Biochem Exam IV
1/87
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
88 Terms
Glycolysis
Occurs in the cytoplasm where ATP is produced by the oxidation of glucose. Requires investment of 2 ATP molecules, while a net gain of 2 ATP, 2 NADH, and 2 pyruvate are produced
Phosphorylated Intermediates
When transforming glucose in the steps of glycolysis, these items keep components in the cell through adding a phosphate group to them → cannot escape cell membrane this way
Hexokinase Reaction
First step that requires Mg2+ to bind the negative phosphates in ATP. Irreversible step that takes phosphate from ATP and attaches it to the 6th carbon on a glucose ring (makes glucose-6-phosphate)
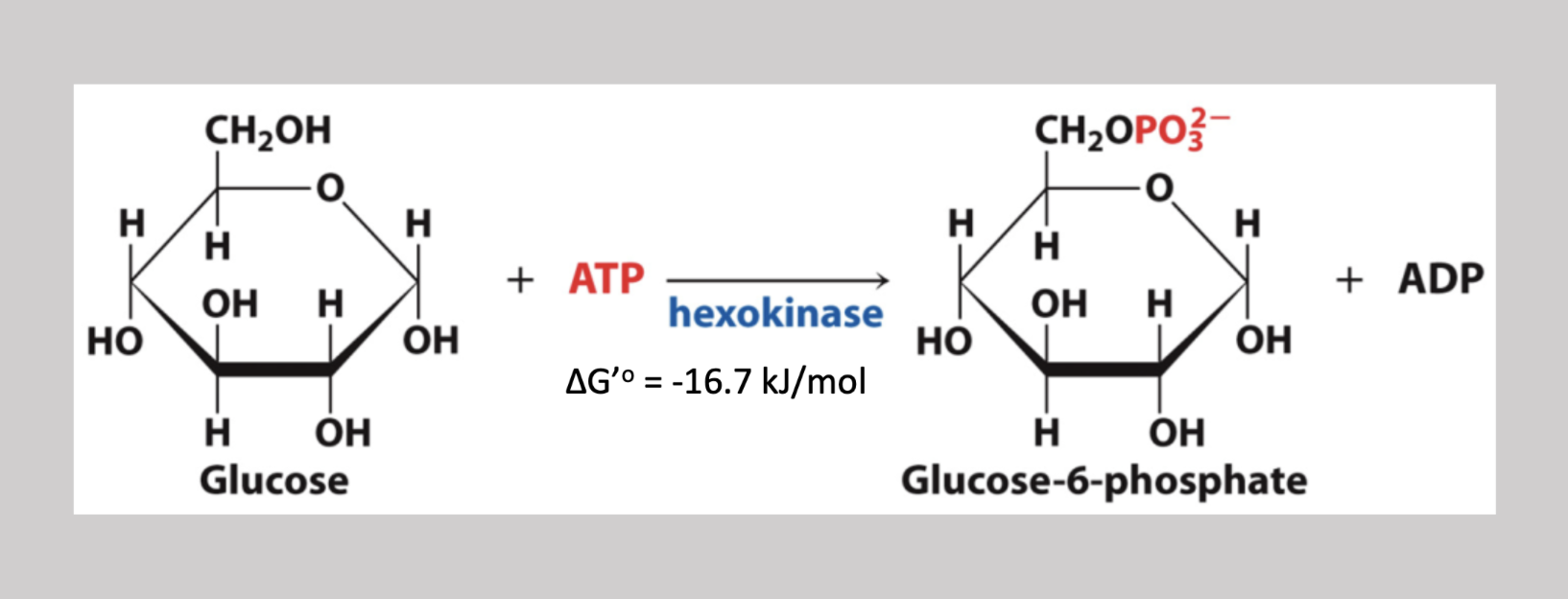
Phosphoglucose Isomerase reaction
Second step reversible reaction that transforms glucose-6-phosphate into fructose-6-phosphate → makes the H on carbon 1 CH2OH
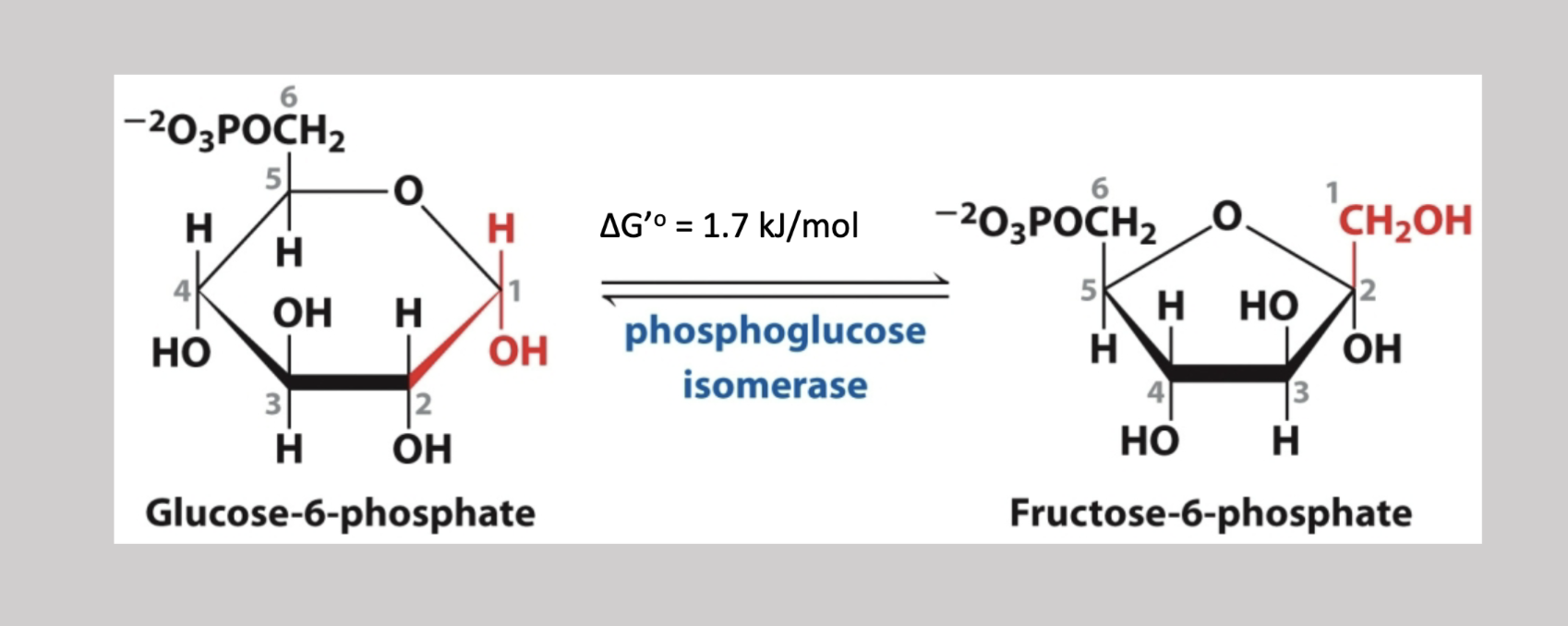
Phosphofructokinase Reaction
Third step irreversible reaction that adds a phosphate to carbon-1 from ATP on fructose-6-phosphate (generates fructose-1,6-biphosphate)
Fructose-2,6-biphosphate
Made by PFK-2 (NOT an enzyme of glycolysis) to regulate and turn on phosphofructokinase (PFK-1) → binds to PFK-1 to increase its affinity for fructose-6-phosphate. This is not generated when glucose levels are low and the cell wants to perform gluconeogenesis
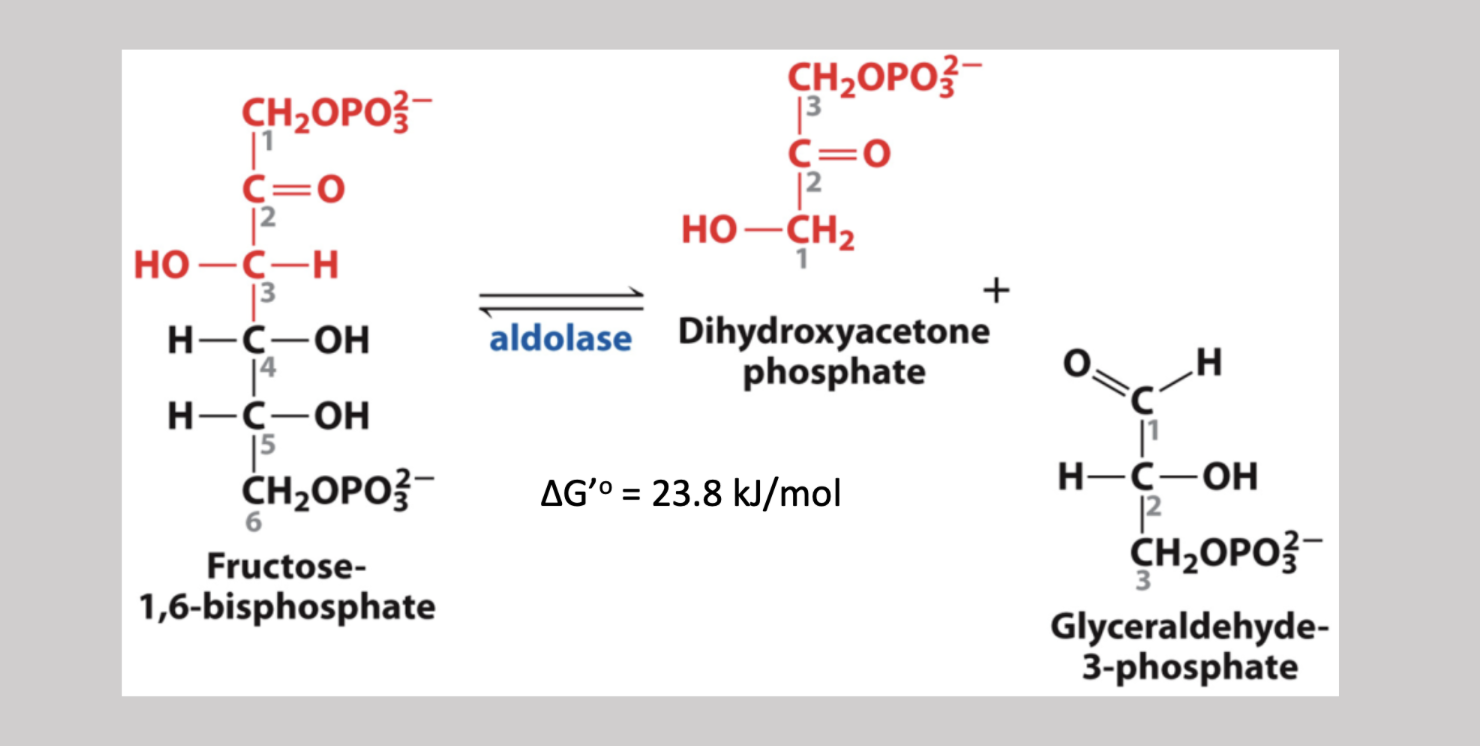
Aldolase Reaction
Fourth step reversible pathway since reactants are low in the cell that severs fructose-1,6-biphosphate into two 3 carbon sugars → dihydroxyacetone (ketone), and glyceraldehyde-3-phosphate (aldehyde)
Triose Phosphate Isomerase Reaction
Fifth step reversible pathway that converts dihydroxyacetone phosphate into an additional glyceraldehyde-3-phosphate in order for it to proceed in glycolysis → it is an induced fit catalytically perfect enzyme that obtains a very high affinity for its substrate

Glyseraldehyde-3-phosphate (GAP) Dehydrogenase Reaction
Sixth step reversible pathway that oxidizes and phosphorylates carbon-1, creating 1,3-Biphosphoglycerate, and reduces NAD+ to NADH

Phosphoglycerate Kinase
Seventh step reversible pathway that transfers the phosphate on carbon-1 of 1,3-Biphosphoglycerate, and attaches it to an ADP (creating ATP) → creates 3-phosphoglycerate
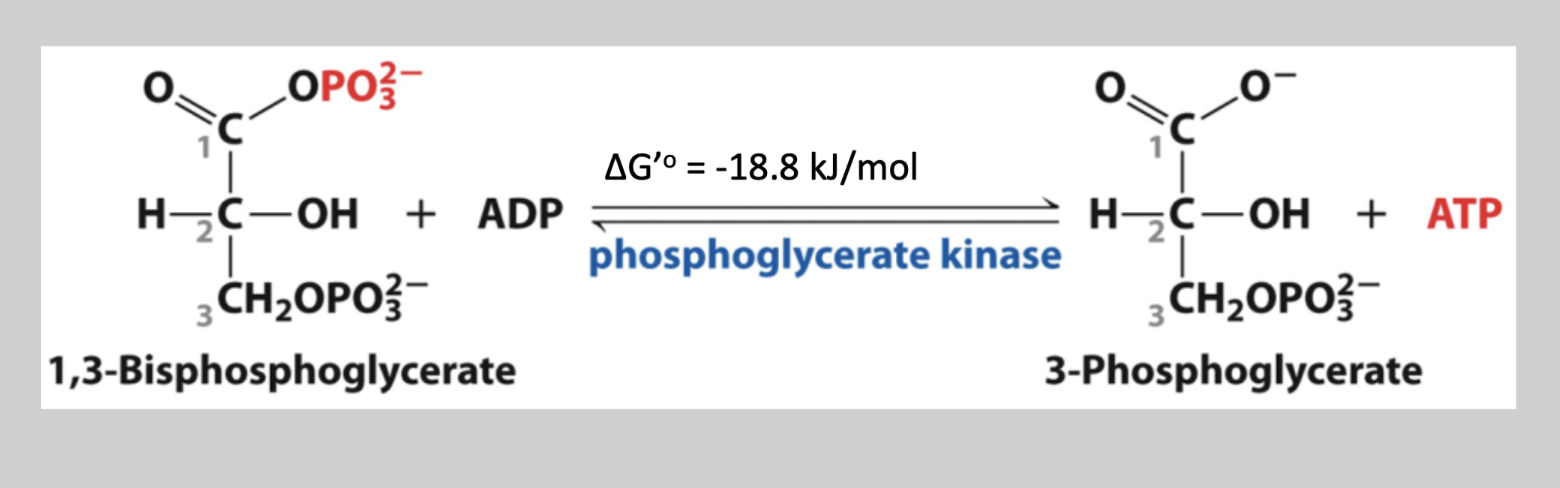
Substrate Level Phosphorylation
The formation of ATP by phosphoric group transfer from a substrate
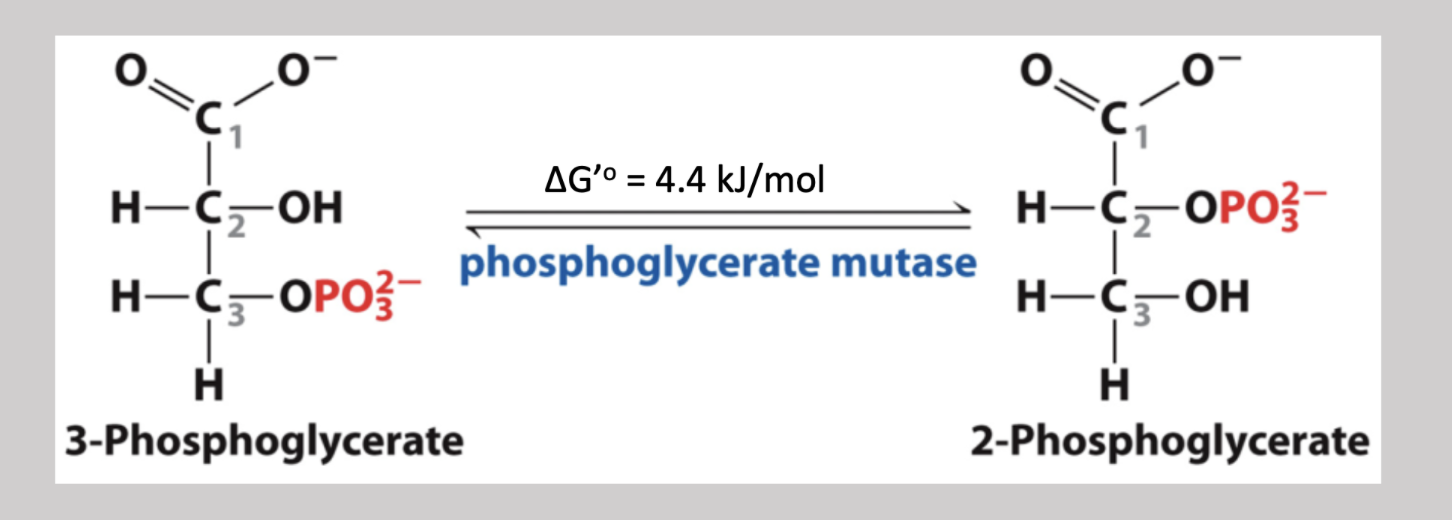
Phosphoglycerate Mutase Reaction
Eith step reversible pathway that transfers a phosphate group from carbon-3 to carbon-2 → 3-phosphoglycerate to 2-phosphoglycerate (isomerase reaction)
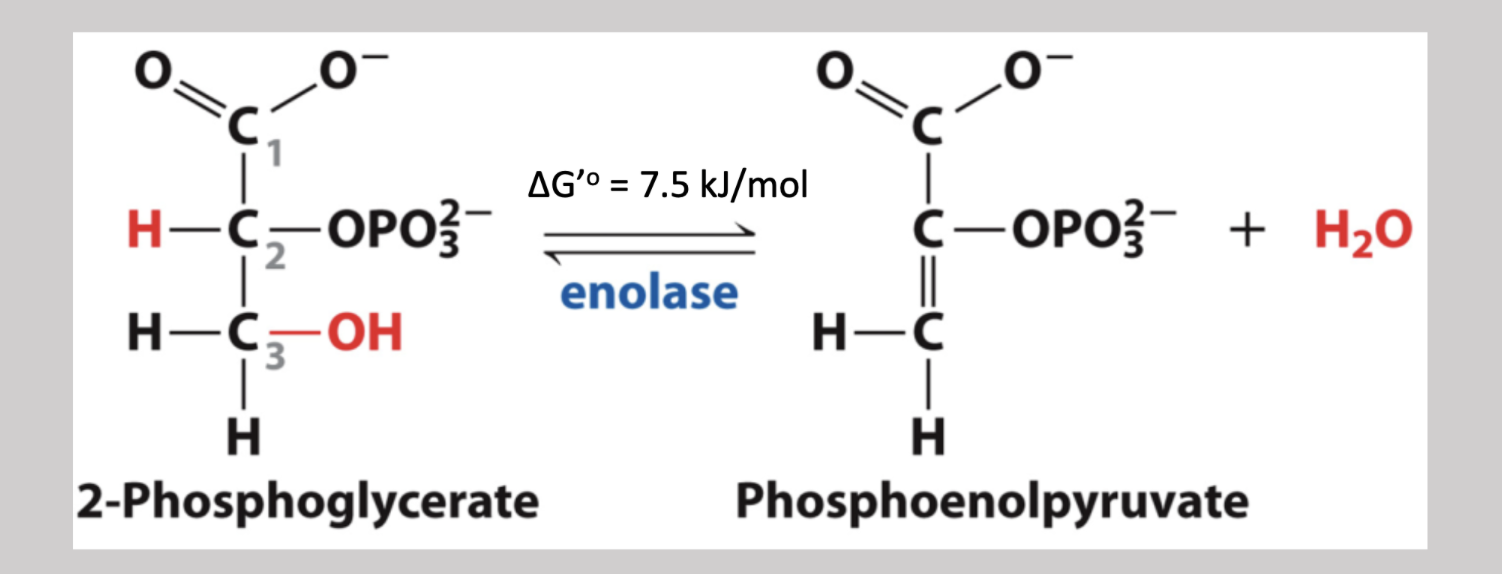
Enolase
Ninth step reversible dehydration pathway that removes an H from carbon-2 and an OH from carbon-3, and in turn generates a double C=C bond as well as yields a water molecule → 2-phosphoglycerate → phosphoenolpyruvate
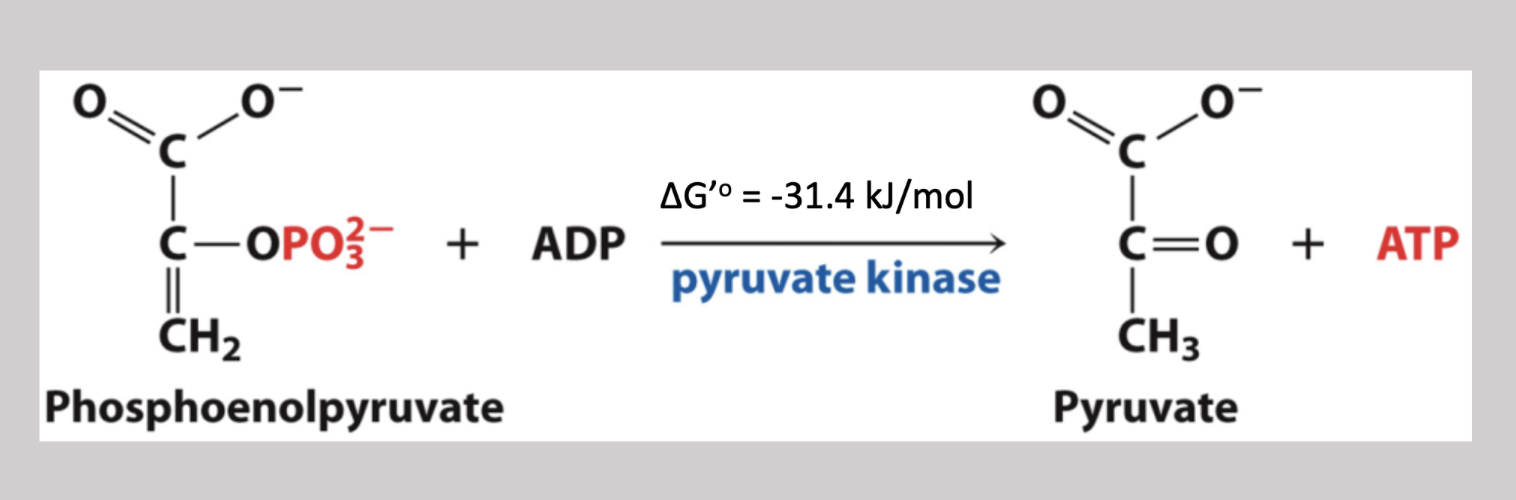
Pyruvate Kinase Reaction
Tenth step irreversible pathway that removes the phosphate on carbon-3 and attaches it to an ADP molecule (creating ATP) → phosphoenolpyruvate → pyruvate
Fates of Pyruvate
Under aerobic conditions, it’s oxidized into acetyl-CoA and under anaerobic conditions, it is reduced to lactate or ethanol. If necessary, it can also be converted into oxalacetate that is used to synthesize amino acids
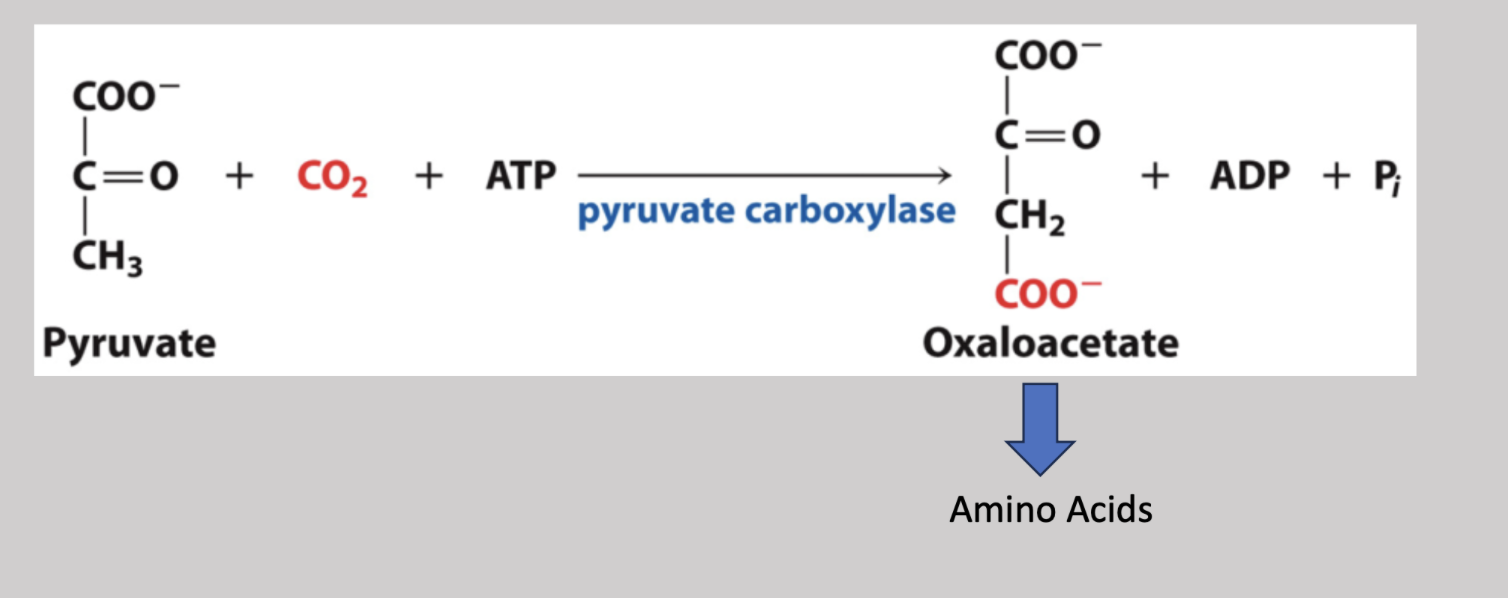
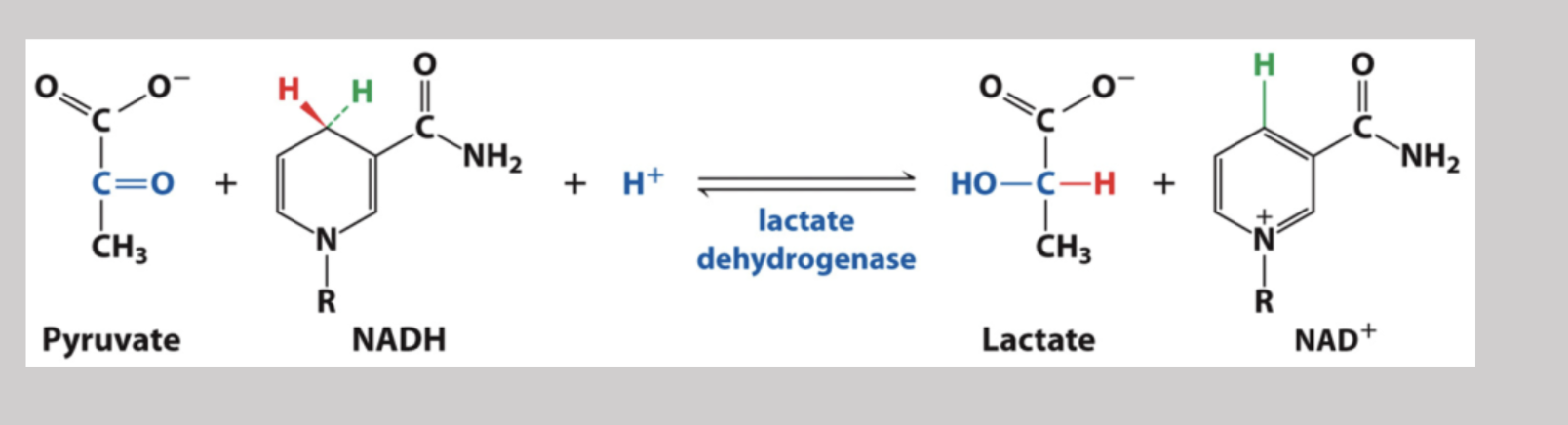
Lactate Dehydrogenase
During excercise, mammals utilize this enzyme to temporarily turn pyruvate into lactate by oxidizing NADH to NAD+ and therefore, reduces pyruvate
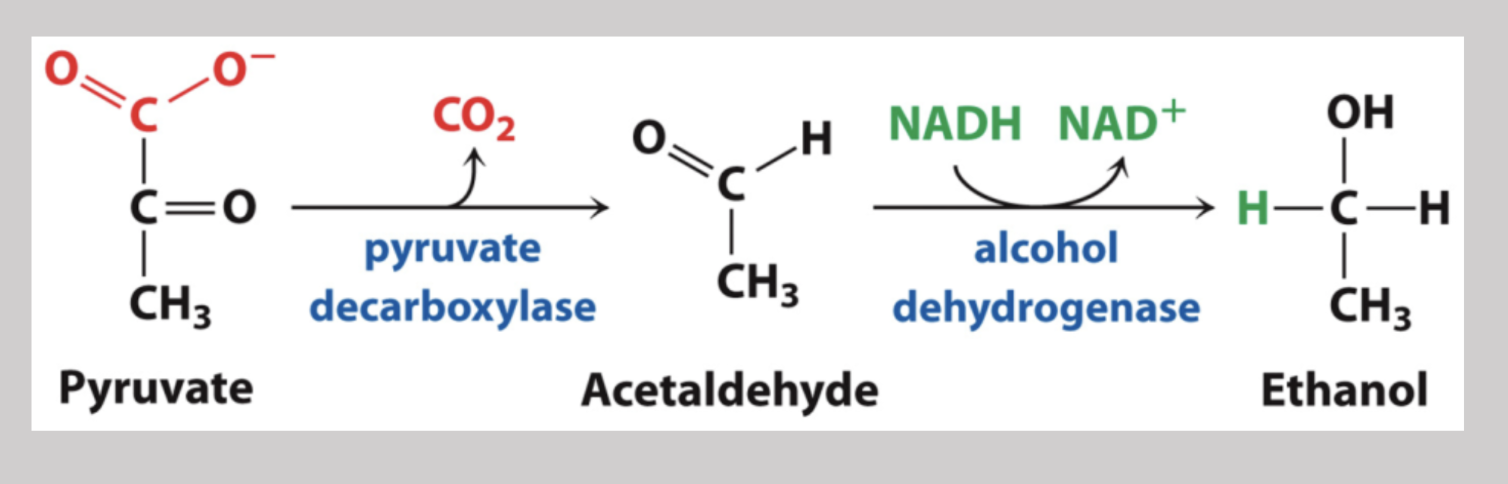
Regeneration of NAD+ in Yeast
Pyruvate generated into acetaldehyde and yields CO2 molecule by way of pyruvate decarboxylase, then utilizes alcohol dehydrogenase to oxidize NADH and in turn, reduce acetaldehyde into ethanol
Gluconeogenesis
The pathway that converts pyruvate and related three and four carbon compounds to glucose → mainly occurs in the liver of mammals, but also occurs in plants, fungi, and microorganisms as well
The First Bypass 1
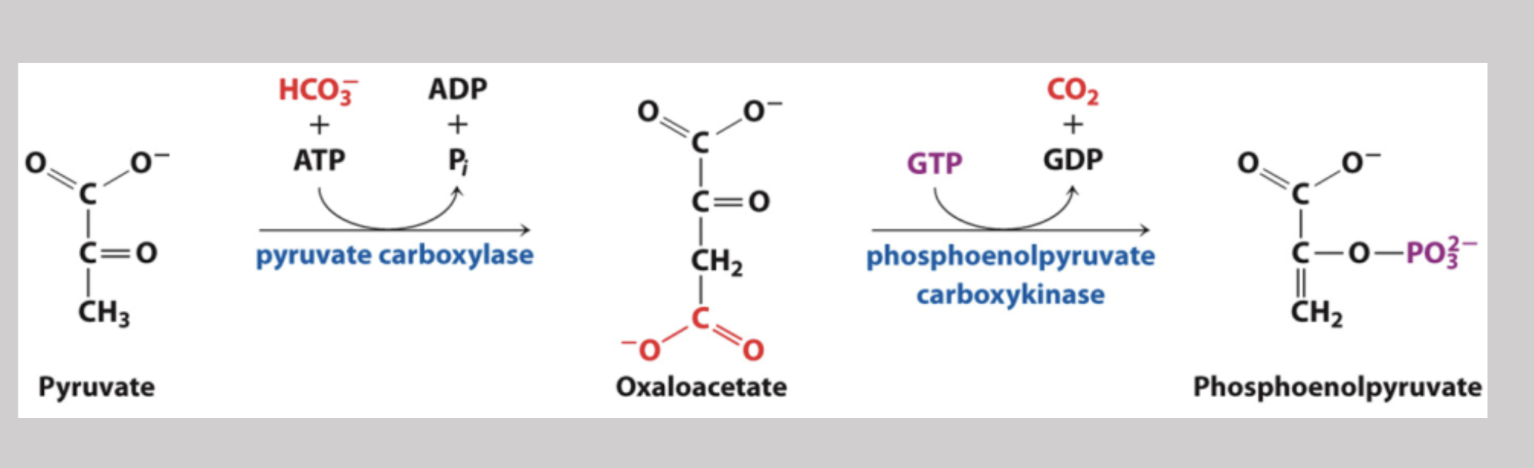
Step one in glyconeogenesis where pyruvate is transferred from the cytosol into mitochondria. Here, pyruvate carboxylase converts pyruvate to oxaloacetate and requires the coenzyme biotin along with ATP
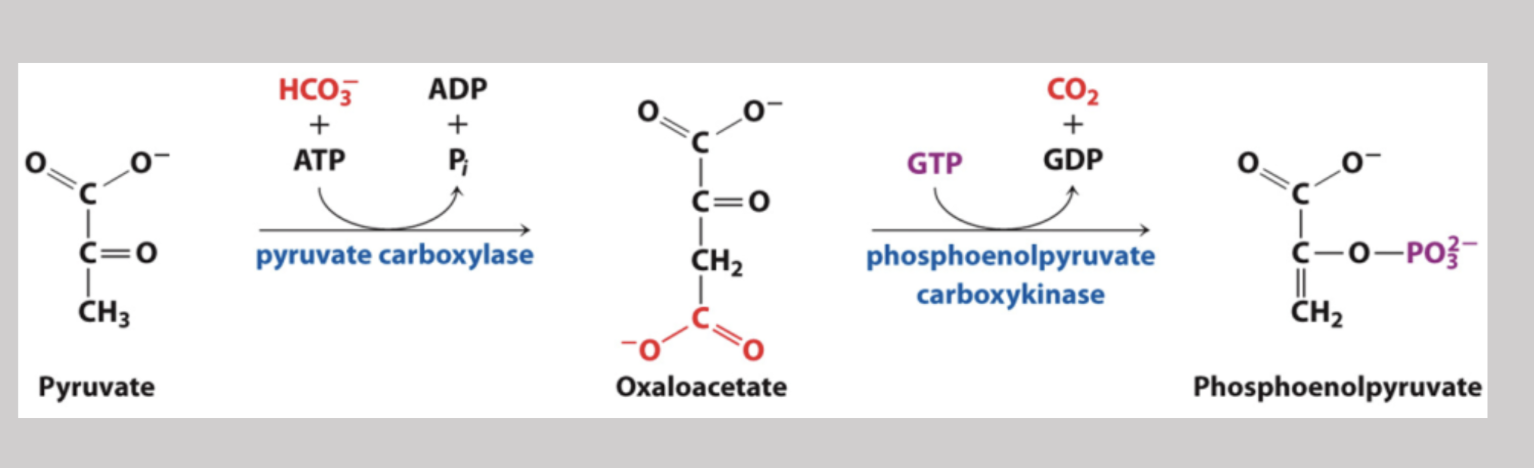
Phosphoenolpyruvate Carboxylkinase
Part of the first bypass in glyconeogeneis where oxaloacetate is converted into phosphoenolpyruvate → a phosphate is added, a CO2 is yielded, and the process oxidizes GTP to GDP (reversible under intracellular conditions)
Fructose 1,6-biphosphatase (FBPase-1)
The enzyme in step 7 of glyconeogenesis that performs fructose-1,6-biphosphate + H2O → fructose-6-phosphate + Pi. IS carried out by hydrolysis of the carbon-1 phosphate, requires Mg2+, and is essentially irreversible
Glucose-6-phosphatase
The enzyme utilized in the last step (third bypass) of glyconeogenesis that utilizes simple hydrolysis to generate glucose + Pi → requires Mg2+, found in the ER, and is an irreversible pathway
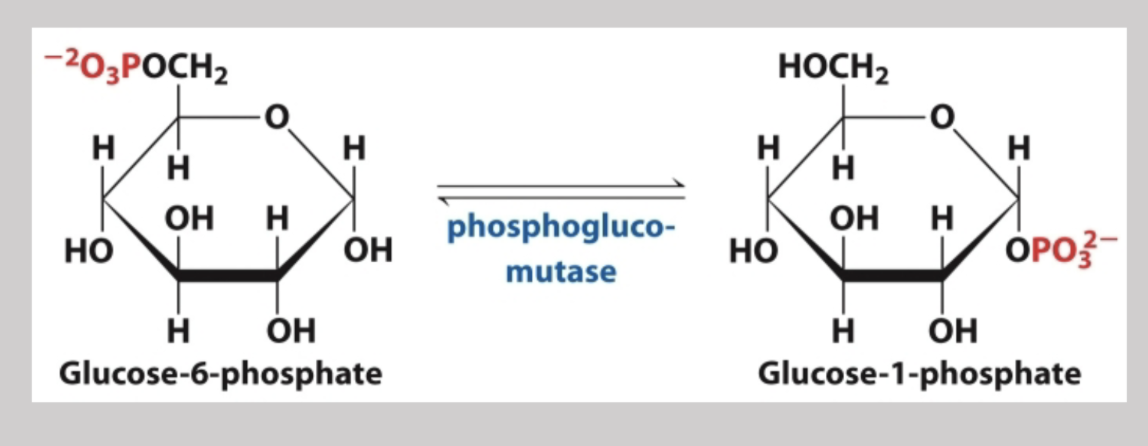
Glycogenolysis
The breakdown of cellular glycogen beginning with glucose-6-phosphate and then broken down into glucose-1-phosphate via phosphoglutomase
Glycogen Synthesis
Consumes the free energy from UTP by transforming glucose-1-phosphate into UDP-glucose utilizing UDP-glucose-phosphorylase and inorganic pyrophosphatase
Glycogen Synthase
Adds glucose to extend the glycogen polymer
Glycogen Branching Enzyme
Catalyzes the formation of the alpha1-6 bonds found at the branch points of glycogen
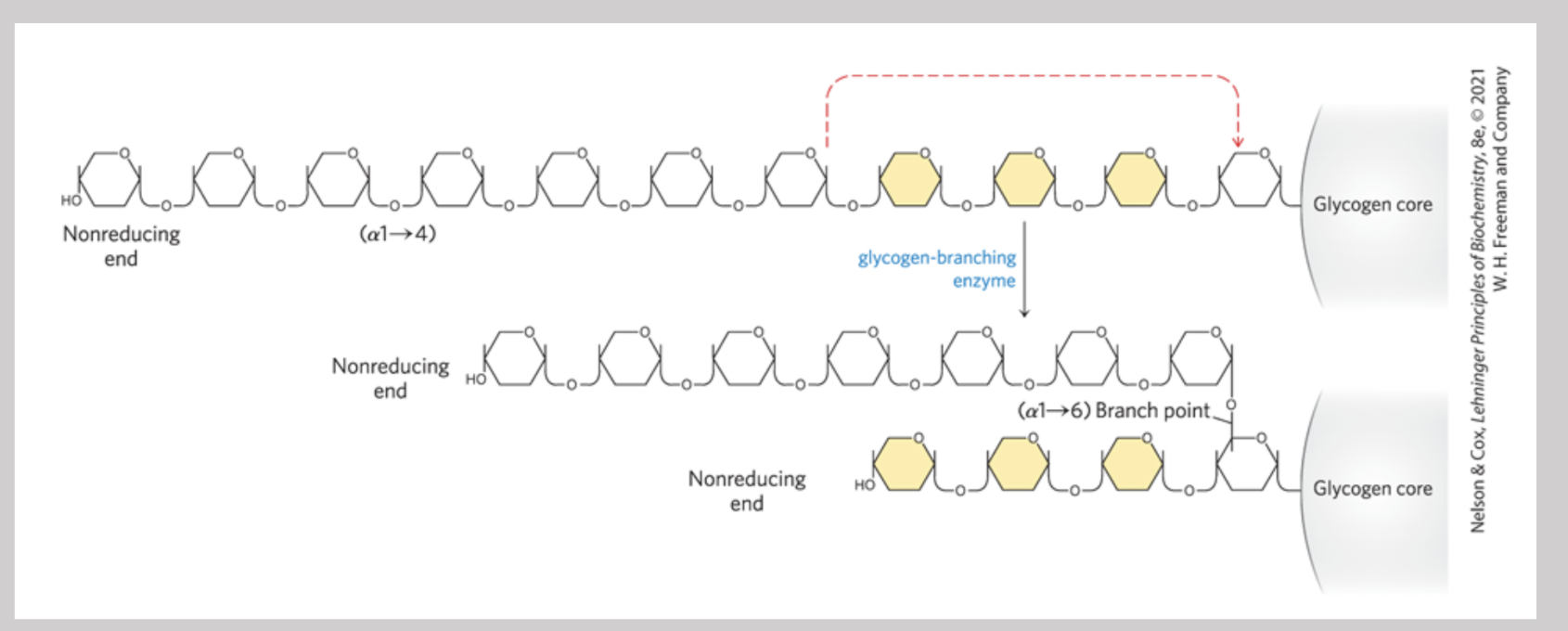
Glycogen Phosphorylase
Enzyme that catalyzes phosphorolytic cleavages at the non reducing ends of glycogen chains → requires a phosphate and acts repetitively until it reaches a point four residues away from an alpha1-6 branch point (makes glucose-1-phosphate)
Debranching Enzyme
Transfers branches onto main chains and releases the residue at the alpha1-6 branch as free glucose
Phosphoglucomutase
Catalyzes the reversible conversion of glucose-1-phosphate to glucose-6-phosphate
Pentose Phosphate Pathway
Alternative pathway for glucose oxidation that occurs in the cytosol of most cells and produces pentoses for nucleotide synthesis and NADPH for the biosynthesis of fatty acids. Begisn with glucose-6-phosphate, then gets oxidized in order to reduce NADP+, then 6-phosphogluconate dehydrogenase that gets oxidized to reduce more NADP+, then produces ribulose-5-phosphate (irreversible pathway)
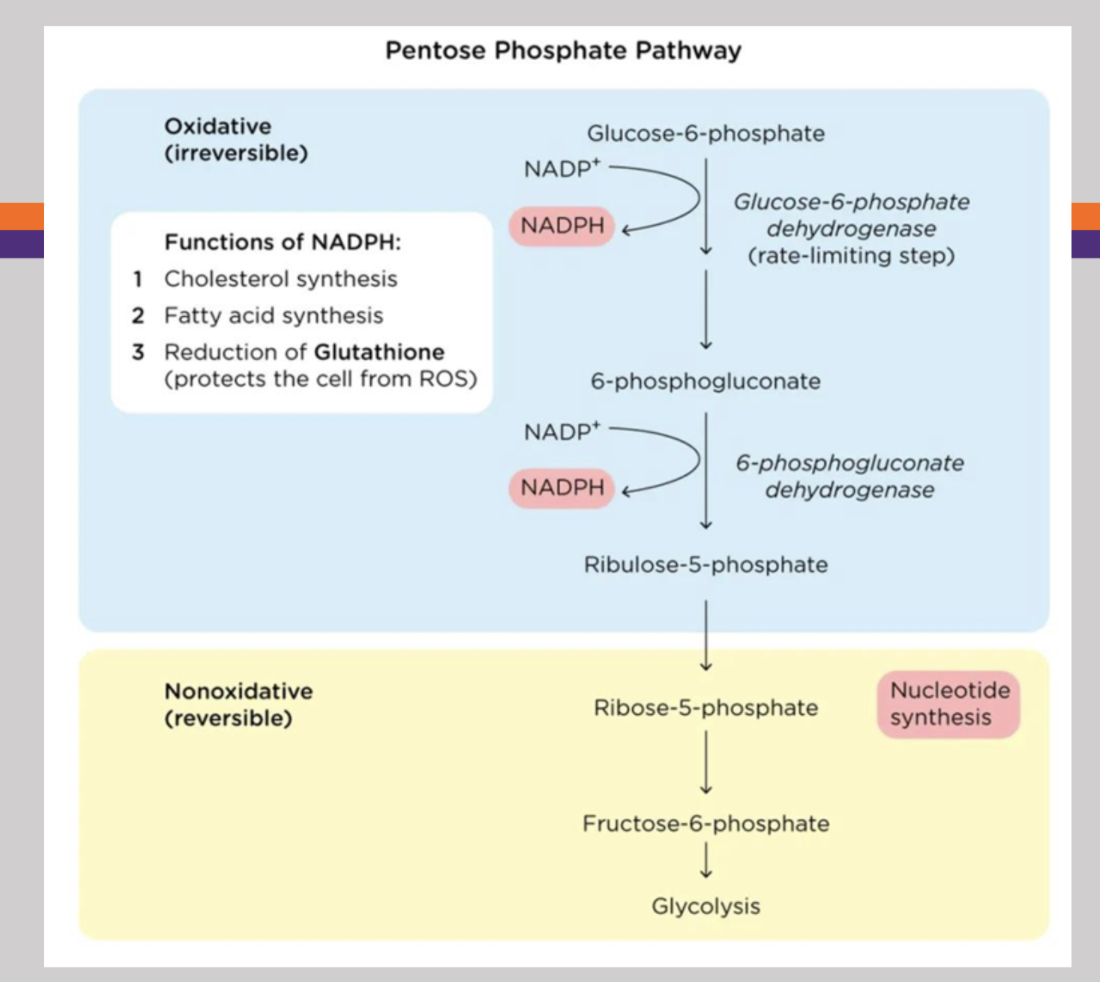
Ribulose-5-phosphate
Product of the pentose phosphate pathway that is utilized in nucleotide synthesis, and can be further converted into fructose-6-phosphate that can also proceed along the glycolysis pathway (reversible pathway)
Citrate Synthase
Catalyzes the first step in the citric acid cycle where water is required for oxaloacetate to receive an acetyl group from acetylCoA and becomes citrate → irreversible
Aconite Isomerase
Catalyzes second step in citric acid cycle that moves OH and H on citrate to make isocitrate
Aconitate
The intermediate product in between citrate and isocitrate in the aconitate isomerase reaction
Isocitrate Dehydrogenase
Catalyzes the third step in the citric acid cycle that generates NADH and CO2 → makes isocitrate into alpha-ketoglutarate
alpha-Ketoglutarate Dehydrogenase
catalyzes the fourth step in the citric acid cycle that yields NADH and CO2 and used coenzyme-A to transform alpha-ketoglutarate into succinyl-CoA
Succinylcholine-CoA Synthetase
Catalyzes the fifth step in the citric acid cycle that breaks off the CoA group, adds a phosphate to it, then gives that phosphate to a histidine residue and releases succinate. That phospho-his then give the phosphate to GDP and generates GTP (energy)
Succinyl Phosphate
The intermediate product formed inbetween succinyl-CoA and succinate by way of succinyl-CoA synthetase
Succinate Dehydrogenase
Embedded in the inner mitochondrial membrane that catalyzes the sixth step of the citric acid cycle. Turns succinate into fumarate → utilizes enz-FAD
Enz-FAD
Becomes enz-FADH2 in the sixth step of the citric acid cycle by oxidizing succinate. Then it re-oxidizes itself and donates its electrons to make QH2 (ubiquinol)
Fumarase
Catalyzes the seventh step of the citric acid cycle and utilizes water to turn fumarate into malate
Malate Dehydrogenase
Catalyzes the eighth step of the citric acid cycle and turns malate back into oxaloacetate to be able to restart the cycle → generates NADH
Intermediate for Glucose
Oxaloacetate → PEP
Intermediate for Pryimidines
Oxaloacetate
Intermediate for Porphyrins and Heme
Succinylcholine-CoA
Intermediate for Purines
alpha-Ketoglutarate
Intermediate for Fatty Acids and Sterols
Citrate
Anaplerotic Reactions
Chemical reactions that replenish intermediates
Tumor Causing
Mutations in succinate dehydrogenase and fumarate
Glial Cell Tumors
Mutant NADPH-dependent isocitrate dehydrogenase that loses nits ability to convert isocitrate to alpha-ketoglutarate and gain ability to convert alpha-ketoglutarate into 2hydroxyglutarate
Hydroxyethyl-TPP
Forms after pyruvate attaches to it and releases a molecule of CO2 in the process
Lipoamide
Receives the acetyl group from TPP
Coenzyme A
Takes the acetyl group off of lipoamide and becomes acetyl-CoA → lipoamide becomes reduced
FAD
Becomes FADH2 and oxidizes lipoamide
NAD+ in Pyruvate Dehydrogenase Complex
Receives electrons from FAD and becomes NADH
Standard Reduction Potential (E’)
Tendency of the oxidized form of a compound to accept electrons → the higher this value, the more susceptible a compound is to getting reduced
Nernst Equation
Entails the actual reduction potential → E = E’ - (0.026/n)ln[Areduced]/[Aoxidized]
Difference in Reduction Potential (deltaE’)
The larger the number, the greater the tendency of electrons to flow from one substance to the other and the greater the change between the system → E’(e- acceptor) - E’(e- donor)
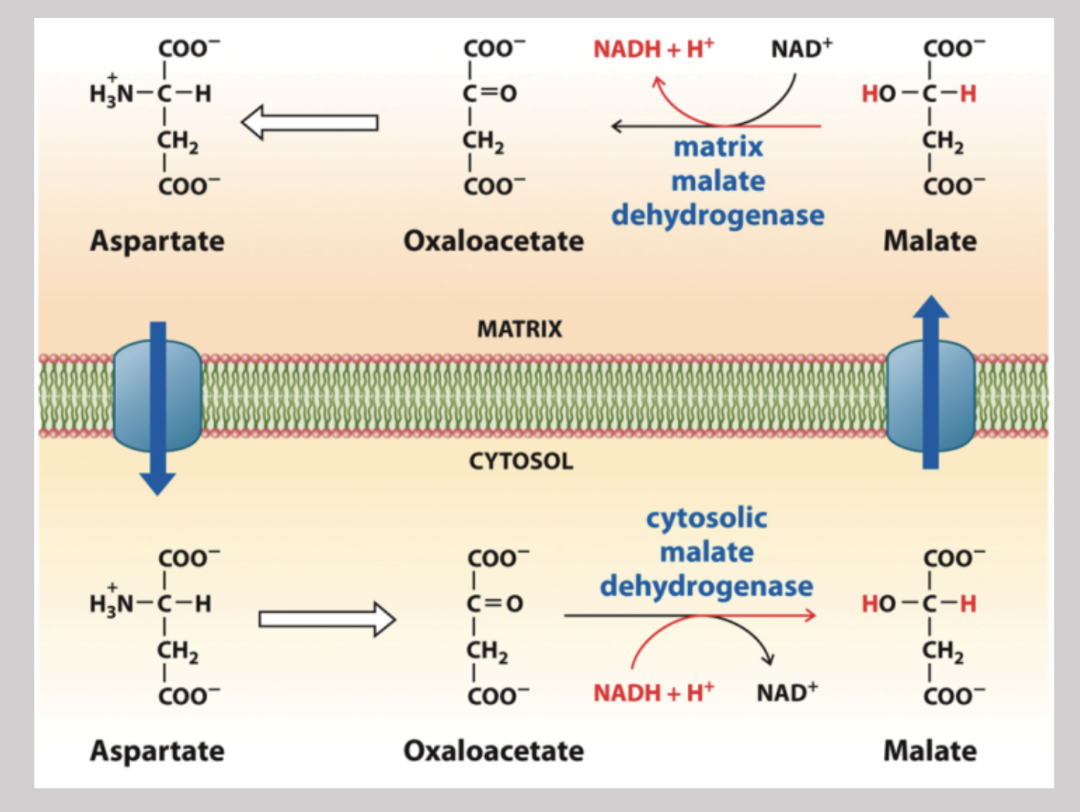
Cytosolic Malate Dehydrogenase
Takes electrons from ocaloacetate and puts them on malate in order to transport it into the mitochondrial matrix
Matrix Malate Dehydrogenase
Regenerates NADH by stripping electrons off of malate and converts it back into oxaloacetate (then back into asparate) so it can re enter the cytosol and start the shuttle system over again
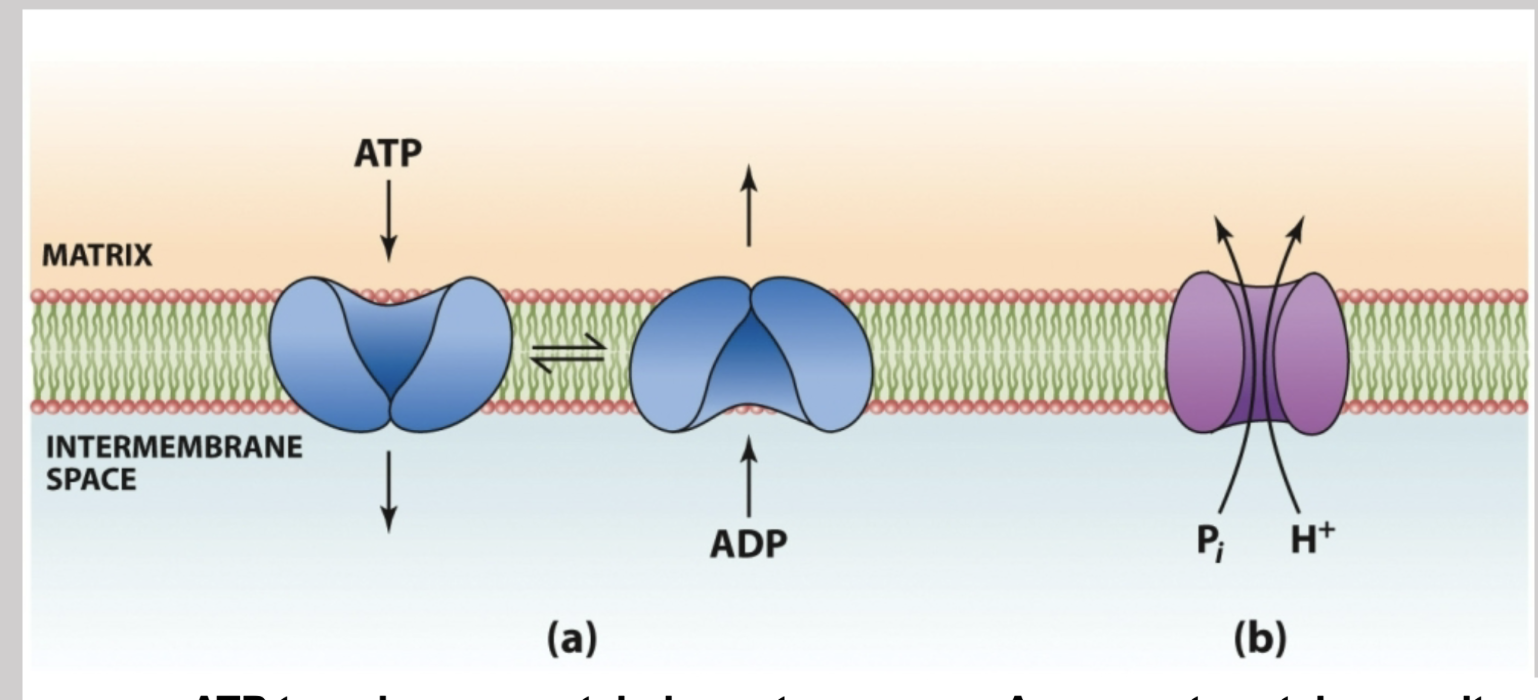
Mitochondrial transport Systems
Moves ADP through its own channel from the inter membrane space into the matrix along with Pi and H+ through another symport protein. In the matrix it is generated into ATP and then sent back down its own channel into the inter membrane space
ETC Complex I
NADH can carry 2 electrons and drops them off to Q at this site → 4H+ get pumped from matrix to inter membrane space
Flavin Mononucleotide
Found in complex I of ETC and accepts electrons from NADH → becomes FNMH2
Iron Sulfur Clusters
Part of complex I in the ETC and can accept one electron at a time from FMNH2
The Q Cycle
QH2 arrives at complex III and gives ONE electron to the ISP and then to cytochrome C while releasing 2H+ into the inter membrane space. The second electron goes to cytochrome B, and then back to Q to make Q-. A second QH2 enters and performs the same process. Total of 4H+ released to create proton gradient
Complex IV
Electrons arrive via 2cytochromeC, they join with oxygen to form water, and in turn 2H+ are pumped into inter membrane space
F0
Bottom component of ATP synthase that rotates protons and pumps them back into the matrix
F1
Upper component of ATP synthase that catalyzes the reaction of ADP +Pi → ATP + H2O
C Subunits
Part of the F0 complex that rotates and contains highly conserved aspartate or glutamate residues that serve as binding sites for protons in the inter membrane space (gamma unit spins with is)
Beta Subunits
Part of F1 complex responsible for that catalytic activity in generating ATP
Open Conformation 1 to Loose Conformation
Binds with ADP + Pi
Loose Conformation to Tight Conformation
Makes ATP
Tight Conformation to Open Conformation 2
Releases ATP
Liver Fed State
Most abundant storage of glycogen. Gluose can also be converted into acetyl-CoA and triacylglycerols
Liver Fasted State
Glycogen and/or amino acids broken down into glucose and triacylglycerols are broken down into acetyl-CoA into Ketone bodies sent out to the blood
Kidney
Rare process, but glutamine is converted to alpha-ketoglutarate to glucose
Adipose Tissue Fed State
Picks up glucose and fatty acids, turns glucose into glycerol and then triacylglycerol
Adipose Tissue Fasted State
Breaks down triacyl glycerols into fatty acids
Muscle Fed State
Converts glucose to glycogen (not main holder of glycogen though)
Muscle Active State
Glycogen converted to glucose, then pyruvate, then alanine or lactate. Or incoming fatty acids and ketone bodies can be converted to acetyl-CoA
Muscle Starved State
Proteins are broken down into amino acids
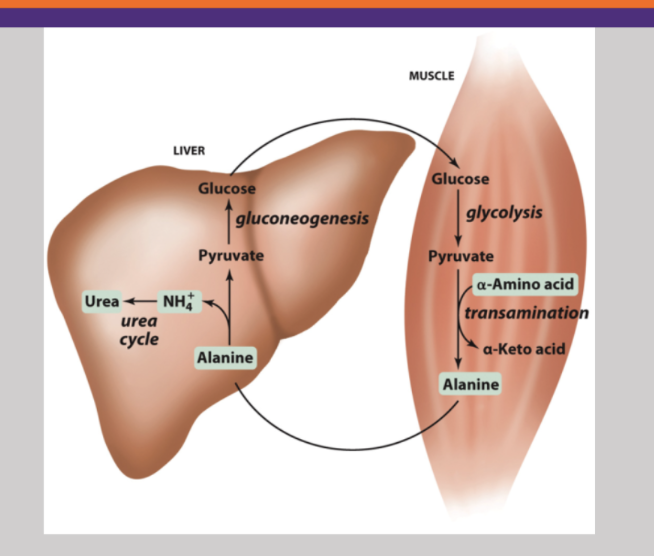
The Glucose-Alanine Cycle
Glucose in muscle cell broken down into pyruvate. Then an amino acid comes in to add on a nitrogen and creates alanine. Alanine is released to the blood and taken up by the liver. Here the nitrogen group is stripped off to undergo the urea cycle, and the re-generated pyruvate gets converted back into glucose to be put back in the blood and repeat the cycle
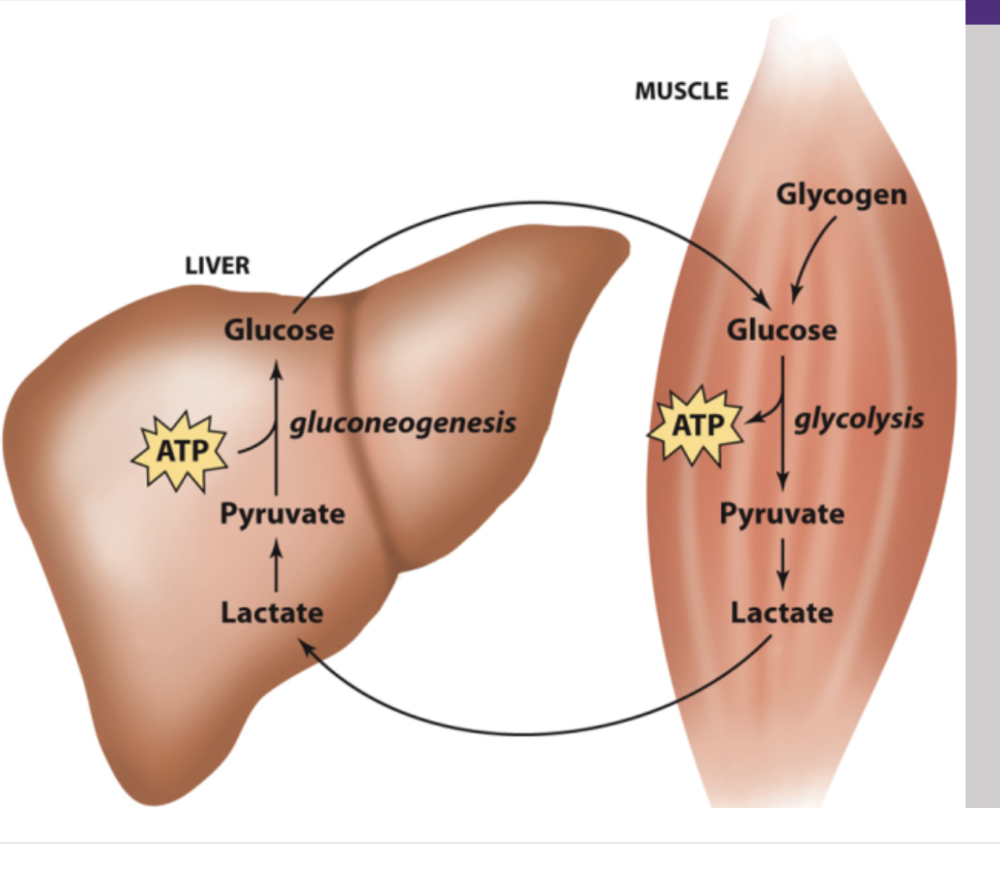
The Cori Cycle
Glycogen in the muscle is broken down into glucose, then pyruvate, and then lactate. The lactate goes into the blood, gets picked up by the liver, re-generates into pyruvate, and then undergoes gluconeogenesis to put glucose back in the blood to repeat the cycle
Pancreatic Islet Cells
Generates glucagon from alpha-cells, and generates insulin from beta-cells
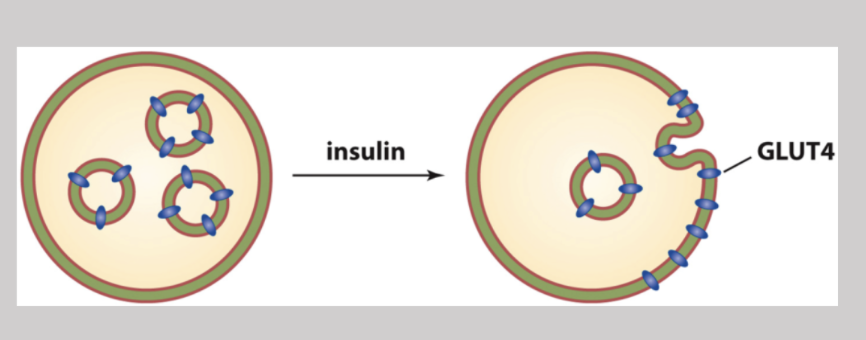
GLUT4
A glucose transporter that gets put on the surface of the cell in the presence of high insulin activity
Glucagon
Signs to STOP making glycogen and release energy by adding phosphates to glycogen synthase to make it less active and to glycogen phosphorylase to make it more active
Insulin
When glucose levels are high, it removes phosphates from glycogen synthase to make it more active, and removes them from glycogen phosphorylase to make it less active
cAMP
A chemical messenger that activates and deactivates a number of proteins. Its presence is increased when glucagon and epinephrine are active to signal a need for releasing energy in a cell (either fight or flight or low glucose response)