Topic 5 - Energy Changes
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
What is conservation of energy
When chemical reactions occur, energy is conserved:
The amount of energy in the universe at the beginning is the same as at the end.
This means if a reaction transfers energy to the surroundings, the product molecules must have less energy than the reactants, by the amount transferred
Exothermic reactions
An exothermic reaction is one that transfers energy to the surroundings, so the temperature of the surroundings increases.
Product molecules must have less energy than the reactants by the amount transferred.
Examples of exothermic reactions include:
combustion
many oxidisation reactions
neutralisation
Everyday examples of exothermic reactions include:
self-heating cans (eg, for coffee)
hand warmers
Endothermic reactions
An endothermic reaction is one that takes in energy from the surroundings, so the temperature of the surroundings decreases
Product molecules must have more energy than reactants
Examples of endothermic reactions include:
thermal decomposition
the reaction of citric acid and sodium hydrogencarbonate
Some sports injury packs are based on endothermic reactions
Exothermic and Endothermic energy profile diagrams
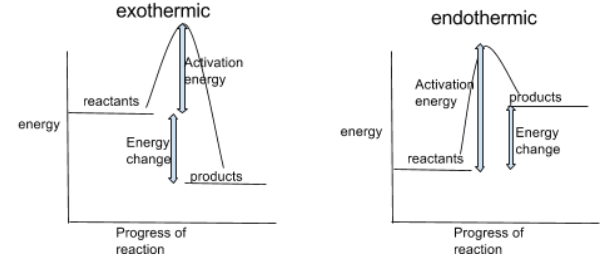
What do energy profiles show
Energy profiles can be used to show the relative energies of reactants and products, the activation energy, and the overall energy change of a reaction
You can tell which reaction is exothermic or endothermic by looking at whether the reactants or products have more energy (higher up the energy scale = higher energy)
Exothermic:
heat is released to the surroundings, so products have less energy than reactants
it’s exothermic because we finished with less energy than we started with
Endothermic:
heat taken in from the surroundings, so products have more energy than reactants
it’s endothermic because we finished with more energy than we started with
Energy change of reactants - what happens during a chemical reaction
Energy must be supplied to break bonds in the reactants
Energy is released when bonds in the products are formed
The energy needed to break bonds and the energy released when bonds are formed can both be calculated from bond energies