasing the temperature caused water molecules to become more ice like
-the hydrophobic effect allows cells to have compartments
-the hydrophobic effect causes the entropy of water to increase
False statements about hydrophobic effect
-increasing temp diminished hydrophobic effect (opposite is true)
-pH and direction matter
-distance matters
-the hydrophobic effect does not allow proteins to fold correctly
-water molecules form strong covalent bonds around the hydrophobic compound
What does one peptide bond plane look like in an oligopeptide?
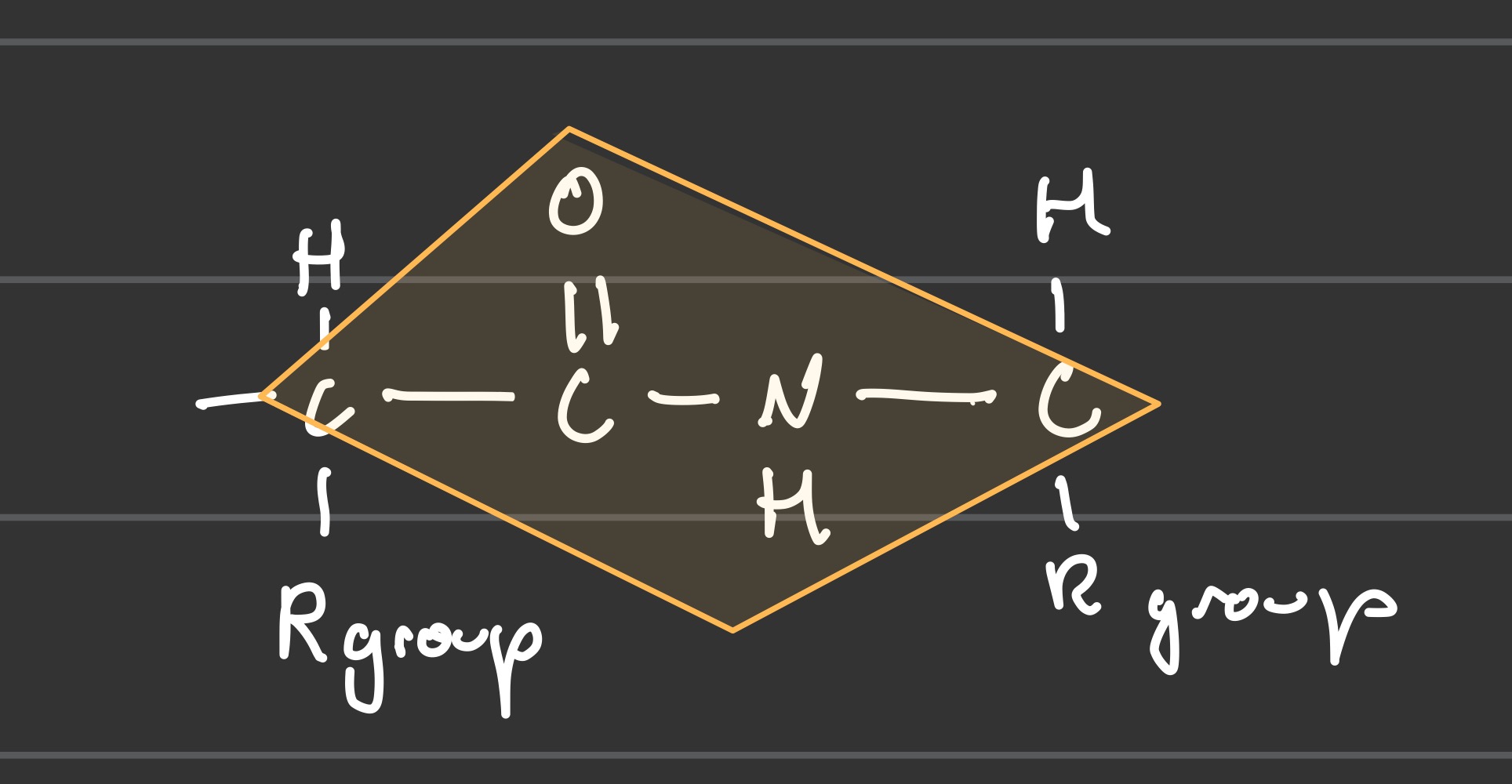
Why are atoms in a peptide bond in a plane shape?
Their resonance forces the peptide bond into a plane shape, as the double bond at the C=O can also resonate to become C=N. The peptide bond has a 40% double bond character
True statements about Myoglobin
-When O2 binds to it, the metal become planar relative to the porphyrin ring
-the proximal histidine forms a covalent bond due to the metal in the heme
-myoglobin has a very low P50 when assayed in vitro
-when Mb is in solution, its interior is dynamic
False stamens about myoglobin
-