1.1 Energy Changes in a System
1/52
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
What is a system?
An object or a group of objects.
What happens when a system changes?
There are changes to the way energy is stored.
What are the changes involved in the way energy is stored when an object is projected upwards?
Kinetic energy as it goes up — it is moving quickly
As it gets higher and slows down — kinetic energy → gravitational potential energy
Once it reaches its highest point (and stops momentarily):
kinetic energy — zero
gravitational potential energy — maximum
What are the changes involved in the way energy is stored when a moving object hits an obstacle?
Starts with kinetic energy
On impact, kinetic energy = zero
Energy is transferred to the thermal energy stores of object and obstacle
Makes them warmer
Some energy is transferred to the surroundings as sound waves
What are the changes involved in the way energy is stored when an object is accelerated by a constant force?
Energy is transferred to its kinetic energy store — it speeds up
What are the changes involved in the way energy is stored when a vehicle slows down?
Energy is transferred away from its kinetic energy store
As brakes are applied, friction causes this energy to be transferred mechanically into the thermal energy stores of the brakes and the air (they heat up)
What are the changes involved in the way energy is stored when you bring water to a boil in an electric kettle?
Energy is transferred electrically from the mains supply to the thermal energy store of the kettle’s metal heating element.
Energy is then transferred by heating into the thermal energy store of the water — causes its temperature to rise until it boils.
What is the equation for kinetic energy? State the units.
kinetic energy = 0.5 × mass × (speed)²
Ek=21mv2
kinetic energy — joules (J)
mass — kilograms (kg)
speed — metres per second (m/s)
What is the equation for elastic potential energy? State the units.
elastic potential energy = 0.5 × spring constant × (extension)²
Ee=21ke2
elastic potential energy — joules (J)
spring constant — newtons per metre (N/m)
extension — metres (m)
What assumption do we make when using the equation for elastic potential energy?
The limit of proportionality has not been exceeded.
What is the equation for gravitational potential energy? State the units.
gravitational potential energy = mass × gravitational field strength × height
Ep=mgh
gravitational potential energy — joules (J)
mass — kilograms (kg)
gravitational field strength — newtons per kilogram (N/kg)
height — metres (m)
What is the typical value of gravitational field strength?
9.8 N/kg
What is the equation for the amount of energy stored in or released from a system as its temperature changes (change in thermal energy)? State the units.
change in thermal energy = mass × specific heat capacity × temperature change
ΔE=mcΔθ
change in thermal energy — joules (J)
mass — kilograms (kg)
specific heat capacity — joules per kilogram per degree Celcius (J/kg°C)
temperature change — degrees Celcius (°C)
What is the specific heat capacity of a substance?
The amount of energy required to raise the temperature of 1 kg of the substance by 1°C.
Required Practical 1 — Specific Heat Capacity (Joulemeter):
Describe a method for investigating the specific heat capacity of different metals.
Use the equation to help you: ΔE=mcΔθ
Zero the balance (removes zero error) and measure the mass of the metal block.
Wrap insulation around the block — reduces heat loss to the surroundings.
Add a few drops of water into the thermometer hole — improves thermal contact.
Insert the thermometer and record the initial temperature.
Insert an immersion heater into the block, connect it to a joulemeter and power supply, and switch it on.
Allow the block to heat up (until temperatures rise by ~15°C), then record the highest final temperature and the total energy transferred from the joulemeter.
Use the equation: ΔE=mcΔθ
Required Practical 1 — Specific Heat Capacity (Voltmeter & Ammeter):
Describe a method for investigating the specific heat capacity of different metals.
Use the equation to help you: ΔE=mcΔθ
Zero the balance (removes zero error) and measure the mass of the metal block.
Wrap insulation around the block — reduces heat loss to the surroundings.
Add a few drops of water into the thermometer hole — improves thermal contact.
Insert the thermometer and record the initial temperature.
Insert an immersion heater into the block, connect it to a power supply with an ammeter in series and a voltmeter in parallel across the heater, start a stopwatch, and switch it on.
Allow the block to heat up (until temperatures rise by ~15°C), then record the highest final temperature, the current from the ammeter, the potential difference from the voltmeter, and the total time in seconds.
Calculate the total energy transferred using E=IVt , then use the equation: ΔE=mcΔθ.
What is the independent variable in RP1?
The energy supplied by the heater
What is the dependent variable in RP1?
The temperature of the material
What are the control variables in RP1?
The mass of the block
The power of the heater
The insulation used
What are the sources of error in RP1 — how can they be fixed?
Heat loss to the surroundings
Wrap the block in an insulator with low thermal conductivity — reduced thermal energy transferring to the air
Poor thermal contact
Add a few drops of water into the thermometer hole — improves thermal contact and ensures an accurate reading
Incomplete energy transfer
Make sure the immersion heater is fully inserted into the material — allows all heat to be transferred usefully
Misreading the thermometer
Use an electronic temperature probe — prevents parallax errors
![<ul><li><p>A student used two different types of thermometer to measure the temperature changes for RP1. </p></li><li><p>Figure 2 shows a reading on each thermometer.</p></li></ul><p>What is the resolution of thermometer B? [1 mark]</p>](https://assets.knowt.com/user-attachments/7bbd8232-062a-4005-831a-fcec8e7e2a12.png)
A student used two different types of thermometer to measure the temperature changes for RP1.
Figure 2 shows a reading on each thermometer.
What is the resolution of thermometer B? [1 mark]
0.1 °C
![<ul><li><p>A student used two different types of thermometer to measure the temperature changes for RP1. </p></li><li><p>Figure 2 shows a reading on each thermometer.</p></li></ul><p>Thermometer A is more likely to be misread. Give one reason why. [1 mark]</p>](https://assets.knowt.com/user-attachments/d601c850-0684-40ab-9ae1-1070f7adef2d.png)
A student used two different types of thermometer to measure the temperature changes for RP1.
Figure 2 shows a reading on each thermometer.
Thermometer A is more likely to be misread. Give one reason why. [1 mark]
Parallax error

![<ul><li><p>A student used the apparatus drawn to investigate the heating effect of an electric heater.</p></li><li><p>Before starting the experiment, the student drew Graph A. </p></li><li><p>Graph A shows how the student expected the temperature of the metal block to change after the heater was switched on.</p></li></ul><p>Describe the pattern shown in Graph A. [2 marks]</p>](https://assets.knowt.com/user-attachments/9d924aca-9a3f-484f-86f2-d35c46454888.png)
A student used the apparatus drawn to investigate the heating effect of an electric heater.
Before starting the experiment, the student drew Graph A.
Graph A shows how the student expected the temperature of the metal block to change after the heater was switched on.
Describe the pattern shown in Graph A. [2 marks]
Temperature increase and time switched on are directly proportional.

![<ul><li><p>A student used the apparatus drawn to investigate the heating effect of an electric heater.</p></li><li><p>Before starting the experiment, the student drew Graph A. </p></li><li><p>Graph A shows how the student expected the temperature of the metal block to change after the heater was switched on.</p></li><li><p>The student measured the room temperature. He then switched the heater on and measured the temperature of the metal block every 50 seconds. </p></li><li><p>The student calculated the increase in temperature of the metal block and plotted Graph B.</p></li><li><p>After 300 seconds, Graph B shows the increase in temperature of the metal block is lower than the increase in temperature expected from Graph A. </p></li></ul><p>Suggest one reason why. [1 mark]</p>](https://assets.knowt.com/user-attachments/57e2af8c-bff5-4e59-98e1-ab4e8c61e024.png)
A student used the apparatus drawn to investigate the heating effect of an electric heater.
Before starting the experiment, the student drew Graph A.
Graph A shows how the student expected the temperature of the metal block to change after the heater was switched on.
The student measured the room temperature. He then switched the heater on and measured the temperature of the metal block every 50 seconds.
The student calculated the increase in temperature of the metal block and plotted Graph B.
After 300 seconds, Graph B shows the increase in temperature of the metal block is lower than the increase in temperature expected from Graph A.
Suggest one reason why. [1 mark]
Energy transfer from the block to the surroundings.

A student uses the same heater to heat blocks of different metals.
Each time the heater is switched on for 300 seconds.
Each block of metal has the same mass but a different specific heat capacity.
Which one of the metals will heat up the most? Give, in terms of the amount of energy needed to heat the metal blocks, a reason for your answer. [2 marks]
Metal | Specific Heat Capacity in J/kg°C |
Aluminium | 900 |
Iron | 450 |
Lead | 130 |
Lead
Needs the least energy to raise temperature by 1°C

What is power?
The rate at which energy is transferred.
or
The rate at which work is done.
What is the equation for power? — involving energy transferred. State the units.
power = energy transferred + time
P=tE
power — watts (W)
energy transferred — joules (J)
time — seconds (s)
What is the equation for power? — involving work done. State the units.
power = energy transferred + time
P=tW
power — watts (W)
work done — joules (J)
time — seconds (s)
An energy transfer of 1 _______ _____ _________ is equal to a power of 1 ____.
joule per second
watt
What are the eight different energy stores?
Thermal
Kinetic
Gravitational potential
Elastic potential
Chemical
Magnetic
Electrostatic
Nuclear
What is meant by a thermal energy store?
The total kinetic and potential energy of the particles in an object.
What are some examples of thermal energy stores? (3)
Human bodies
Hot coffees
Stoves
What is meant by a kinetic energy store?
The energy a system has because it is moving.
What are some examples of kinetic energy stores? (3)
Runners
Buses
Comets
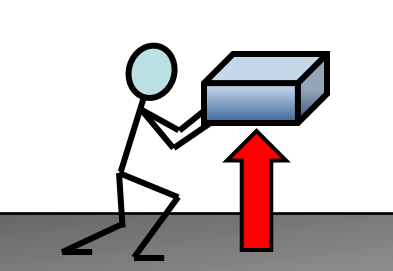
What is meant by a gravitational potential energy store?
The energy a system has because of how high above the ground it is.
What are some examples of gravitational potential energy stores? (3)
Aeroplanes
Kites
Mugs on a table

What is meant by a chemical energy store?
The energy stored in chemical bonds.
What are some examples of chemical energy stores? (3)
Food
Fuels
Batteries


What is meant by an elastic potential energy store?
The energy stored in a system when it is stretched or squashed.
What are some examples of elastic potential energy stores? (3)
Drawn catapults
Compressed springs
Inflated balloons

What is meant by a thermal energy store?

What is meant by a magnetic energy store?
The energy stored when two repelling magnetic poles have been pushed together or two attracting magnetic poles have been pulled apart.
What are some examples of magnetic energy stores? (2)
Fridge magnets
Compasses

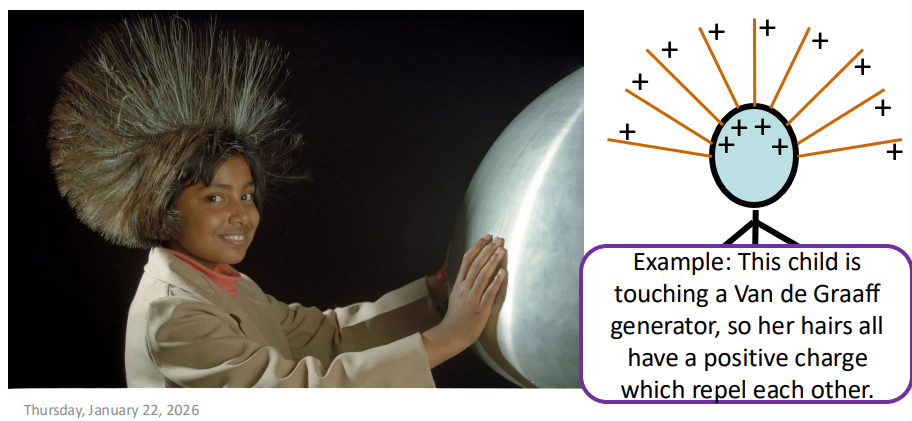
What is meant by an electrostatic energy store?
The energy stored when repelling charges have been moved closer together or attracting charges have been pulled apart.
What are some examples of electrostatic energy stores? (2)
Thunderclouds
Van De Graaff generators
What is meant by a nuclear energy store?
The energy stored in the nucleus of an atom.
What are the four ways energy can be transferred?
Mechanical
Electrical
Heating
Radiation
What is meant by the mechanical energy transfer pathway?
Energy is transferred mechanically when a force is used to move an object a certain distance.
This is called the work done by the force.
What is meant by the heating energy transfer pathway?
Energy is transferred by heating when there is a temperature difference — so energy flows from a hotter area to a colder area.
What is meant by the electrical energy transfer pathway?
Energy is transferred electrically when a charge flows around a circuit.
This is called the work done when an electrical current flows.
What is meant by the radiation energy transfer pathway?
Energy is transferred by radiation when it is transferred by waves or particles.
![<ul><li><p>In a ride at a theme park, a person is strapped into a pod that is attached to two stretched bungee cords. </p></li><li><p>The bungee cords behave like springs. </p></li><li><p>The figure shows a person using the ride.</p></li></ul><p>Which energy store increases as the bungee cords are stretched? [1 mark]</p>](https://assets.knowt.com/user-attachments/346c10e1-568e-4e79-a50c-8d12c6a4de35.png)
In a ride at a theme park, a person is strapped into a pod that is attached to two stretched bungee cords.
The bungee cords behave like springs.
The figure shows a person using the ride.
Which energy store increases as the bungee cords are stretched? [1 mark]
Elastic potential

![<ul><li><p>A fire piston is a special type of syringe that can be used to start fires.</p></li><li><p>Figure 2 shows a fire piston.</p></li><li><p>The plunger is pushed quickly downwards and compresses the air. </p></li><li><p>When the air is compressed quickly, the temperature of the air increases.</p></li></ul><p>How does an increase in temperature affect the air particles inside the piston? [1 mark]</p>](https://assets.knowt.com/user-attachments/af638318-fb7e-443a-805f-17ef12bfba98.png)
A fire piston is a special type of syringe that can be used to start fires.
Figure 2 shows a fire piston.
The plunger is pushed quickly downwards and compresses the air.
When the air is compressed quickly, the temperature of the air increases.
How does an increase in temperature affect the air particles inside the piston? [1 mark]
The mean kinetic energy of the particles increases.