GW 3.5 Greenhouse Gases
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
For most solids and liquids, what is the emissivity and absorptivity like?
Fairly smooth functions of wavelength
They may be higher at some wavelengths and lower at others
They usually don’t wiggle up and down much
Usually fairly close to 1
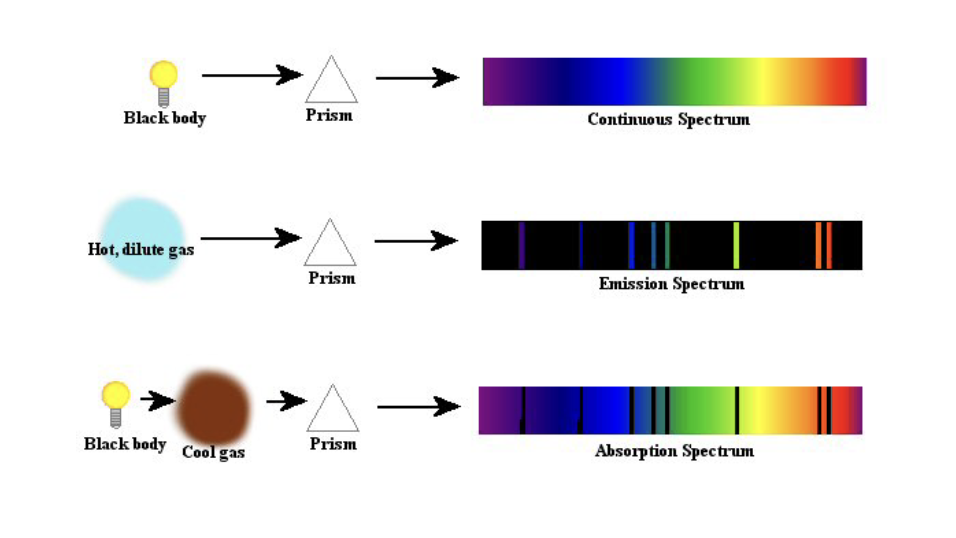
What does absorption for gases depend on?
Strongly depends on wavelength and the amount of gas
In fact, gases can only absorb and emit electromagnetic radiation (light) at certain discrete energies
Why can only particular frequencies and wavelengths of light interact with particular gases?
The energy of photons is proportional to their frequency
What causes the greenhouse effect?
Gases in the atmosphere absorbing the radiation that Earth emits
Earth mostly emits infrared light between 5-30 μm
The gases that absorb a lot of this light are the most imporatnt greenhouse gases
What is the most important greenhouse gas on Earth?
H2O water vapor
Concentration depends strongly on temperature (the hotter it is, the more humid it can be)
Why is CO2 a “very good” greenhouse gas?
It absorbs a lot at wavelengths that Earth emits where H2O doesn’’t
What are “window” regions?
Wavelengths where radiation emitted at Earth’s surface can make it all the way to space without being absorbed
This happens despite all of the greenhouse gases in Earth’s atmosphere