inorganic 2 final
1/129
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
130 Terms
Atomic Orbitals
Solution to SWE
n
principal quantum number
radial part of the wave function
l
orbital quantum number (angular momentum quantum number)
0, 1, 2... (n-1) => s, p, d, f…
determines the shape of the atomic orbitals.
ml
magnetic quantum number
+l -> -l
determines directionality of atomic orbitals => px, py, pz
Degenerate Orbitals
possess the same energy
ms
magnetic spin quantum number
+½ or -½
therefore, there are only 2 electrons per orbital
Effective Nuclear Charge (Zeff)
arises from incomplete shielding of the nuclear charge by the atomic electrons
Slater’s Rule
method of estimating Zeff experienced by electrons in different atomic orbitals
Zeff = Z – S
Z = nuclear charge
S = screening (or shielding) constant
Ionization Energy
energy required to remove a valence electron from an atom in
the gas phase
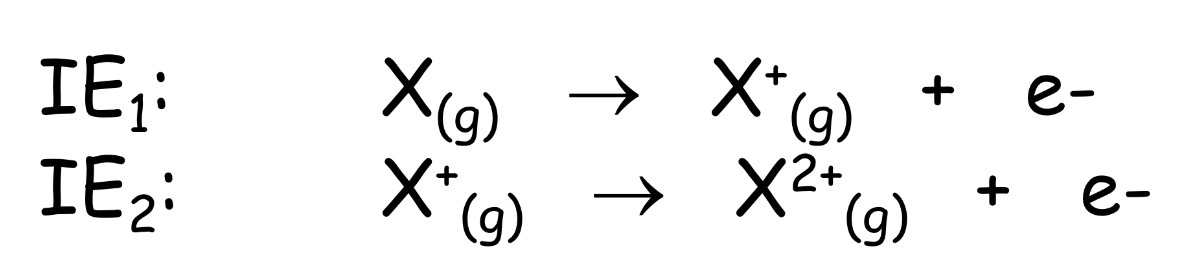
Electron Infinity
minus the internal energy for the gain of an electron by an atom in
the gas phase
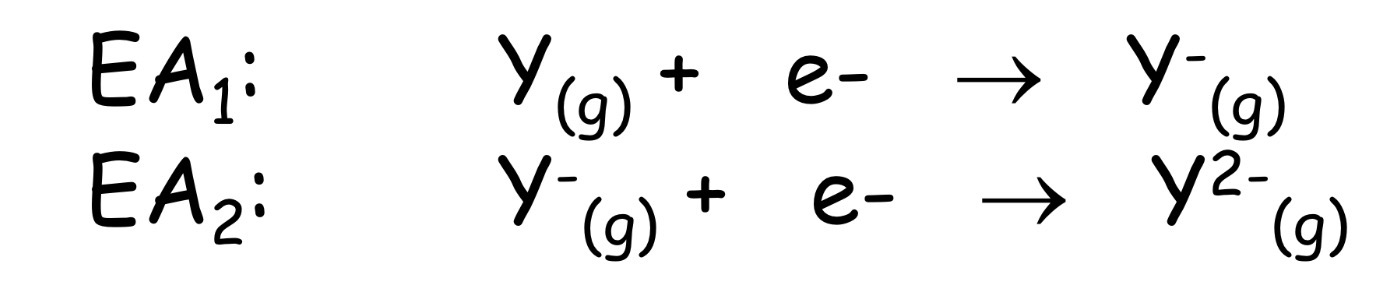
Electronegativity
the electron withdrawing power of an atom in a molecule (x)
Covalent Radius
half of the internuclear separation in a homonuclear X-X single bond
Van der Waals Radius
half of the distance of closest approach of two non-bonded atoms of X
Soft Acids
prefer to bond to soft bases
Hard Acids
prefer to bond to hard bases
Lewis Structures
show patterns of bonding in molecules.
dots represent electrons.
lines represent shared electron pairs
Lone Pairs
unshared pairs of valence electrons on atoms (influence shape and chemical
reactivity)
Resonance Structures
occurs when there are two or more structures with the same
possibility
VSEPR (Valence Shell Electron Pair Repulsion)
model for predicting molecular shape in molecules with central p-block atoms
Linear

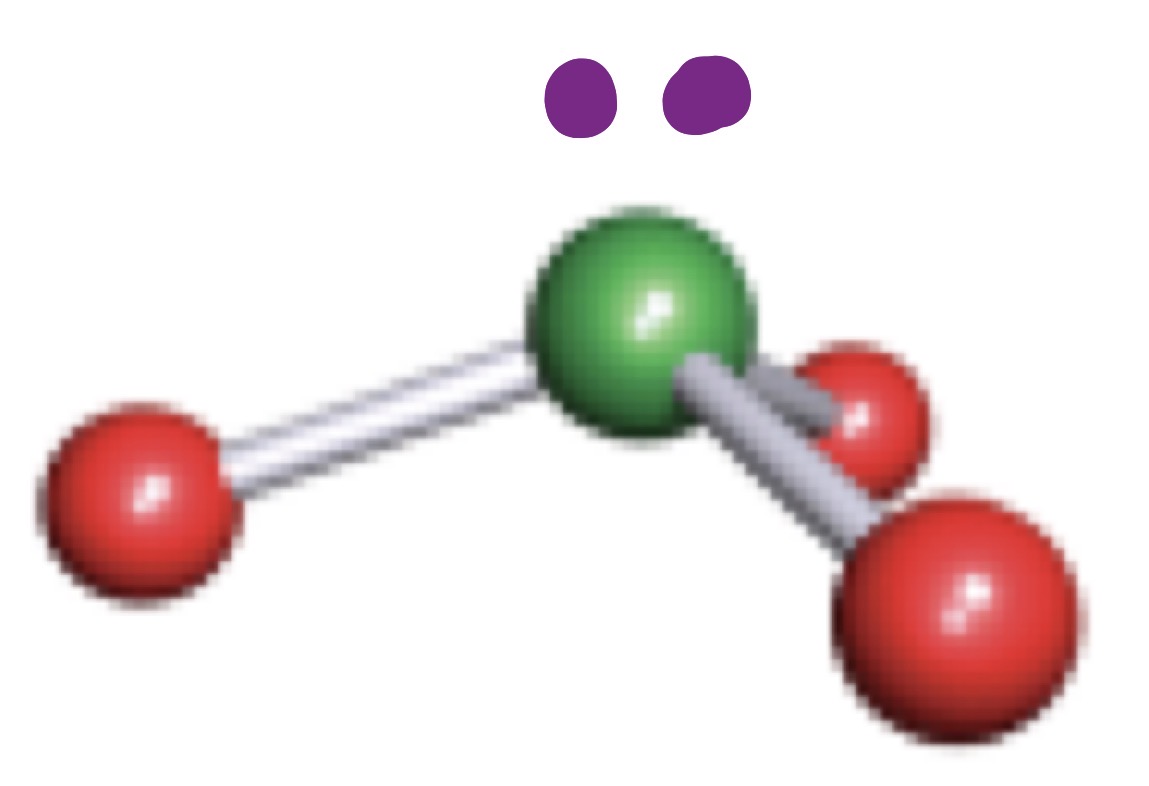
Bent
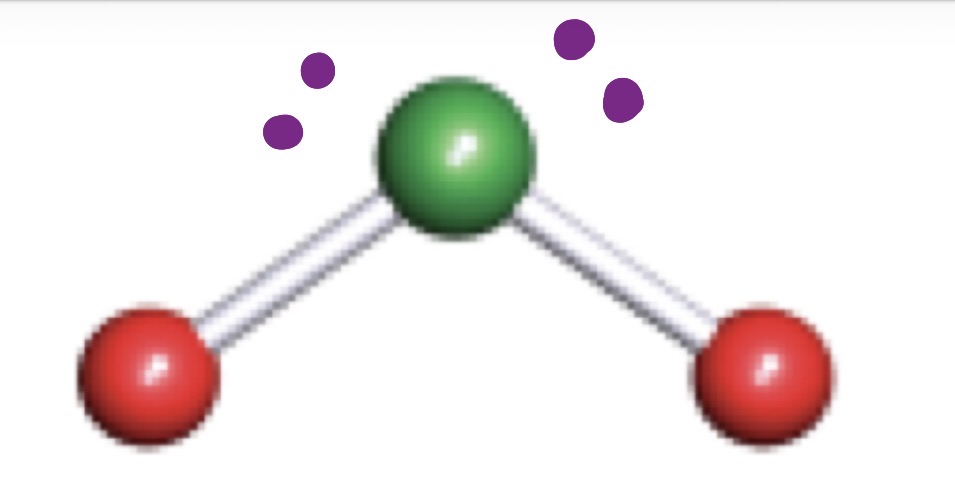
Trigonal Planar
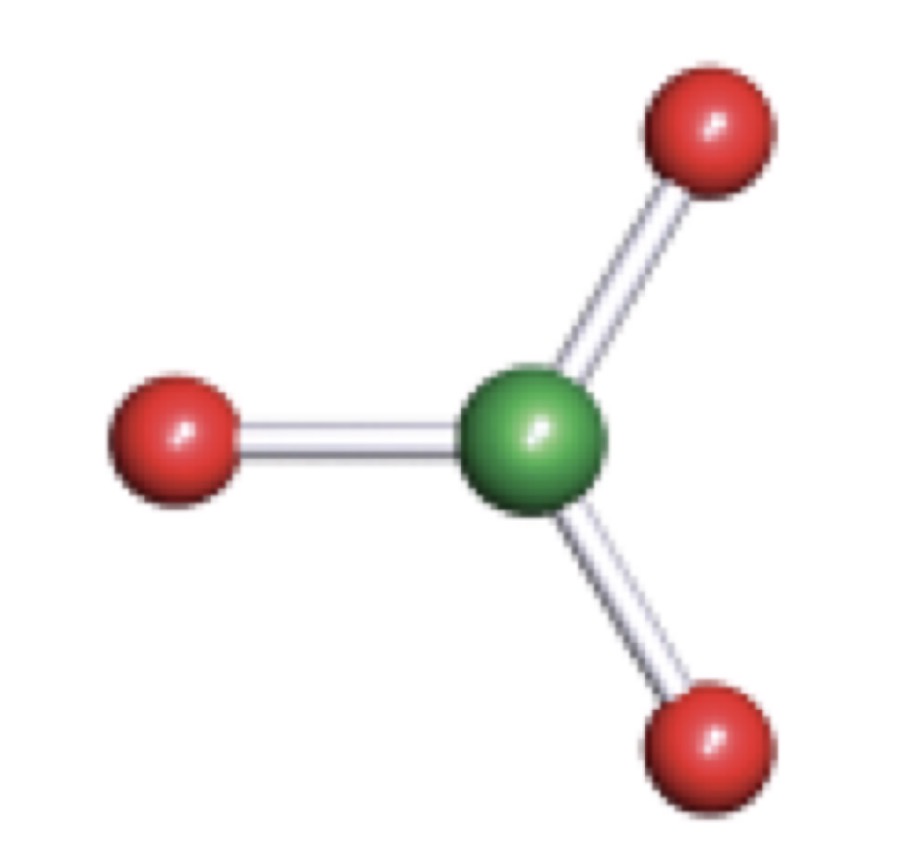
T-Shaped
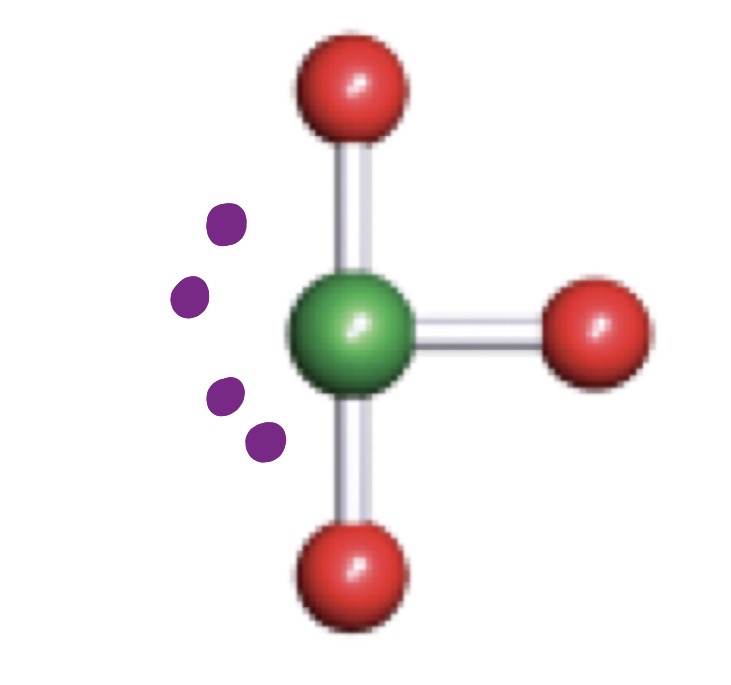
Trigonal Pyramidal
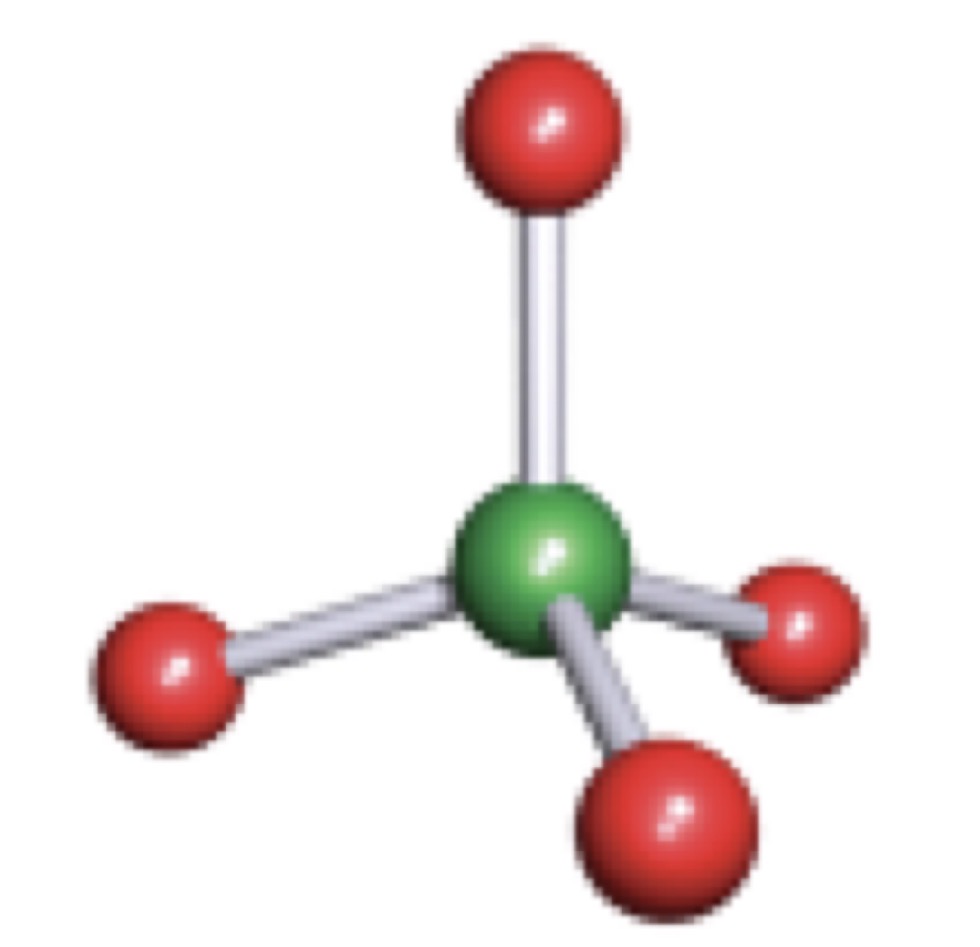
Tetrahedral
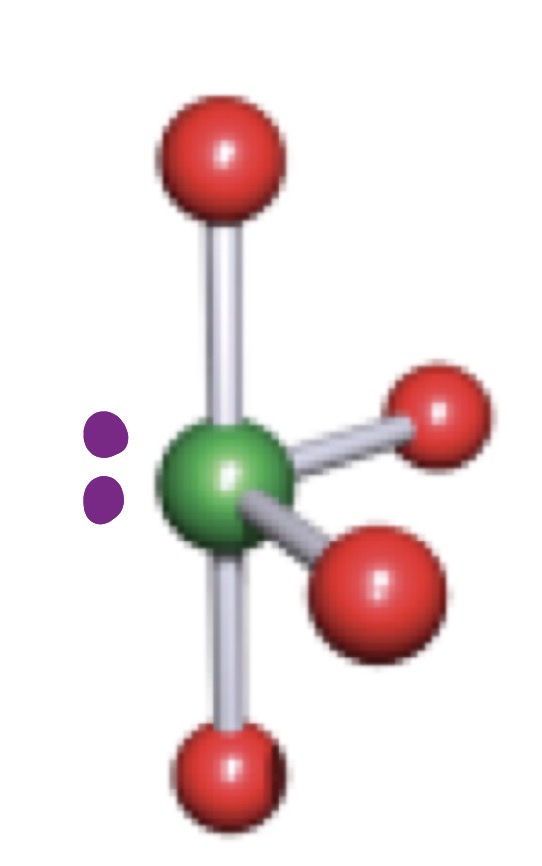
See-Saw
Square Planar
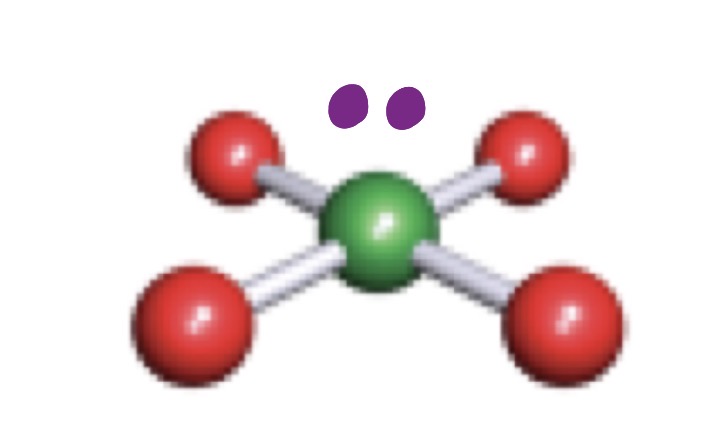
Trigonal Bypyramidal
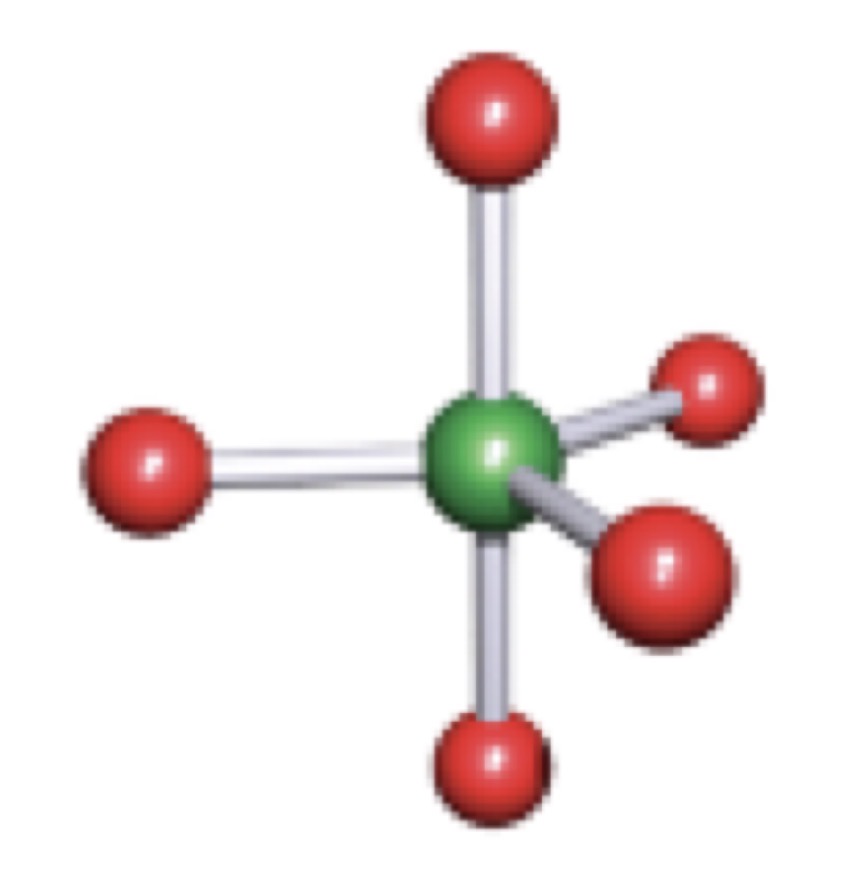
Square-Based Pyramidal
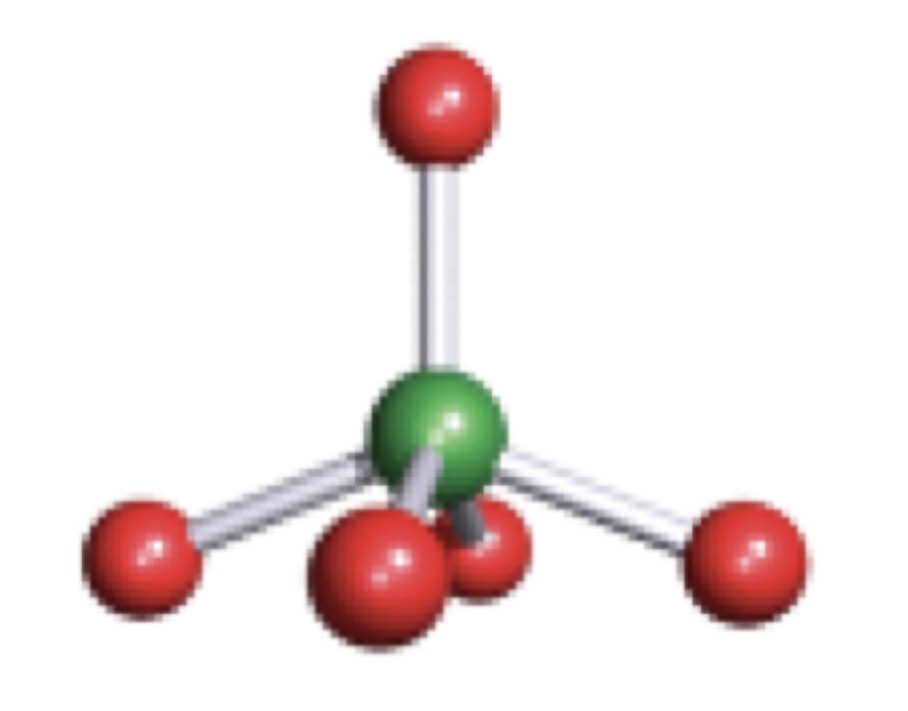
Pentagonal Planar
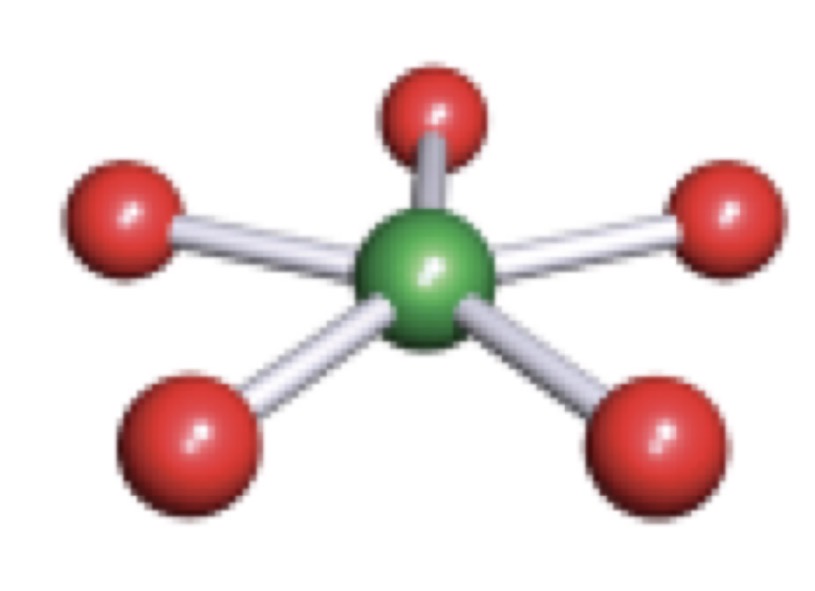
Octahedral
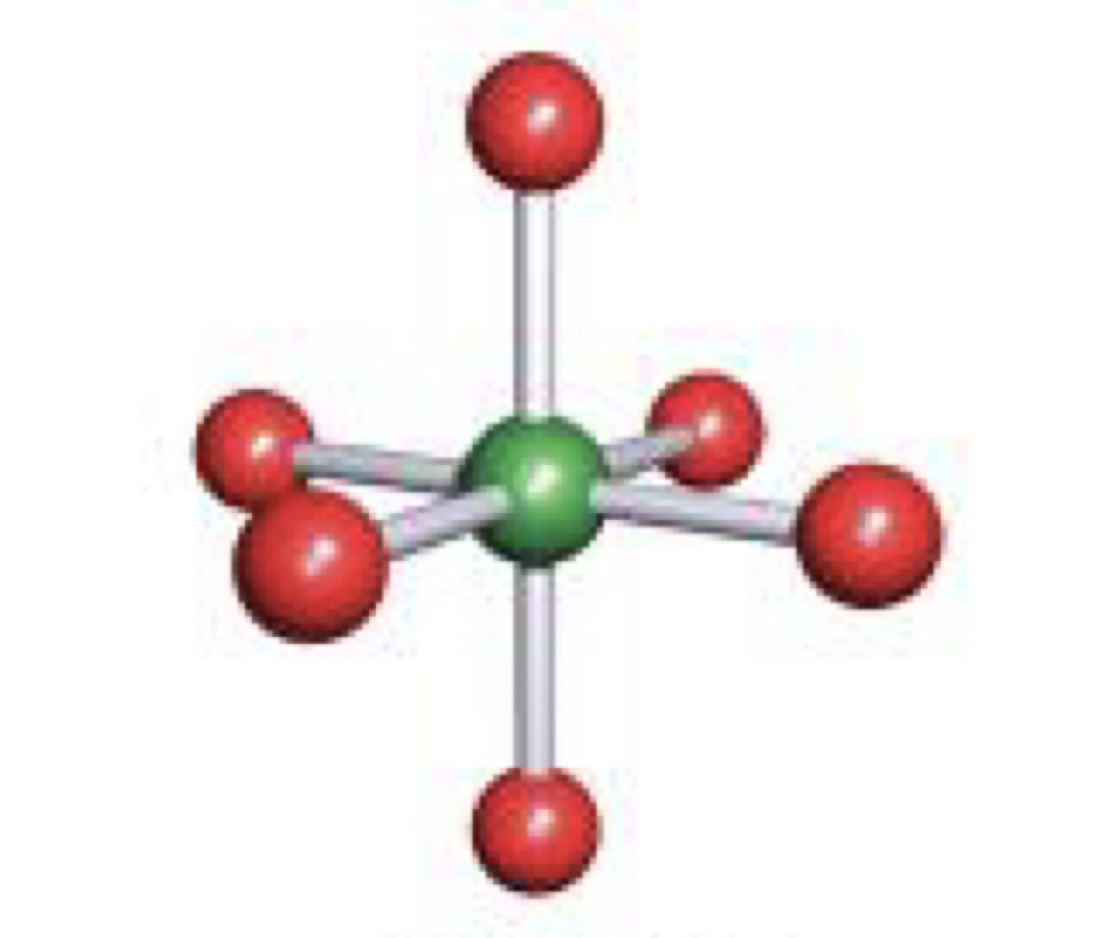
Pentagonal Bipyramidal
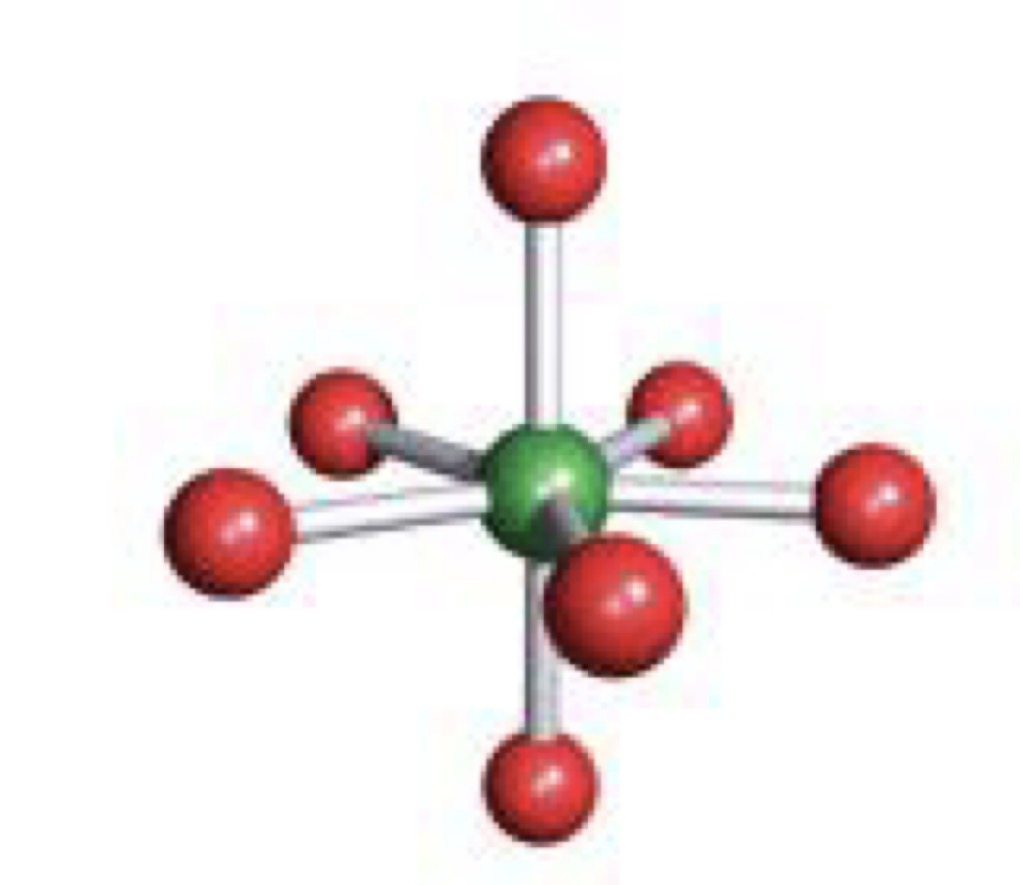
Square Antiprismatic
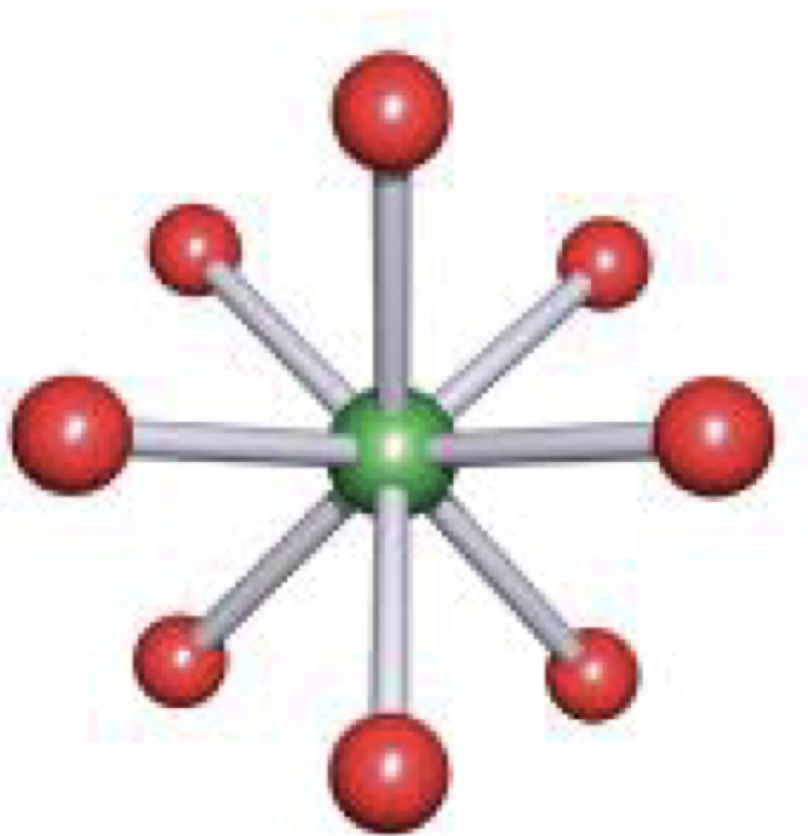
Cis Isomer
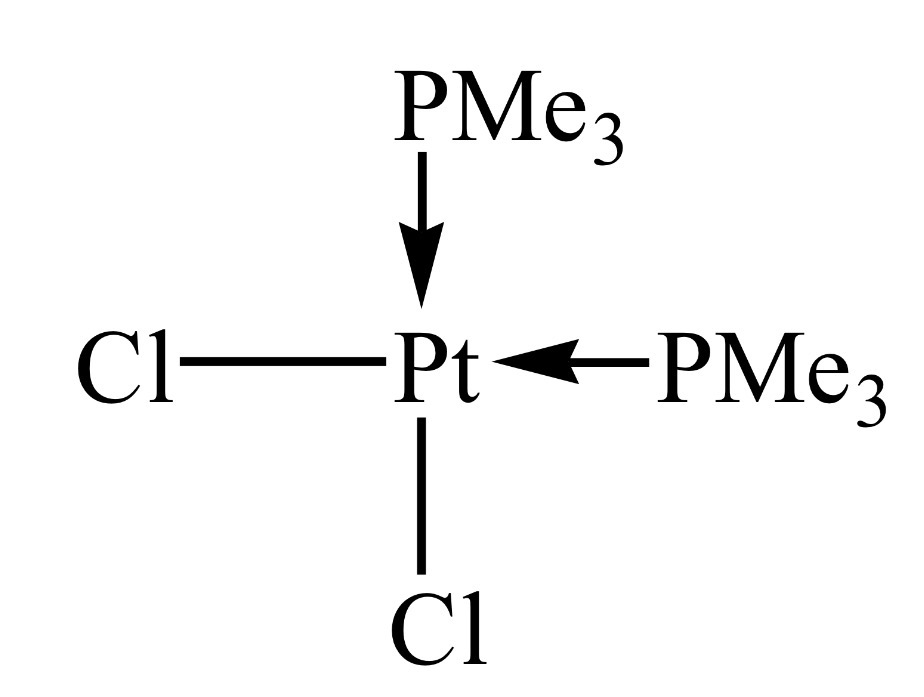
Trans Isomer
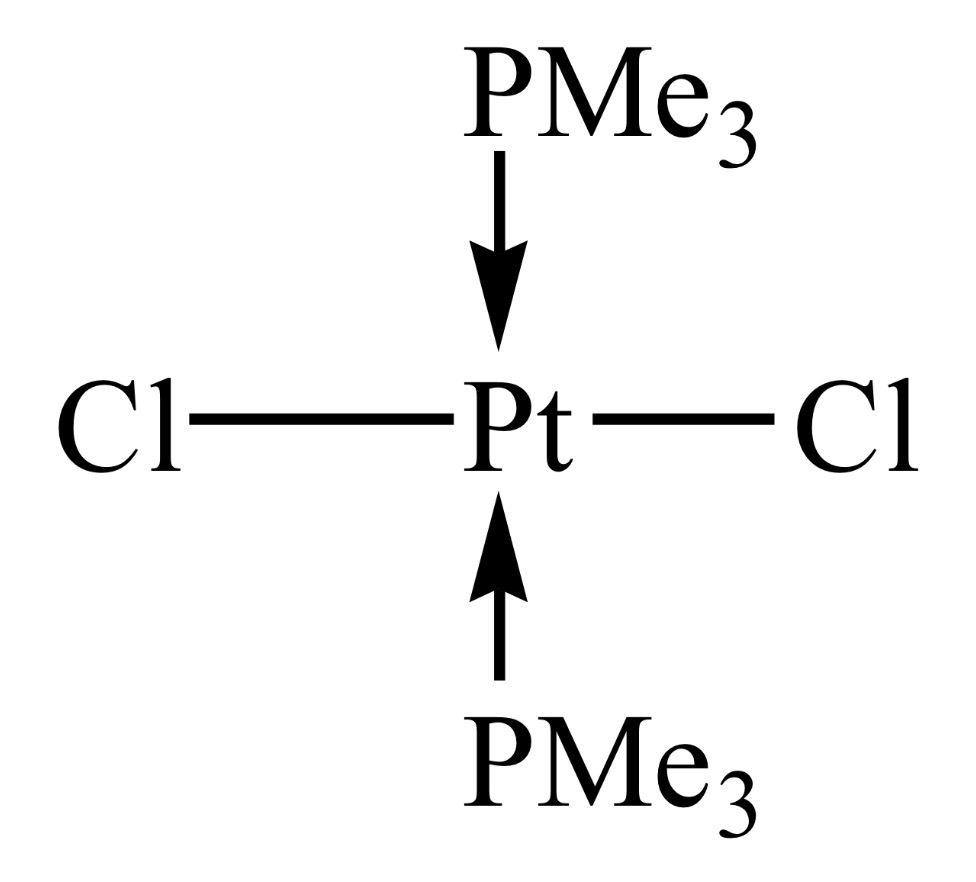
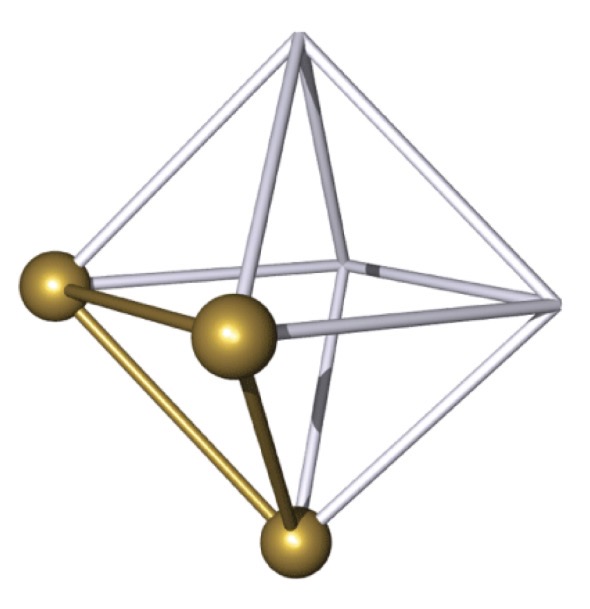
fac-isomer
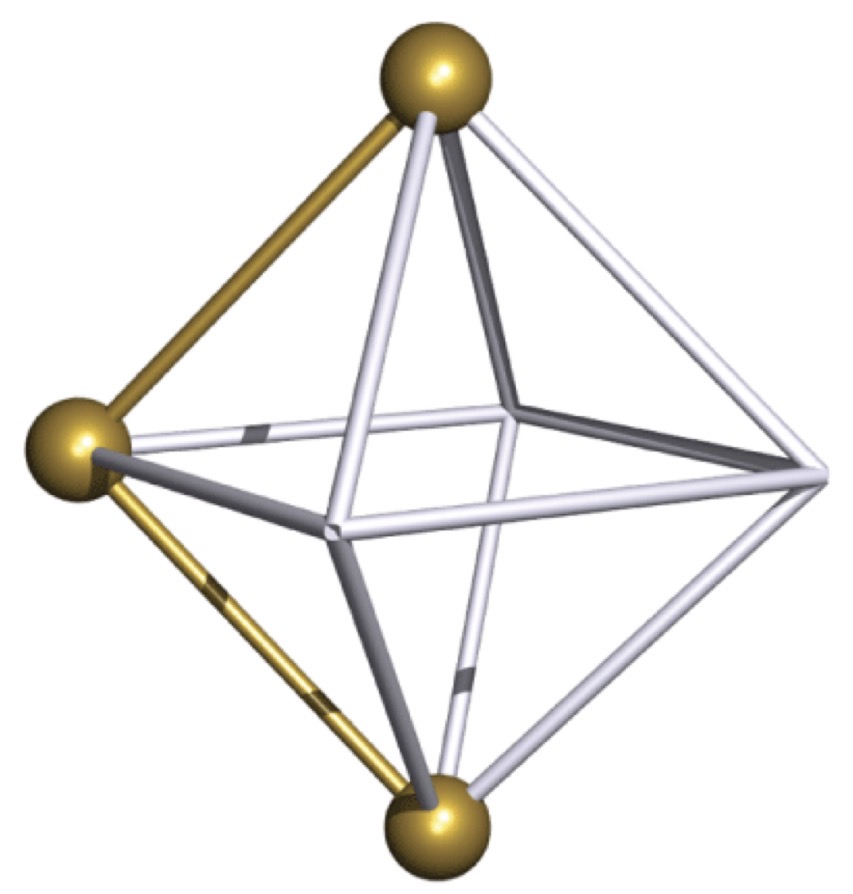
mer-isomer
Valence Bond (VB) Theory
describes bonds in terms of the spin pairing of electrons
Pi
unhybridized p- and d-orbitals may participate in the formation of ___ bonds
Molecular Orbital Theory
the valence shell atomic orbitals are combined to give molecular orbitals (spread over the entire molecule); the method is termed the Linear Combination of Atomic Orbitals (LCAO)
valence electrons are then added to the resulting molecular orbitals; we assume the core electrons are not involved in bonding
Inert Pair Effect
As you go down a group and atoms get larger, the stability of the +1 oxidation state increases, indicating that the atom does not want to give away their s electrons. Their s electrons become more inert.
e.g. Tl: [Xe]6s24f145d105p1
Tl+: [Xe]6s24f145d10
Metallic Radius
half of the distance between the nearest neighbour atoms in a solid state metal lattice, and is dependent on coordination number
Metallic Structures
strong, non-directional cohesive forces
many metals are polymorphic (exist in more than one crystalline form)
Increases
the electrical conductivity of a semiconductor _________ with temperature
Extrinsic Semiconductors
contain dopants, impurities introduced into a semiconductor in minute amounts (1 in 106 atoms) to enhance electrical conductivity.
P-Type
Ga-doped Si yields a _____ semiconductor
N-Type
As-doped Si yields an ______ semiconductor
Intrinsic semiconductors
the extent of occupation of the upper band increases with increasing temperature
Band Gap
Label A
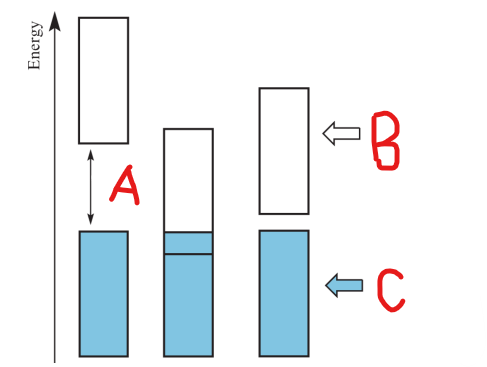
Conduction Band
Label B
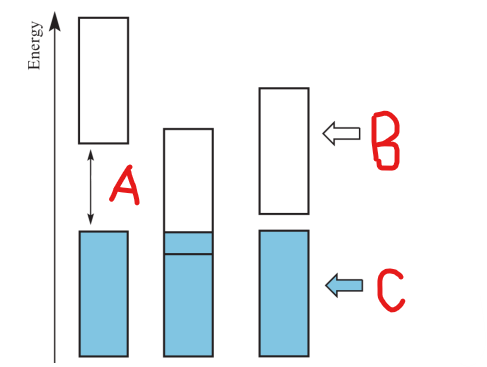
Valence Band
Label C
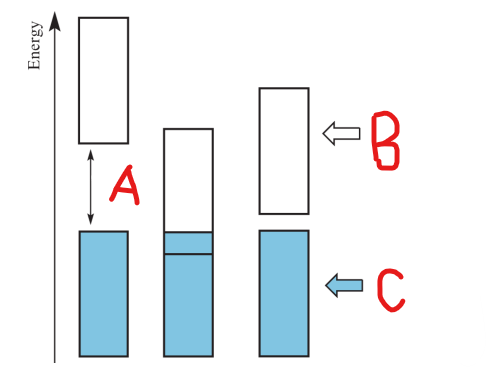
Band Gap
occurs when there is a significant energy difference between two bands
Metals
exhibit high electrical conductivity
Electrical conductor
offers a low resistance to the flow of electrical current
Increases
the electrical conductivity of a wire _______ with temperature
A Band
(in Band Theory) a group of MOs, the energy difference between which are so small that the system behaves as a continuum, non-quantized variation of energy within the band is possible
Ionic Bonding
non-directional electrostatic interactions between charged species (ions)
Lattice Energy
change in internal energy that accompanies the formation of one mole of solid from its constituent gas-phase ions.
Coordination Complex
central atom or ion is coordinated by one or more molecules or ions (ligands) which act as Lewis bases (electron donors), forming coordinate bonds with the central atom or ion
Donor Atoms
In a coordination complex, atoms that are directly bound to the central atom or ion are ________
Lewis Acid
In a coordination complex, the central atom acts as a ________ (electron acceptor)
coordinate (dative) bond
donation of an electron pair from a Lewis base to a Lewis acid; results in the formation of an adduct
Chelate Effect
when dealing with comparable uncharged ligands, polydentate ligands form more stable complexes than monodentate ones
largely driven by a favorable increase in entropy (disorder, S > 0)
Denticity
the number of donor atoms through which a ligand coordinates to a metal ion
Chelate Ring
Coordination of a polydentate ligand to an ion leads to the formation of a _______
6-Membered
small metal cations (e.g. 1st row transition metals) favour the formation of __________ rings
5-Membered
large metal cation favour the formation of ___________ rings
Cryptand
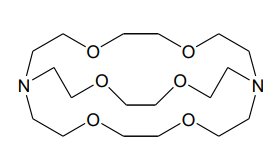
Crown Ethers
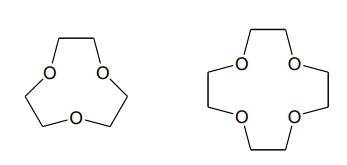
Macrocycles
form more stable complexes than with open-chain complexes (favoured by Go )
Macrocyclic Ligands
Crown ethers, cryptands
Reduction
gain of one or more electrons.
loss of oxygen (or any other more electronegative element).
gain of hydrogen (or any less electronegative element).
Oxidation
loss of one or more electrons.
gain of oxygen (or any other more electronegative element).
loss of hydrogen (or any less electronegative element)
0
the oxidation state of an element is _____, whether it exists as atoms (e.g. Ne), molecules (e.g. O2 , P4 , S8 ), or an infinite lattice (e.g. Si).
the charge on the ion
in monatomic ions, the oxidation number is equal to _____
-2
the oxidation number of oxygen is usually ____
-1
In H2O2 and O22- the oxidation number of oxygen is _____
-1
the oxidation number of hydrogen is +1 except when it is bonded to metals in binary compounds. In these cases, its oxidation number is ____
0
The oxidation number of oxygen in ozone (O3) is _____
-1
the oxidation number of fluorine is always ____
Bronsted Acid
can act as a proton donor
Bronsted Base
can act as a proton acceptor
HF
hydrofluoric acid
HCl
Hydrochloric Acid
HBr
Hydrobromic Acid
HI
Hydroiodic Acids
HX
Hydrogen Halides
Oxoacids
a compound that contains oxygen, at least one other element, at least one hydrogen bound to oxygen, and produces a conjugate base by proton loss.
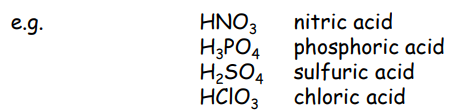
perchloric acid
HClO4
chloric acid
HClO3
chlorous acid
HClO2
hypochlorous acid
HClO
Monobasic Acids
donate only one hydrogen ion () or proton per molecule to a base during an acid-base reaction
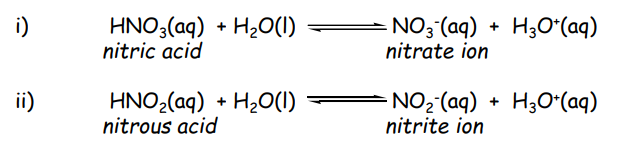
Dibasic Acids
compounds containing two replaceable hydrogen atoms (protons) per molecule, which dissociate in two steps to produce two hydronium ions (H+) in aqueous solution
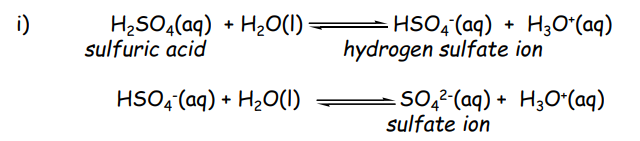
Sulfurous Acid
H2SO3
prepared by dissolving SO2 (g) in H2O
is not isolable in its pure form
Bell’s Rule
empirical method for estimating Ka
n = number of “hydrogen free” oxygen atoms
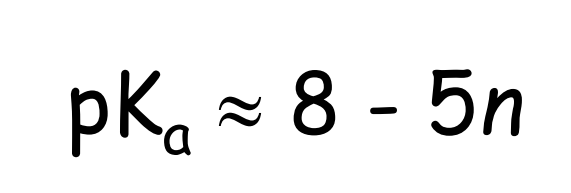
Hydroxides
NaOH, KOH, RbOH, CsOH, LiOH
Lewis Acid
electron pair acceptor
Lewis Base
electron pair donor
Hydration
solvation by water