BRS - initial flashcard bulk 1
1/1439
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
1440 Terms
what is a neurone?
excitable non-dividing cells of the CNS and PNS
transfer electrical signals
heterogenous morphology
in terms of morphology what are the 4 types of neuronal cells:
unipolar
pseudo-unipolar
bipolar
multipolar
unipolar neurone features:
one axon projection
no dendritic projection
pseudo-unipolar features
one axonal projection that divides into 2
no dendritic projection
bipolar features:
one axonal projection
one dendritic projection
multipolar features:
one axonal projections
multiple dendritic features
recall the 3 types of multipolar neurones:
purkinje
pyramidal
Golgi
recall the 3 main structural components of a neurone:
cell body (perikaryon)
axon
dendrites
describe the difference in the function of a dendrite and an axon:
an axon is used to transmit signals to other neurones
a dendrite receives signals from other neurones
Recall the five types of cell in the CNS:
Neuron
Oligodendrocyte
Astrocyte
Microglia
Ependyma
name 4 neuroglia:
Oligodendrocyte
Astrocyte
Microglia
Ependyma
describe the role of oligodendrocytes:
they produce glia which produce myelin
allow signals to move faster across nervous system - amplify signal
what is the most abundant type of cell in CNS?
astrocyte
describe the role of microglia?
they are immune cells
neuronal macrophages
describe the role of ependymal cells:
epithelial cells that line the cerebral ventricles (brain) and spinal cord
describe the role of an astrocyte:
structural support
mop up excess NT and waste
What is the difference between oligodendrocytes & Schwann cells?
Oligodendrocytes → produce myelin CNS, myelinate number of axons
Schwann cells → produce myelin in PNS, only myelinate single axonal segment
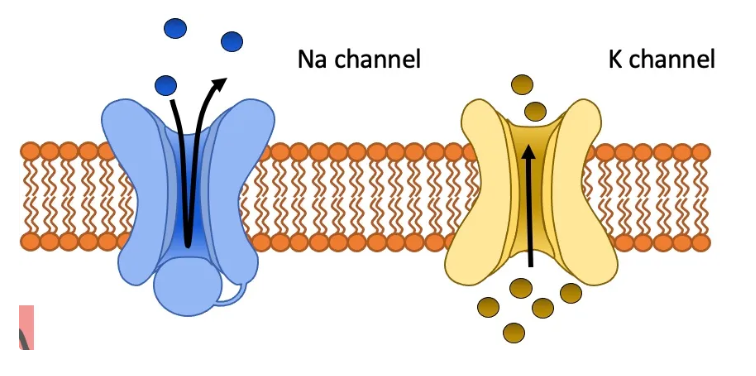
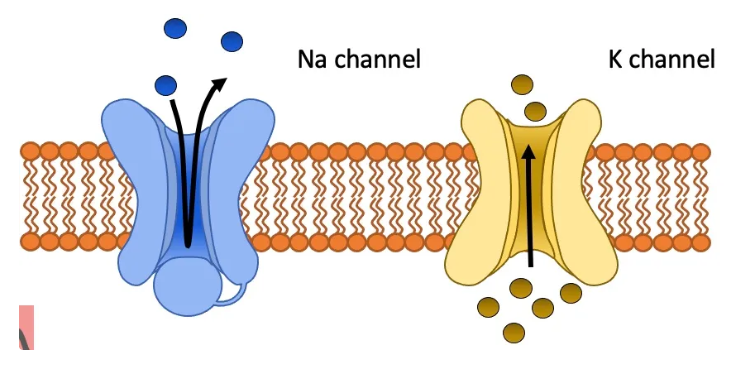
what are the 4 main physiological ions:
Na+
K+
Ca2+
Cl-
which ions have high extracellular concentrations:
Na+
Cl-
which ions have high intracellular concentrations:
K+
define action potential:
the change in membrane potential that occurs in a neurone when a signal is being transmitted
what value is RMP:
-70mV
What is the state of the VGSCs and VGKCs during RMP?
closed
what causes membrane depolarisation?
opening of VGSCs
influx of Na+
further depolarisation makes it -40mV
what causes membrane repolarisation?
opening of VGKCs
efflux of K+
membrane repolarisation restores it to -70mV
how is RMP restored?
by Na+K+ATPase as AP leaves there is an imbalance between Na and K
so pumps 3Na+ out of cell and 2K+ in
resting configuration:
Na+ enters vestibule and upon phosphorylation → 3Na+ ions transported through protein
active configuration:
Na+ removed from cell, phosphate detaches from pump
conformational change
K+ enters vestibule
nodes of ranvier definition
The gaps between the myelin sheath on axons aren’t covered in myelin
what is saltatory conduction?
AP jumps using nodes of ranvier instead of whole axon
what happens at the end of an axon?
AP unable to jump across axon terminal → stops here
Describe the process of neurotransmission across a synapse. (7)
membrane depolarisation due to Na+ influx
membrane repolarisation due to K+ efflux
AP triggers VGCCs at pre-synaptic terminal to open
Ca2+ influx causes vesicles exocytosis
NT binds to receptors on post-synaptic neurone
Influx of Na+ causes AP in post-synaptic neurone
NT dissociates from receptor and recycled in synaptic cleft (metabolised by enzymes/recycled by transporter proteins)
What happens to neurotransmitters after an action potential has been triggered?
disassociate from receptor and metabolised by enzymes in synaptic cleft
can also be recycled by transporter proteins
Recall the three types of communication that axons use to communicate with other neurones.
Axodendritic synapse (axon-to-dendrite)
Axosomatic synapse (axon-to-somatic cell)
Axoaxonic synapse (axon-to-axon)
Order them in speed from fastest to slowest.
Axoaxonic
Axosomatic
Axodendritic
What is a neuromuscular junction?
neurones to muscle
Which neurotransmitter works at the neuromuscular junction?
Acetylcholine
What type of signalling is involved between the nerve and effecter cell?
paracrine NT release, only uni-directional
Which NT is involved in NMJs?
ACh
which post-synaptic receptor is involved in the NMJ?
nicotinic ACh receptors (nAChR)
Describe how the transmission of signal from a neurone causes muscle cells to contract.
ACh binds to nAChR
stimulates the influx of Na+ causing depolarisation into muscle cell and sarcoplasm
depolarisation causes Ca2+ in muscle cell to be released from sarcoplasmic reticulum
Ca2+ binds to myofibrils causing muscle contraction
What is an end plate potential?
local depolarisation of muscle fibres as a result of NTs binding to post-synaptic membranes
graded potential depends on size of stimulus
What are miniature EPP?
smaller release of ACh
How is excitation of the post-synaptic neurone coupled to muscle contraction (excitation-contraction coupling)?
ACh binds to nAChR activating it in skeletal muscle membrane (sarcolemma)
stimulates the influx of Na+ causing depolarisation of sarcolemma produces AP
AP travels through T-tubules
depolarisation from sarcolemma activates dihydropyridine receptors (DHPR) causing conformational change
this change is transmitted from RyR on sarcoplasmic reticulum → open and release Ca2+ from intracellular stores
Ca2+ activates myofibril so muscle contraction occurs
3 disorders of NMJ:
botulism
MG
LEMS
botulism features:
inhibits release of ACh from pre-synaptic receptors
muscle paralysis as can’t depolarise or contract
MG features:
autoimmune disorder where antibodies block nAChR
causes fatiguable weakness with repetitive use as muscles can’t contract or relax
LEMS
autoimmune disorder where antibodies block VGCCs
calcium can’t enter presynaptic neurone - no ACh release
How can myasthenia gravis (MG) be distinguished from LEMS?
In MG, there tends to be facial muscle weakness which is not typically characteristic of LEMS.
what is flux in the context of diffusion?
The number of molecules that cross a unit area per unit of time.
membrane potential definition:
difference in voltage between the inside and outside of the cell
voltage definition
difference in charges between two areas to create a gradient
current definition
current describes the movement of these ions across a concentration gradient
typical membrane potential of a resting cell:
-70mV
means inside of cell is slightly more negative than outside
When does movement of molecules across a membrane stop?
when an equilibrium has been achieved
What is an electrochemical equilibrium?
The state at which the concentration gradient of certain particles in and out of an environment has been opposed and balanced out by an electrical one
no net movement of molecules
equilibrium potential definition
membrane potential at which electrochemical equilibrium has been reached
potential prevents further movement of molecules across cell membrane
What factors affect the opening and closing of ion channels? (3)
transmembrane voltage (membrane potential) e.g. VGCs
presence of activating ligand (attaching to them)
mechanical forces
What is the Nernst equation used for?
to calculate the equilibrium potential of a cell using the concentrations of ions on either side of the membrane
relies on assumption that membrane is permeable to ion in question
What is the Goldman-Hodgkin-Katz (GHK) equation used for?
more accurate model of Nernst equation
cell membrane has varying permeability to ions at different times
What are the typical intracellular and extracellular conc. of Na+ and K+?
Na+ 150mM extracellular, 10mM intracellular
Eq usually +72mV
K+ 5mM extracellular, 150 mM intracellular
Eq usually -90mV
What is depolarisation?
membrane potential becomes more positive → 0mV
What is repolarisation?
membrane potential becomes more negative (towards RMP)
What is overshoot?
membrane potential increases above 0mV
What is hyperpolarisation?
membrane potential decreases below RMP
what are graded potentials?
change in membrane potential dependent on strength of stimulus
many small stimulus, so to produce AP must reach threshold
What happens to graded potentials over time?
they decrease with distance
why do graded potentials decrease over distance
charge leaks out ion channels in membrane
cytoplasm and membrane offer resistance so signal weakens
What happens if the graded potential reaches a threshold?
triggers action potential
What is the difference between an action potential and a graded potential?
graded potentials → variable in size, can summate decay with distance
action potentials → fixed size, no decay, all-or-nothing events
Where does an AP occur?
in excitable cells
state 3 examples of excitable cells:
neurones
muscles cells
some endocrine tissue
What is the purpose of APs? (3)
transmission of information reliably and quickly over long distances
cell-cell communication
activates intracellular processes
what does permeability depend on?
conformational state of ion channels
What is the conformational state of ion channels at depolarisation?
VGSCs open
What is the conformational state of ion channels at sustained depolarisation?
VGSCs inactivated, VGKCs open
What is the conformational state of ion channels at hyperpolarisation/ repolarisation?
VGSCs closed, VGKCs open
Name the 5 phases of the AP
RMP
depolarisation
upstroke (after -55mV threshold is reached)
repolarisation
hyperpolarisation
describe the relation between permeability and RMP stage
membrane more permeable to potassium than sodium ions due to leak channels
Na+/K+/ATPase maintains gradients
-70mV
membrane potential closer to equilibrium of potassium (-90mV) than sodium (+72mV)
describe the relation between permeability and depolarisation stage
membrane becomes more permeable to sodium than potassium as VGSCs open
causes sodium influx
depolarisation occurs
describe the relationship between permeability and the upstroke phase:
reaches threshold of -55mV
PNa increases bc VGSCs open quickly→ Na+ influx
PK increases bc VGKCs open slowly → K+ efflux
less K+ efflux than Na+ influx
membrane potential → Na+ eqm potential (+72mV)
describe the relationship between permeability and repolarisation phase:
PNa decreases due to sustained depolarisation
VGSCs inactivated so no further influx of Na+
PK increases as more VGKCs open and remain open so K+ efflux
membrane potential → K+ eqm potential
Describe the hyperpolarisation phase.
VGKCs are initially open so K+ leaves the cell
membrane potential moves closer to K+ eqm
VGKCs then close
membrane potential returns to RMP
What is the purpose of the refractory period?
prevents neurone being re-stimulated immediately to allow for unidirectionality and limiting frequency
state 2 types of refractory period:
absolute
relative
describe the absolute refractory period :
all VGSCs inactivated
no AP triggered regardless of stimulus strength
describe the relative refractory period:
some VGSCs have returned to resting state
many VGKCs open and membrane hyperpolarised
AP only triggered if stronger-than-normal stimulus reaches threshold
what happens during the absolute refractory period:
Na activation gate is open
Na inactivation gate is closed (Na can’t come in)
eventually both gates become closed as this period continues (activation becomes closed due to repolarisation)
no new AP can be triggered even with very strong stimulus
what happens during the relative refractory period:
Na+ channels have recovered
inactivation gate becomes open
AP may be triggered if stimulus is stronger than normal
Describe two factors that affect the conduction velocity/propagation distance of an AP.
axon diameter: larger diameter → increase in AP velocity as less resistance and depolarisation spreads quicker
myelination: increased myelination → increase in AP velocity
Example of a common neurological condition that results from reduced myelination.
Multiple Sclerosis - degeneration of myelin sheath, nerve starts to lose its myelin = Slower nerve impulses → saltatory conduction not as effective
How are APs propagated across an axon by saltatory conduction?
threshold reached so VGSCs open
depolarisation
AP moves further along axon
Na+ sense the decay (resets)
AP activated in another region of the axon
What does saltatory conduction ensure?
Prevents AP spread
Increases resistance
Decreases capacitance
What is pharmacology?
the study of a chemical substance that interacts with a specific target within a biological system to produce a physiological effect
What are the three questions that we can ask to assess how drugs have an effect on hosts?
Where is the effect produced?
What is the target for the drug?
What is the response produced after interaction with this target?
where is the brain is the effect of heroin produced?
peri-aqueductal grey region (analgesia)
ventral segmental area (euphoria)
solitary nucleus (cough suppression)
What are the responses to heroin?
euphoria
analgesia (painkiller)
cough suppression
What is the target (receptors) for heroin?
Opioid receptors in the brain (exists naturally to interact with endorphins)
What is the main goal of pharmacology companies when producing drugs?
To produce a drug that looks very similar to an endogenous compound, but has stronger effects.
What are the four main drug target classes?
Receptors
Enzymes
Transport proteins
Ion channels.