Chem 215-1: Quiz 1
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
When is a resonance structure lower in energy?
More atoms with a completed octet
Fewer atoms nonzero formal change
A -1 formal charge appearing on more electronegative atom (and +1 on less electronegative)
Why are resonance structures stabilized?
Delocalization of electrons because electrons are less confined
When is a structure highly stabilized?
When there are more equivalent resonance structures
What do nonequivalent resonance structure do?
They have a small contribution to the hybrid
What features indicate that another resonance structure exists? (not ranked)
Lone pair on an atom adjacent to a multiple bond
Atom lacking an octet adjacent to a multiple bond
Atom with a lone pair adjacent to an atom lacking an octet
Polar multiple bond (can lead to no octet and formal charges on 2 atoms)
Ring of alternating single and multiple bonds (benzene)
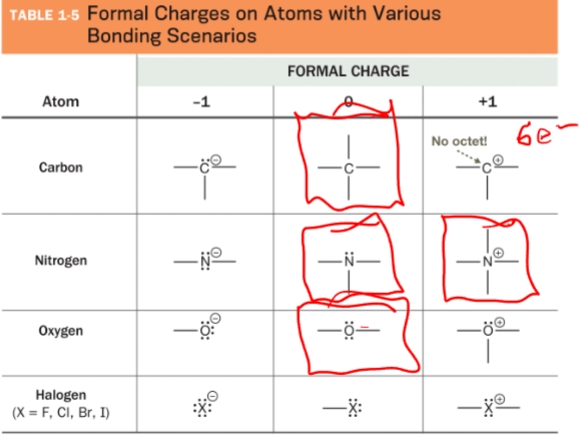
Just memorize
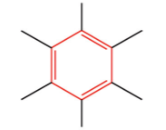
What is this?
Arene/aromatic compound
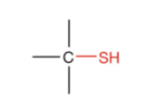
What is this?
Thiol
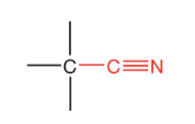
What is this?
Nitrile
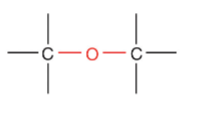
What is this?
Ether
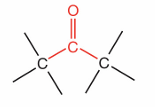
What is this?
Ketone
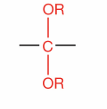
What is this?
Acetal
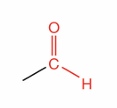
What is this?
Aldehyde
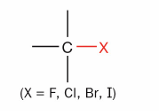
What is this?
Alkyl halide
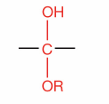
What is this?
Hemiacetal
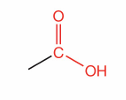
What is this?
Carboxylic acid
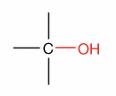
What is this?
Alcohol

What is this?
Epoxide
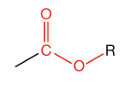
What is this?
Ester
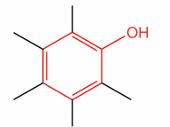
What is this?
Phenol
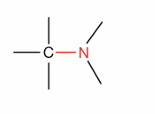
What is this?
Amine
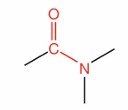
What is this?
Amide
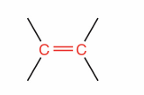
What is this?
Alkene
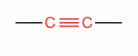
What is this?
Alkyne
In dash wedge notation, which one is towards you?
Wedge
Do alkanes contain functional groups?
No
What are the basic alkanes?
Straight-chain / linear alkanes
No branches
What are the prefixes for nomenclature?
Meth, eth, prop, but, pent, hex, hept, oct, non, dec
What are substituted alkanes?
If a hydrogen atom of an alkane is replaced by another atom or group of atoms → substituted
What are the prefixes for halo substituents and NO2 substituents?
Fluoro, chloro, bromo, iodo, nitro
What are the prefixes for substituents?
di, tri, tetra, penta, hexa, hepta, octa, nona, deca
How to determine if a ring is a root or substituent?
Compare number of carbons in ring vs in longest continuous chain (if in ring, NOT in chain)
Called cycloalkane if root (ex. cyclopropane)
Called cycloalkyl if substituent (ex. cyclopropyl)
What is isopropyl?
End of carbon chain with two methyls off of it
Iso___yl refers to a chain of carbons where a methyl exists on C-2 (looks like carbon chain with 2 methyls off it)
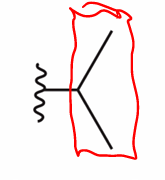
What is tert-butyl
Carbon chain with 3 methyl groups coming off it
All tert means is that the terminal carbon is attached to 3 other carbons (3 methyl groups)
ADD VSEPR HERE
When atoms are connected by a multiple bond, are they in the same plane or different?
Those atoms and atoms to which they are directly bonded prefer to lie in same plane
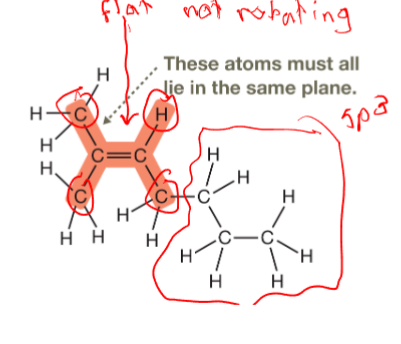
Why does cis-1,2-dichoroethene have a higher boiling point than its trans counterpart?
Due to the dipole that’s formed
How do you determine if a molecule has two distinct configurations about the double bond?
See if there are two different atoms/groups attached to each double bonded C (on C has 2 different groups)
What happens to the bond length as hybridization goes from sp3 to sp2 to sp?
Become shorter (+ stronger)
Has more s character, so bond length decreases because s orbital is closer to the nucleus than the p orbital
What happens to the effective electronegativity as hybridization goes from sp3 to sp2 to sp?
Increases (more polar portions of the molecule)
What does a node represent?
Surfaces where there is a zero probability of finding an electron
What are the relative energy levels of bonding, antibonding and nonbonding MOs?
Bonding is significantly lower in energy than contributing AOs
Antibonding is significantly higher in energy than its contributing AOs
Non-bonding has the same energy as contributing AOs
What are the HOMO and LUMO?
HOMO = highest energy w/ an electron, LUMO = lowest empty
ADD Last two slides from lecture 4, about hybrid atomic orbitals