Chemistry 116 - Chapter 3: Physical Properties of Solutions Part 2
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
What are colligative properties?
Properties that depend only on the number of solute particles, not their identity.
What happens to vapor pressure when a nonvolatile solute is added?
Vapor pressure decreases because fewer solvent molecules escape into the vapor phase.
State Raoult’s Law for a nonvolatile solute.
X_solvent = mole fraction of solvent
P° = vapor pressure of pure solvent

What is an ideal solution?
A solution that obeys Raoult’s law at all concentrations.
How do you calculate total vapor pressure when both components are volatile?
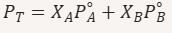
What does “miscible” mean?
Two liquids that are completely soluble in each other in all proportions.
Why does adding a solute raise the boiling point?
Solute lowers vapor pressure → requires higher temperature to reach 1 atm.
Formula for boiling-point elevation.
ΔT_b = increase in boiling point
K_b = boiling‑point elevation constant
m = molality
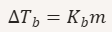
What does Kb represent
Molal boiling-point elevation constant (°C/m), unique to each solvent.
Boiling point of a 1.00 m aqueous nonelectrolyte solution?
Δ𝑇𝑏 = 0.513°𝐶 → new boiling point = 100.5°C.
Why does adding solute lower freezing point?
Solute disrupts solid formation → requires lower temperature to freeze
Formula for freezing-point depression.
ΔT_f = decrease in freezing point
K_f = freezing‑point depression constant
m = molality
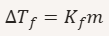
What does 𝐾𝑓 represent?
Molal freezing-point depression constant (°C/m).
Relationship between freezing point and solute particle count?
More particles → greater freezing-point depression.
Why do electrolytes show larger colligative effects than nonelectrolytes?
They dissociate into multiple ions, increasing particle count.
Why doesn’t 1 M NaCl double the freezing-point depression compared to 1 M methanol?
Ion pairing reduces the number of free particles.
Define the van’t Hoff factor (i).
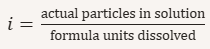
Expected i values (ideal):
NaCl → 2
CaCl₂ → 3
K₂SO₄ → 3
Sucrose → 1
Why does i decrease at higher concentrations?
More ion pairing occurs.
Colligative property formulas for electrolytes.
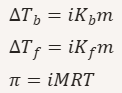
Order the freezing points of:
0.050 m CaCl₂, 0.15 m NaCl, 0.10 m HCl, 0.050 m HC₂H₃O₂, 0.10 m C₁₂H₂₂O₁₁
Lowest → Highest:
0.15 m NaCl → 0.10 m HCl → 0.050 m CaCl₂ → 0.10 m sugar → 0.050 m acetic acid
What is osmosis?
Movement of solvent from low solute concentration → high solute concentration through a semipermeable membrane.
Define osmotic pressure (π).
Pressure needed to stop osmosis.
Osmotic pressure formula.
𝜋 = 𝑀𝑅𝑇
π = osmotic pressure
M = molarity
R = gas constant
T = temperature (K)
What does isotonic mean?
Two solutions with equal osmotic pressure.
Effect of electrolytes on osmotic pressure.
They increase π because they produce more particles (use iMRT).
What does Henry’s law describe?
he relationship between the solubility of a gas in a liquid and the partial pressure of that gas above the solution.
State Henry’s law in equation form.
k_H = Henry’s law constant
P = partial pressure of gas
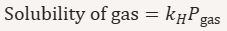
How does increasing gas pressure affect gas solubility (at constant temperature)?
Higher pressure → higher solubility (directly proportional).
Units commonly used for Henry’s law constant 𝑘𝐻?
Typically mol/(L·atm).
In the soda example, what happens to CO₂ solubility after the bottle is opened?
𝑃CO₂ drops from several atm to about 0.0004 atm, so CO₂ solubility decreases sharply and the drink goes flat.