Chem. Oceano. Unit 8: Solubility + Exchange
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
Processes Relating to Gas Exchange
1. Photosynthesis (CO2, O2)
2. Respiration (CO2, O2)
3. Nitrogen Cycle (NH3, N2, N2O)
4. Sulfate Reduction (H2S)
5. Methanogenic Fermentation (CO2, CH4)
6. Algae Growth + Osmotic Regulation (DMS + CCNs)
Way Gases Exist in the Ocean
1. Bubble (gas phase)
2. Solute (surrounded by H2O molecules)
3. Trapped within a solid (ice)
[A]
The absolute concentration or amount/volume of a gas.
Dalton's Law of Partial Pressures
PT = P1 + P2 + ...
Ideal Gas Law
PAPV = nART
T
The absolute temperature in kelvin (K = 273.15 + _____ C).
CA
The volume mixing ratio (VA/Vair).
XA
Mole fraction (nA/nair)
CA = XA
The volume mixing ratio is equal to the mole fraction as a function of the ideal gas law.
STP
Standard temperature (0C) and pressure (1 atm).
22.414 L
At STP, 1 mole of any ideal gas occupies the same volume as ________________.
Mass
Units ~ ppm.
Volume
Units ~ ppm-v
Pascal (Pa)
The SI unit of pressure.
Gas Equilibrium Concentration in Seawater
Means the rates of ingassing + outgassing are equal as functions of salinity + partial pressure.
K
The equilibrium constant equal to [Asw]/[Ag].
Henry's Law Formula
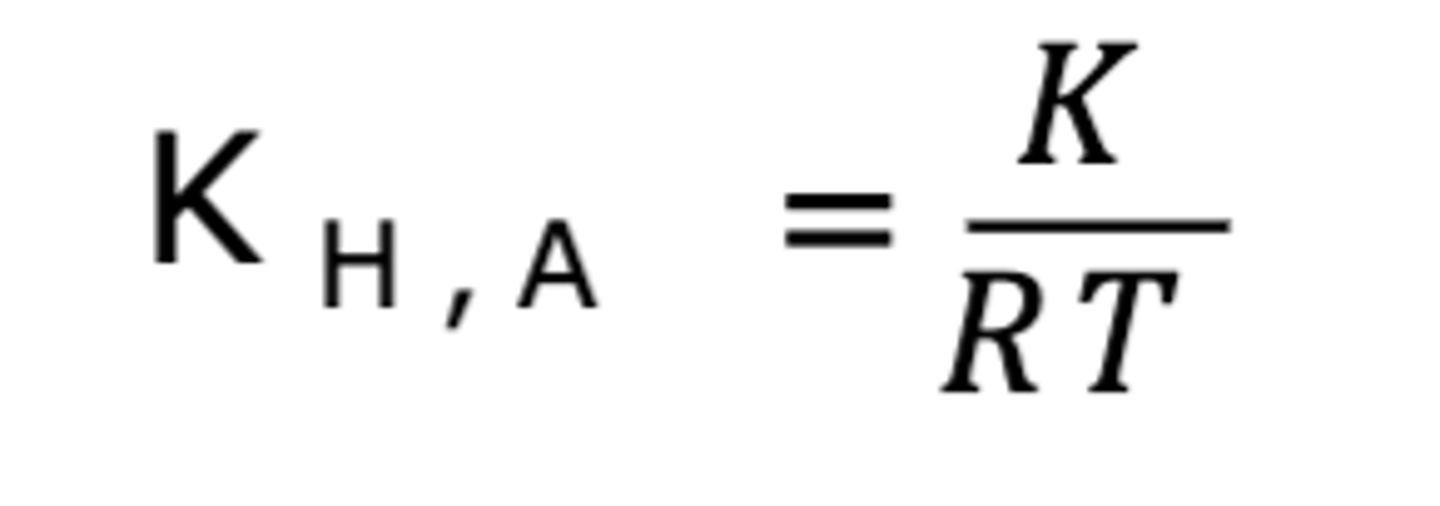
Proportion
Gases dissolve in seawater in ____________ to their partial pressures.
Henry's Law
Describes atm. to ocean dissolution where gases dissolved in seawater, in proportion to their pp.
Bunsen Solubility Coefficient
A version of Henry's Law Coefficient that describes how many lires/mL of gas can be dissolved per L of seawater at 1 atm.
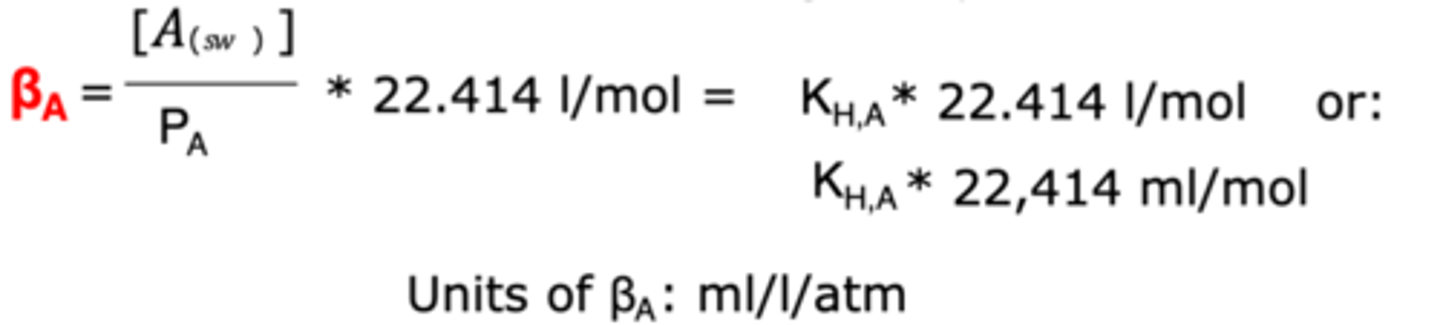
Decreases
The solubility of gases in seawater ____________ w/ temperature.
Decreases
Gas solubility ______________ w/ salinity caused by electrostatic forces exerted by salt ions (higher salinity = lower dissolution).
Normal Atmospheric Equilibrium Concentrations (NAEC)
The gas concentrations determined from Bunsen solubility coefficients obtained for a given water mass under equilibrium conditions.
Disequilibrium
There can be _____________________ between the concentrations of gas in the air and surface sea water.
% Saturation
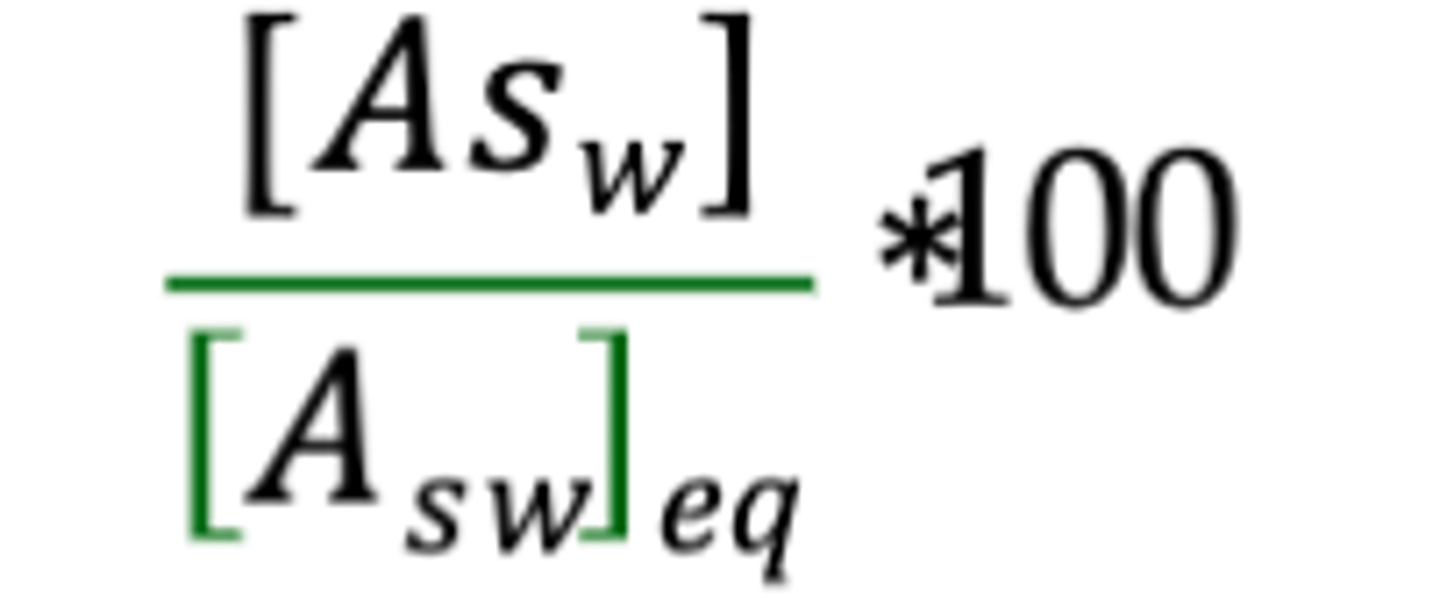
100%
At equilibrium, seawater is ___________ saturated.
Supersaturated
When [Asw] > [Asw]eq.
Deviation from Equilibrium Causes
1. Physical effects
2. Chemical/biological effects
Physical
Non-reactive gases are only affected by ______________ processes (ex. temperature + salinity).
Physical, Biological, + Chemical
Reactive gases (ex. O2, CO2) can be influenced by one or all of _____________________________ processes.
5 Physical Effects Causing Deviations from NAEC
1. Bubble injection
2. Fast changing atm. conditions (T/P)
3. Post-equilibrium water temp. changes
4. Nonlinear effects of mixing
5. Sedimentary seeps/hydrothermal vent sources.
Sea Surface Slicks/Surfactants
Do not create deviations from NAEC but can preserve/erase pre-existing deviations by slowing/accelerating re-equilibrium.
Bubble Injection
AT high winds, air are be forced directly into the sea surface as gas bubbles leading to ONLY supersaturation.
Fast Changing Atmospheric Conditions/Kinetic Consideration
Occurs when environmental conditions change faster than equilibrium can be restored leading to under/supersaturation.
Post-Equilibrium Temperature Changes
The sun can warm the subsurface water after NAEC has been reached, leading to the oversaturation of CO2 and O2.
Nonlinear Effects of Mixing
Because the line of solubility as a function of T is concave upward (nonlinear), it can only increase saturation - stronger on inert gases.
Physical Subsurface Sources
Hydrothermal vents/seeps release gas in vent water leading to under/supersaturation.
Physical Obstructions to Air-Sea Fluxes
1. Slicks
2. Surfactants
3. Sea Ice
Slicks
Slow the exchanges through the air-sea interface and delay the equilibration after a change in conditions.
Surfactants
Acceleration the exchanges at the air-sea interface + equilibration.
Sea Ice
Works like a slick but thicker - greatly slows/blocks the exchanges at the air-sea interface.
Inert Gases
Are not affected by biological processes + are useful tracers of physical processes.
Subsurface Bubbles
Mediate gas exchange by affecting the solubility of gases.
Undersaturated Gases
In deep water, more soluble + temp. dependent gases are ____________________ and sank before reaching 100% saturation from cooling.
Supersaturated Gases
The less soluble gases near equilibrium that reached 100% before they sunk.
Surface/Subsurface Peak
The strongest for most temp. sensitive gases - warming by the sun increases % saturation = more temp. sensitive + oversaturated.
Biological Processes Affecting NAEC
1. Photosynthesis
2. Aerobic Respiration/Microbial Decomposition
3. Anaerobic Respiration
4. Methanogenic Respiration
Photosynthesis
Consumes CO2 producing O2 in shallow, sunlit waters (coastal upwellings).
Aerobic Respiration/Microbial Decomposition
When algae sink too deep for photosynthesis, ______________________ consumes O2 producing CO2 (pycnocline + surface of sediments).
Anaerobic Respiration + Methanogenic Fermentation
Occurs where O2 is gone (anoxic waters) producing CO2, H2S, and CH4 (cold seeps).
Vertical Oxygen Profile Features
1. Surface
2. Subsurface peak
3. Pycnocline/OMZ
4. Deep water
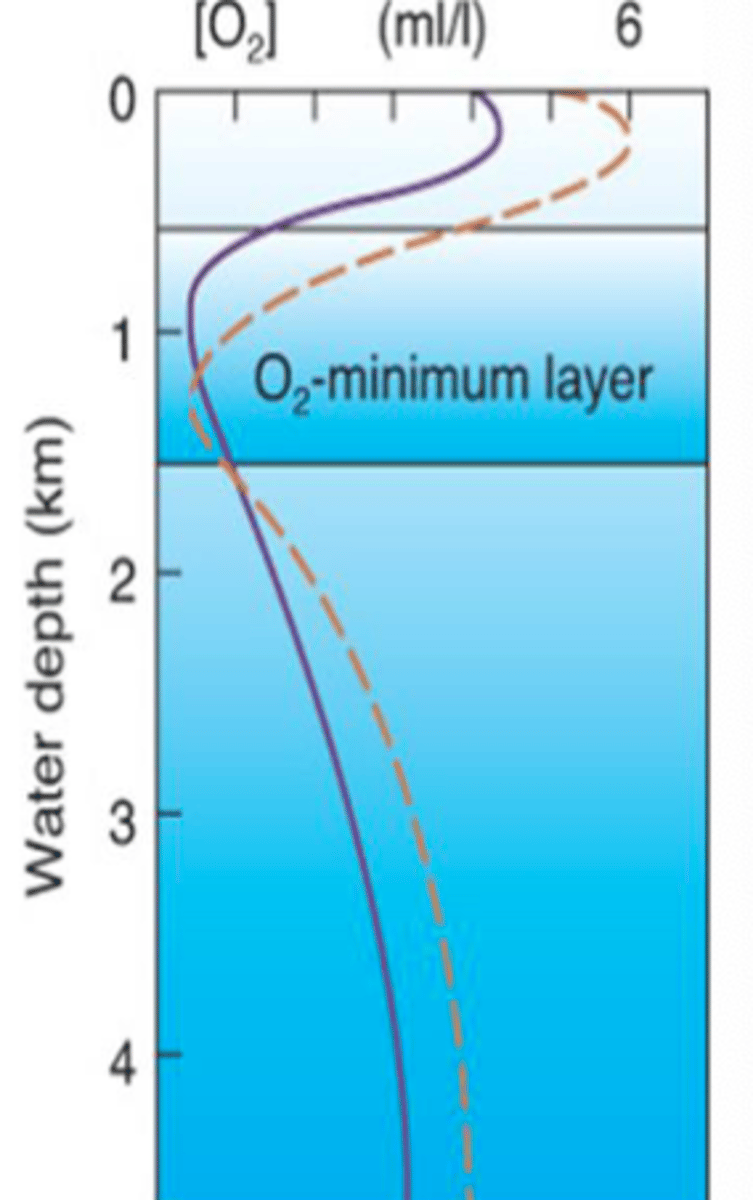
Surface
Air-sea interface - exchange of gases w/ air.
Subsurface Peak
Exhibits the maximum photosynthesis.
Pycnocline/Oxygen Minimum Zone (OZM)
Some sinking of OM gets stuck here and respiration by animals/decomp. by microbes consumes O2.
Decreases
The strong density stratification exhibited by the pycnocline/OMZ ________________ vertical mixing and diffusion from above/below waters.
Deep Waters
Have higher O2 since water brought by the THC from the NA was was cold/had a higher NAEC when it sunk.
Anoxia
Total depletion of O2.
Hypoxia
A less severe version on anoxia w/ low O2.
Anoxic Ocean Regions
1. Open ocean
2. OMZ
3. Ocean floor
4. Estuaries
5. Seas w/ estuarine circulation
Conditions for Open Ocean Anoxia
1. Large supply of OM = respiration/decomp. = ↓ O2
2. Pycnocline/geological barriers prevent mixing w/ O2-rich waters.
OMZ Anoxia
Occurs in the pycnocline where OM gets stuck and respiration by animals/decomp. by microbes consumes O2.
Ocean Floor Anoxia
Occurs when the OMZ intercepts the continental slope and near areas of upwelling.
Estuary Anoxia
River runoff + little evap increases primary production in the surface layer and decomp in the deep layer - no mixing (geological barrier - fjords) leads to O2 depletion in the deep layer
Estuary-Like Sea Anoxia
Occurs where partially restricted basins behave like estuaries where warm river water sits on top of deep water (strong strat.) + the production of algae in surface water = decomp in deep water/deep water O2 deficit (ex. Black Sea, Baltic Sea).