Polyesters and Polyamides
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Condensation polymers
Any kind of polymer formed through monomers joining, and a small molecule is formed as a by-product, such as water or HCl
They are formed with functional groups at both ends of the molecule
Reactions
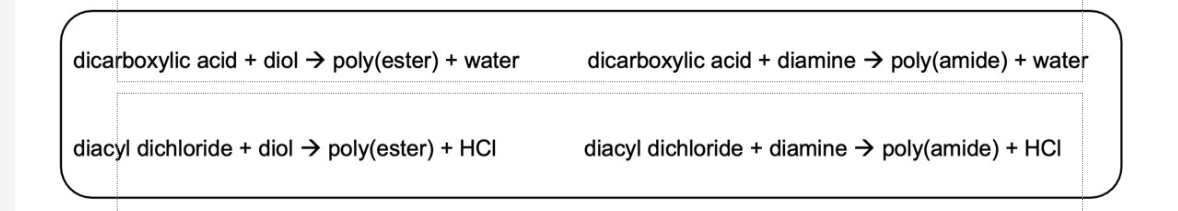
Condensation Polymers: Diacid + Diol
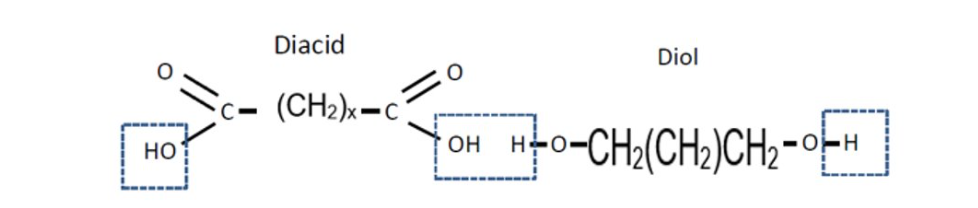
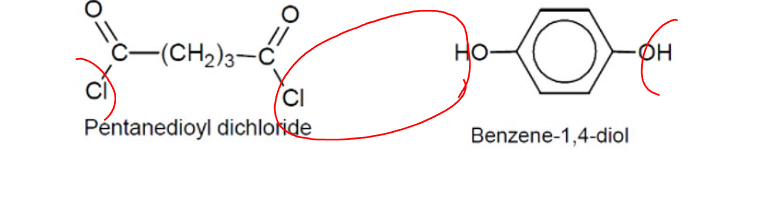
Condensation Polymers: Diacyl chloride and diol
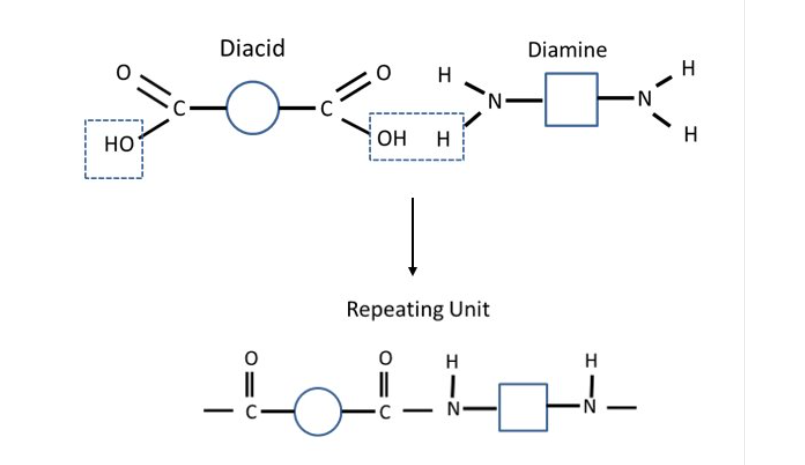
Condensation Polyamides: Dicarboxylic acid and diamine
Properties of condensation polymers
Polyesters: Used as wool, cotton substitutes, carpet and rugs. Bullet proof vests and some flame-retardant clothing are made from polyesters
Polyamides: Elastic and used in underwear, fishing nets and synthetic fibres
Why are polyesters and polyamides biodegradable?
It contains an ester bond which can be hydrolysed
Reason: It contains a polar C-O bond so the delta positive C can be attacked by the nucleophile
Hydrolysis of Polyesters: Acid and Base hydrolysis
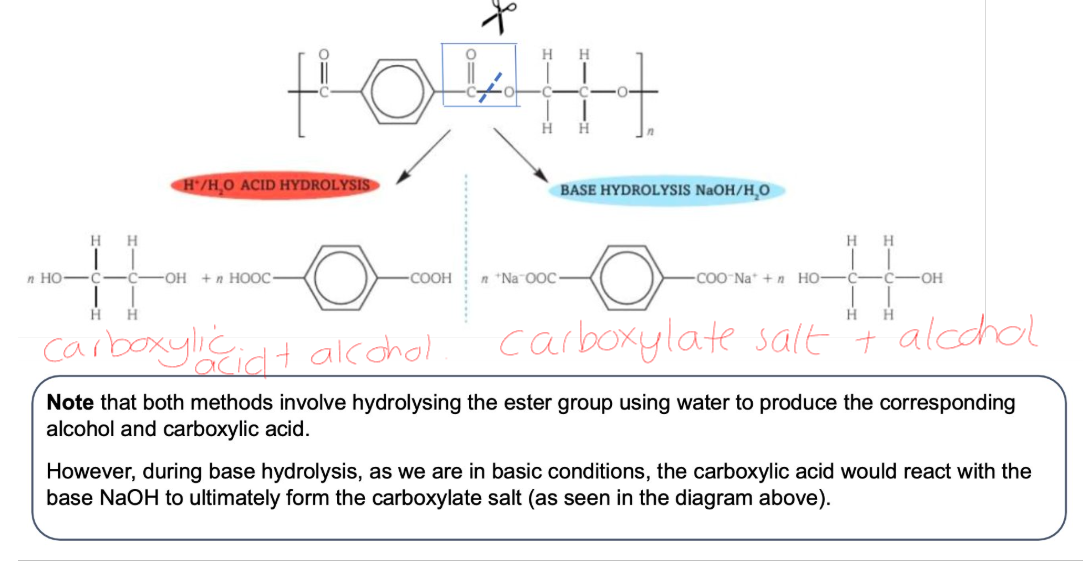
Hydrolysis of Polyamides
Similar to the hydrolysis of polyesters except the CONH group is hydrolysed
Calculations involving polymers: 2 different monomers
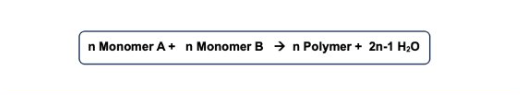
Calculations involving polymers: Bifunctional monomer

Advantages of recycling polyalkenes
Recycling saves using crude oil to make plastic
It reduces landfill
Disadvantages of recycling polyalkenes
Plastics need to be collected, transported and sorted which uses energy and power and is therefore expensive