Chp 25 - Glycogenesis (Glycogen Synthesis)
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What is the path for Glycogen formation?
Glucose → G6P → G1P → UDP-Glucose → Glycogen(n+1)
What is UDP-glucose? Why is it formed?
UDP-glucose → High energy Activated carrier
Used because glycosidic bonds expensive to make
Is Glycogen building just the reverese of glycogen breakdown?
No. Different processes
Which enzymes are involved with Glycogen building?
Hexokinase/Glucokinase
Phosphoglucomutase → Forms a-1,4 bonds
UDP-glucose phosphorylase
Glycogen synthase
Branching enzyme → Forms a-1,6 bonds (branches)
What are the 5 steps to making Glycogen
G6P formation from Glucose (via Hexokinase)
G!P formation from G6P (via Phosphoglucomutase)
UDP-glucose formation from G1P (via UDP-glucose pyrophosphorylase)
a-1,4 bond formation
a1,6 bond formation for branching
What happens in steps 1 and 2
Glucose —Hexokinase→ G6P
In liver: Glucokinase used
1 ATP SPENT
G6P —Phosphoglucomutase→ G1P
reversible reaction from glycogen breakdown
What happens in step 3? What is the reaction that takes place?
G1P + UTP —UDP-Glucose pyrophosphorylase→ UDP-Glucose + PPi
1 UTP SPENT
Phosphate-uridine group of UTP transferred to G1P
Two remaining phosphatebgroups form PPi (pyrophosphate)
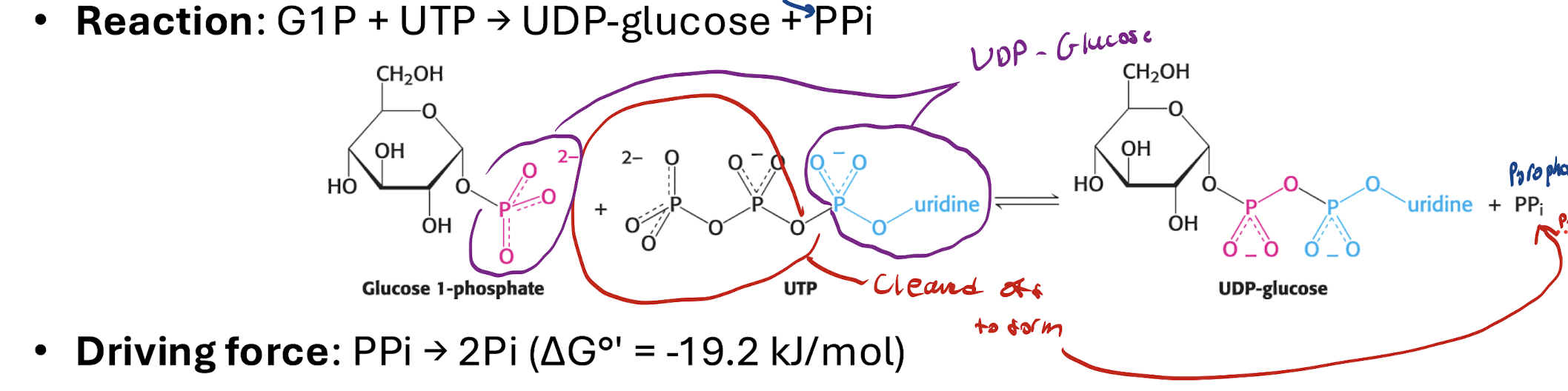
How can Step 3 be reveresed? What enzyme is needed and what has to happen?
PPi present → the Step 3 reaction is reversible
PPi needs to be broken down to make reaction non-reversible
PPi —Pyrophosphatase (via ATP)→ 2Pi
Irreversible reaction (-∆G), strong forward driving energ
PPi breaking down means reaction can’t ne reversed since no PPi to reverse the reaction
What is the energy investment for Step 3?
1 ATP, 1 UTP
= 2 ATP equivalents needed for step 3
What happens in Step 4 of glycogen building? What has to happen first before Glucose could be added via Glycogen Synthase?
Forms a-1,4 glycosidic bonds
UDP-glucose + Glycogen(n) → Glycogen(n+1) + UTP
Glycogenin must prime by adding initial 8-10 glucose from the core
Glucose added via Glycogen Synthase (can’t start new chains or make branching points, so glycogenin has to start the chains and then branching enzyme will do branches)
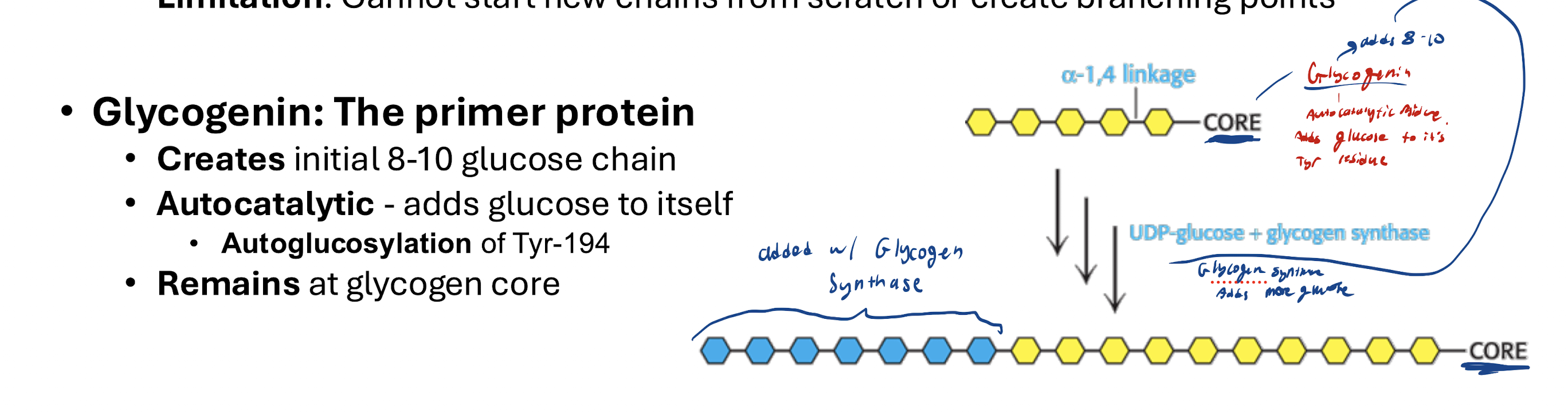
How does Glycogenin add glucose to itself?
Autocatalytic (adds glucose to itself) via Autoglucosylation of Tyr-194. Remains at the glycogen core. (Priming step)
When is Glycogen Synthase most active? How does that compare with glycogen phosphorylase?
Glycogen synthase → Most active when in dephosphorylated (synthase a) b-form activated by G6P
Reciprocal regulation compared with Glycogen Phosphorylase
Most active when phosphorylated
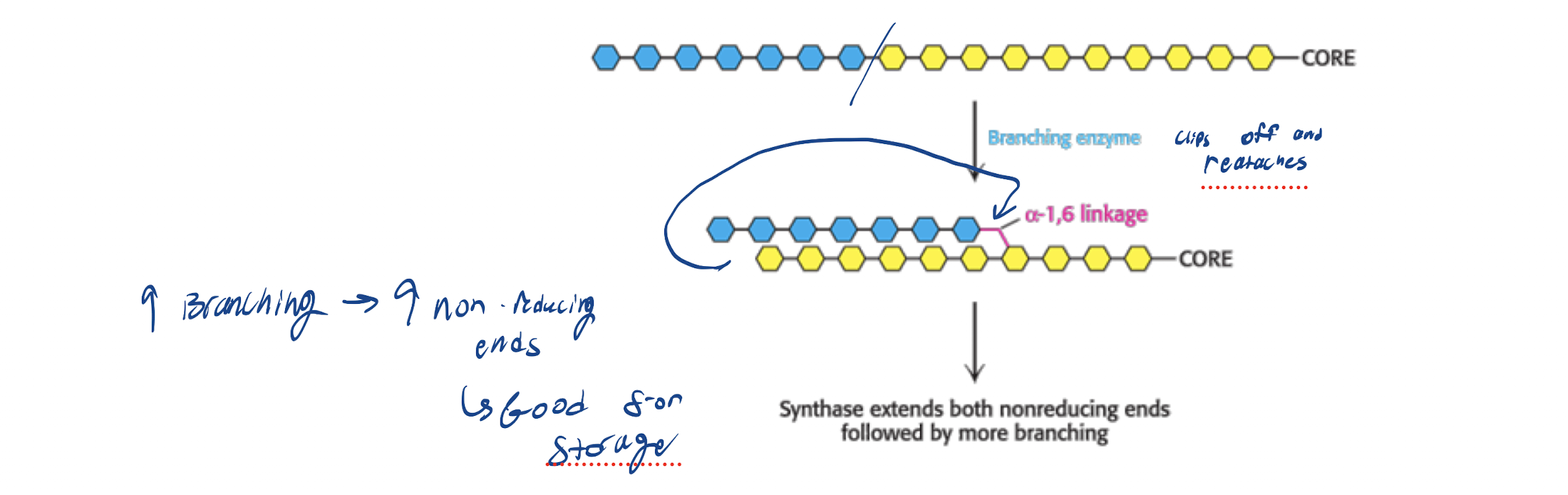
What happens in Step 5? How is it done?
Branching enzyme creates a-1,6 bonds
Takes a 7 glucose segment from a-1,4 chain → Creates a-1,6 bond at a branching location every 12 glucose residues
What is the advantage of branching?
Increased surface area for further synthesis
Inc branching → Inc non-reducing ends → good for storage
What is the cost to store energy? (the net cost). What steps are requiring energy?
G6P formation uses ATP
Not counted toward the Energy cost because this step is necessary regardless when doing metabolism
UDP-glucose activation: 1 UTP → UDP + PPi → 2Pi
Counted because not done during metabolism
True energy cost: 1 UTP = 1 ATP equivalent per glucose being stored
50 glucose being stored → True cost of 50 UTP
Does Glycogenolysis have any energy cost?
0 ATP cost (because uses a phosphorylase enzyme)
30-32 ATP with oxidative phosphorylation produced per Glucose molecule broken off
Since it costs 1 ATP to store a glucose as glycogen and doesn’t cost any to breakdown and mobilize glucpse, what does that mean? What does this allow the body to do?
Body is able to return 30-32 ATP when breaking off one glucose only sepnding 1 ATP → 3000% return on investment
Allows body to survive in Fasted States and performance when exercising
With the alternative Gluconeogenesis (with it costing 6 ATP per glucose) why is that even done if glycogen is much more efficient to use?
Body could run out of glycogen stores, or the Glucose is needed for something else
How would Glycogenolysis and Glyconeogenesis running at the same time be a futule cycle? How does the cell make sure this doesnt happen
Glycogen would be broken down and stored at the same time
Every storage cycle would spend 1 ATP equivalent every time glucose is stored (wasting)
Solution - Reciprocal Regulation (something activating one will inhibit the other)
Synthesis on → Breakdown off
Breakdown on → Synthesis off
What path is active in the fasted state (high glucagon and epi)?
Glycogenolysis (Glycogen breakdown) is active
How does glucagon and epi being present affect glycogenolysis via the adenylyl cyclase path? What is the pathway
PKA activated via Glucagon/Epi → Glycogen Synthase (active when dephosphorylated) AND Glycogen Phosphorylase (active when phosphorylated) → Glycogen Syntahse switches ot it’s b form (inactive form) and Glycogen Phosphorylase switches to it’s a form (active form)
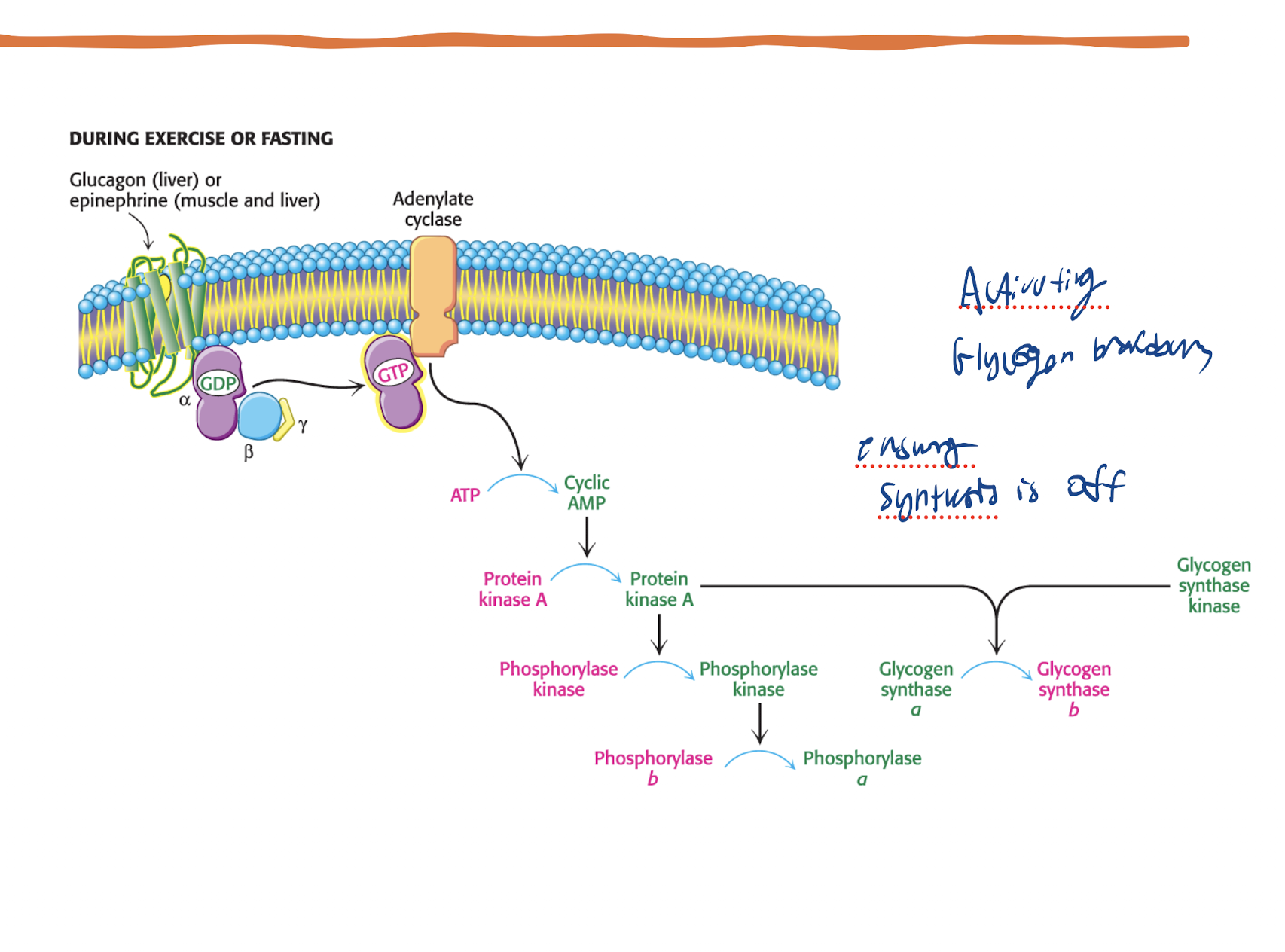
What does Glycogen synthase kinase do in the PKA path gor Glycogenolysis?
Another enzyme keeping Glycogen synthase in it’s inactive b form (phosphorylated in this form)
What path is active when in the FED state (insulin active)
Fed State → Glycogenesis active
makes sense beacuse body just got glucose and can now convert it to glycogen
How does INSULIN being present affect glycogenesis via the adenylyl cyclase path? What is the pathway.
No Glucagon signaling → PKA NOT activated
No activation of phosphorylase kinase which would have kept synthase in inactive form if activated
Insuling → Activates Akt (Activating Protein Kinase B) → Phosphorylates Glycogen synthase kinase (turns it off, which stops the inhibition signal of glucogen synthase kinase to glycogen synthase)
So, Insulin signaling DOES NOT activate PKA and PHOSPHORYLATES Glycogen Synthase Kinase (deactivating it) so neither of them can inhibit Glycogen synthase
Thus inhibition of inhibition signal, keeping glycogen synthase active.
What other enzyme is needed to activate glycogen synthase from it’s inactive form (Glycogen synthase b, phosphorylated) into it’s active form (Glycogen Synthase a, dephosphorylated)
Protein Phosphatase 1 (PP1) →
DEphosphorylates glycogen phosphorylase a (inactivating it)
DEphosphorylates Phosphorylase Kinase (inactivating it)
DEphosphorylates Glycogen Synthase b (activating it to glycogen synthase a)
→ Reciprocal Regulation
What determines the default state of Glycogen Phosphorylase in tissues
PP1 activity (whether it is stimulated or inhibited)
What is the structure of PP1 in the muscle?
Catalytic Subunit—Glycogen-binding subunit (Gm)
Gm subunit and all it’s enzymes connected directly to PP1 (so PP1 is highlyy active on the enzymes in the Gm)
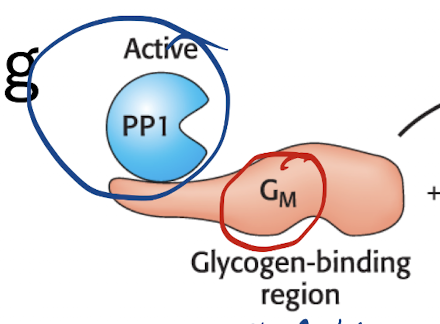
What is the state PP1 is always in the muscle? What does that mean?
Always active
Glycogen phosphorylase in it’s inactive dephosphorylated b form
glycogen synthase is in it’s “a” dephosphorylated active form
means glycogen is being made by default in the muscle until signaling reaches
How is PP1 affected by Epi signaling? What’s the result? in muscle
Epi activates PKA → Gm subunit phosphorylated (and the enzymes) → PP1 dissociates from Gm → Another phosphorylation of PP1 binds it to a phosphorylated inhibitor, deactivating PP1
Result:
Glycogen Synthase Kinase→ Phosphorylated → b (inactive form)
Glycogen Synthase Kinase then phosphorylates Glycogen Synthase (a, active, dephosphorylated form) → Phosphorylated Glycogen Synthase, b form, inactivated → No Glycogen formation.
Glycogen Phosphorylase Kinase → Phosphorylated
Glycogen Phosphorylase Kinase then phosphorylates Glycogen phosphorylase (b, inactive, dephosphorylated form) → Phosphorylated Glycogen phosphorylase, a form, activated → Glycogen breakdown
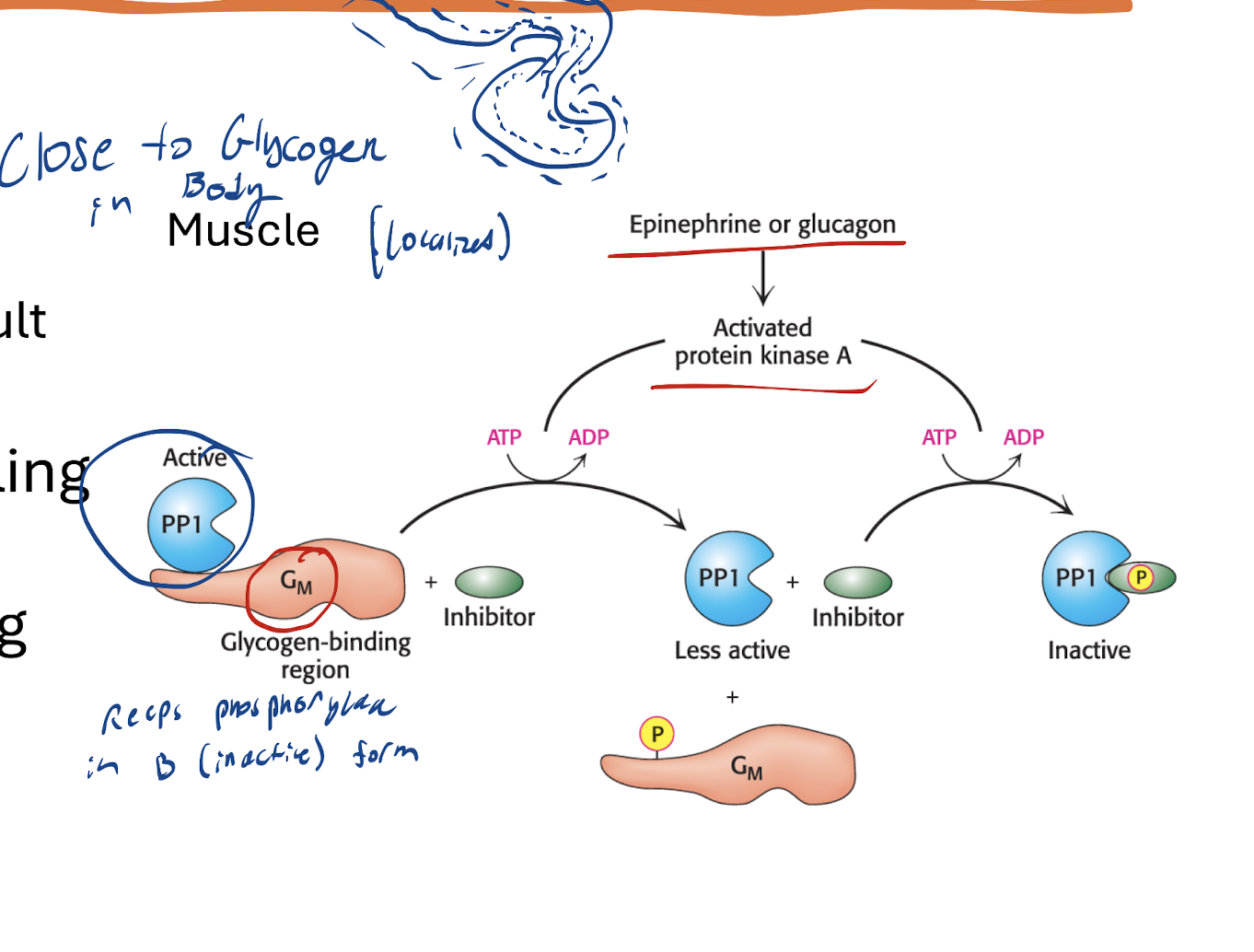
How does regulation of PP1 of the muscle make sense?
Makes sense because muscle received Epi signal. Needs more energy for contraction
Needs to stop making glycogen and break it down instead for energy production
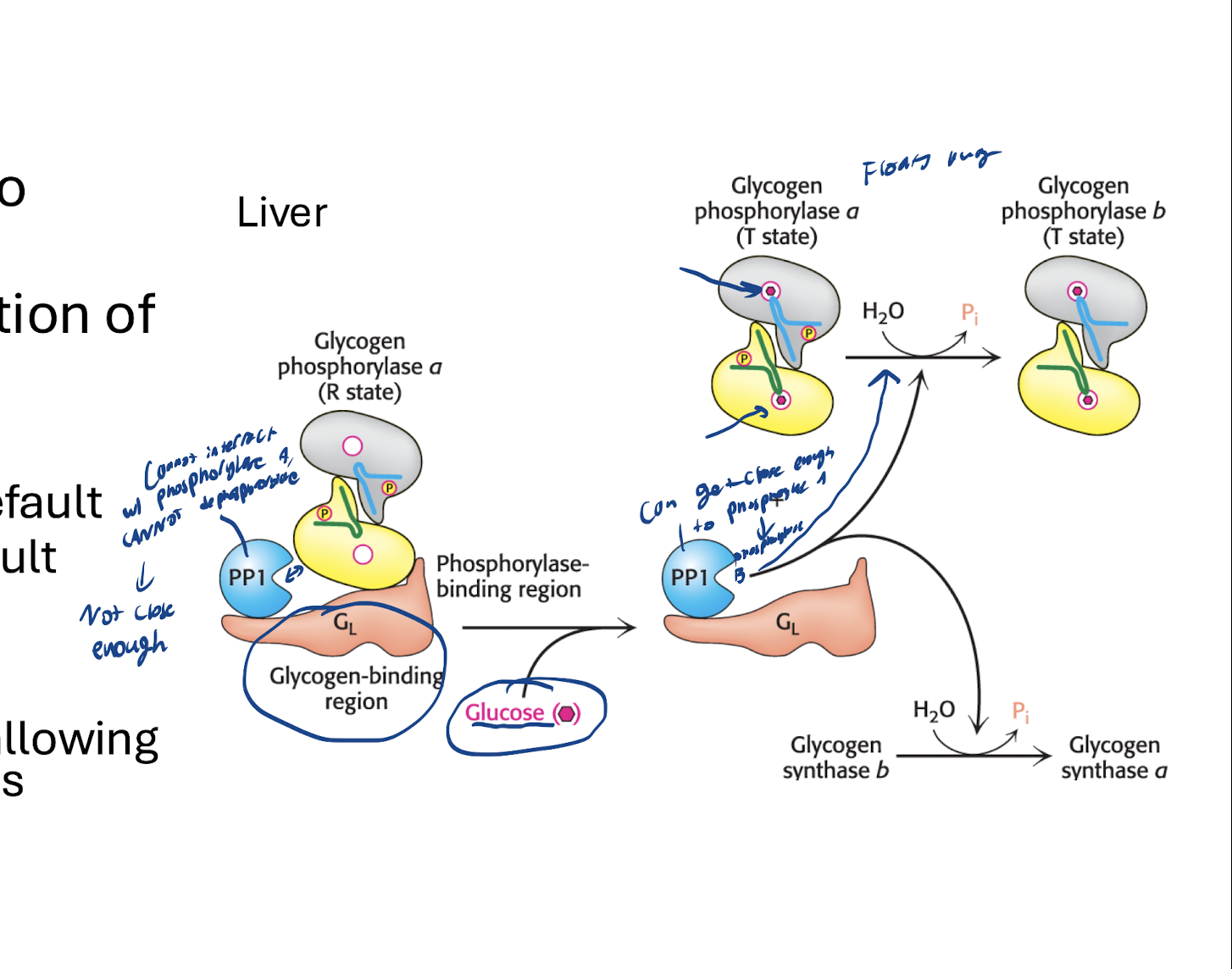
What is the structure of GL in the liver (glycogen-binding subunit)? What does the structuer of it mean?
PP1-GL-Glycogen Phosphorylase a (R state, active)
PP1 is inhibiting enzymes of GL, BUT is not close enough to Glycogen Phosphorylase a to inhibit that
— — → Means that Glycogen Phosphorylase is in a (active form) by deafult.
Glycogen Synthase in b (inactive form) by default
How does glycogen phosphorylase being in it’s active form by default and glycogen synthase being in it’s active form by default make sense for the liver?
Liver wants to break down it’s glycogen inherintly so it can share it with the rest of the body
What is the mechanism to switching to glycogen synthesis in the liver upon glucose entry
Glucose enters and binds to glycogen phosphorylase a’s binding region → Glycogen phosphorylase. dissociates and floats away → PP1 can now react with glycogen phosphorylase a activating it to glycogen phosphorylase b (inactive, T state) → Glycogen synthase is dephosphorylated by PP1, switching to active “a form”
What happens when normal reguation of glycogen storage diseases fails? Two types?
Glycogen storage diseases
von Gierke Disease (GSD I) - Liver Dysfunction
McArdle Disease (GSD V) - Muscle Dysfunction
What is wrong in GSD I, what are the symptoms and how does that happen?
Glucose-6-phosphatase defficiency → Liver can’y realease free glucose into blood (bc has to be converted G6P→Glucose)
Liver becomes enlarged, fasting hypoglycemia (can’t release glycose)
What is wrong in GSD V, what are the symptoms and how does that happen? What is something interesting that’s noted with it
Muscle Phosphorylase defficiency → Can’t breakdown a-1,4 bonds, so myscle can’t breakdown it’s ow glycogen → Exercise intolerance → no glycogen can be broken down for energy so body has to rely on BGL and other energy sources
HOWEVER experience “second wind” when pushing past failure b/c body switches to fatty acid oxidation
Wjat’s the main way Glycogen metabolism is connected to glycolysis and gluconeogenesis?
Glycolysis - G6P from breakdown enters glycolysis path for fast ATP
Gluconeogenesis → Glucose provided when glycogen sotres depleted. Helps maintain BGL
What’s the main way glycogen metabolism is connected w/ Hormonal signaling and tissue specialization. Central theme?
Hormonal signaling → Insulin/glucagon direct different glycogen pathways
Epi links glycogen breakdown with fight/gflight
Tissue specialization: Liver still serves as hub for glucose homeostasis, muscle uses glycogen to meet energy demands
Glucose homeostasis and energy mobilization in coordinated metabolic network is glycogen’s central function