PHSYL 372 Motor Disorders
1/77
Earn XP
Description and Tags
imported from hannah-mukuhi on quizlet
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
What is myasthenia gravis (MG)
immune system attacks AChR receptor of neuromuscular junctions with antibodies
Cause of MG
- unknown
- may be bacterial or viral infection
What is a distinctive symptom of Myasthenia Gravis (MG) that involves the eyelid?
Ptosis: drooping of eyelid due to ocular muscles being affected
What is a distinctive symptom of Myasthenia Gravis (MG) that affects the mouth?
Mouth hanging
What is a distinctive facial expression associated with Myasthenia Gravis (MG)?
Snarling smile
What respiratory symptom can occur in Myasthenia Gravis (MG)?
Dyspnea due to weakness of respiratory muscles
Treatment of MG
- remove thymus
- immunosuppressants
- anticholinesterase drugs
Why is it thought that MG is caused by bacteria or viral infection
herpes simplex has same sequence as Ach receptor
Immune response that causes MG
- T and B cells destroy herpes virus
- mistake Ach receptor as herpes
- begin autoimmune attack
Which muscles' Ach receptors are affected first
Muscles that control eye lids
Role of MG antibodies in endocytosis
Ach receptors that are bound to antibodies have a higher endocytosis rate than those not bound
How was it determined that MG was an autoimmune disease
- injected antigen of Ach receptor from fish into mouse in hopes that the mouse immune system will develop antibodies for it
- the antibodies will be later tagged with fluorescent molecules and visualized
- however fish receptor was close enough to mouse that mouse antibodies started attacking its own receptors
What type of antibody is rituximab?
Designer monoclonal antibody
What does rituximab target in the treatment of MG?
B cells in intermediate stages
How does rituximab help in Myasthenia Gravis (MG)?
It helps to stop the attack on acetylcholine receptors.
Which types of B cells does rituximab not target?
Immature or plasma B cells
What should patients be cautious about while on rituximab?
They should be careful not to catch new infections.
What does a neuromuscular junction look like?
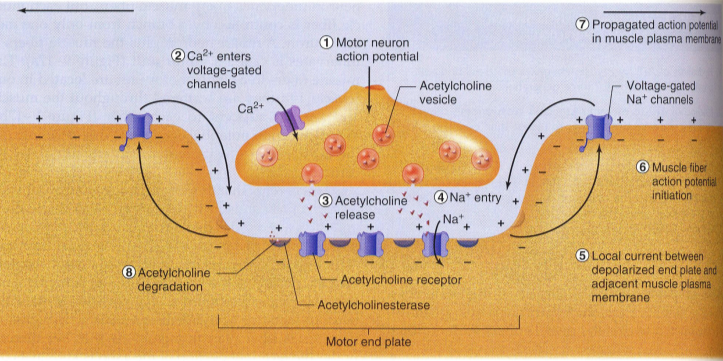
What is neostigmine
anti-cholinesterase drug that reduces ACh-esterase activity preventing breakdown of Ach
Why does neostigmine mechanism of action work
Ach levels are elevated therefore higher probability of binding untargeted Ach receptors
Why is neostigmine useful as medicine?
It is reversible and needs to be taken daily
What is multiple sclerosis (MS)
immune system attacks myelin and associated oligodendrocytes, in late stages axons with no myelin die
Cause of MS
- unknown
- only people with epstein-Barr virus get MS
Distinctive symptoms of MS
scars near vessels in BBB causing breakdown and invasion of B and T cells
Cognition affected in 50% of MS patients
Stages of MS
1. gradual decline in myelination and increase in deficits
2. symptoms flare up with periods of relief in between
Treatment of MS
- corticosteroid to suppress immune system
- rituximab
Consequence of demyelination in neurons
conduction of AP is impaired
How does current regulation work in myelinated neurons?
Current is kept from one node of Ranvier to the next.
What happens to current in demyelinated regions of neurons?
The current is lost through the membrane.
What happens after years of demyelination
axons upregulate Na channels however cannot cope with increased metabolic load and die
What is the animal model of MS?
Inject myelin proteins or CNS tissue together with agents that stimulate the immune system
Treatment: Immunosuppressants, but not effective in halting disease
What is Guillain-Barre syndrome
immune system attacks Schwann cell myelin in PNS via polyclonal antibodies
Symptom of Guillain-Barre syndrome
- motor and sensory neuropathy
- can lead to paralysis depending on severity
progresses from distal extremities first (unlike MG)
How does Guillain-Barre syndrome progress in body
- affects distal extremities first and other muscles later
- longer axons are more vulnerable
Cause of Guillain-Barre syndrome
follows EBV or mononucleosis infection
Treatment of Guillain-Barre syndrome
- suppress immune system
- intravenous rituximab
- support breathing
- disease often resolves by itself
Consequence of myelin sheath loss in Guillain-Barre syndrome
impairs nerve transmission
What happens after Guillain-Barre syndrome is resolved
myelin and nerve damage is spontaneously repaired
What is a motor unit
A motor neuron and all of the muscle fibers it innervates
Motor neurons are huge so they use lots of ATP
Use sustained Ca currents and Ca activated K currents to slowly pace firing to match muscle properties
Poor Ca buffering
Types of motor units
- eye muscle: 1:1 muscle/nerve ratio
- biceps: 750:1 muscle/nerve ratio
What is amyotrophic lateral sclerosis (ALS)
motoneuron death
widespread mitochondrial damage
Cause of ALS
uncertain
possibilities:
toxins
trauma
oxidative tress and SOD1 mutation
calcium exitotxicity
Symptoms of ALS
- muscle weakness
- spasms
- paralysis
frontotemporal dementia
Distinctive characteristic of ALS
mislocalization TDP 43 from nucleus to cytoplasm
Types of ALS
- sporadic
- familial
Role of TDP 43
- transcriptional repression
- RNA splicing
- mRNA transport
- microRNA maturation
What happens when TDP 43 migrates to cytoplasm
disrupts RNA processing and leads to cell death
Function of SOD1 enzyme
protects mitochondrial cells from ROS species
What does SOD1 mutation lead to
leads to SOD1 aggregates, oxidative stress and mitochondrial damage
Relationship of SOD1 with ALS
single mutated SOD1 gene or enzyme dysfunction causes ALS
What happens to Ca in ALS
ALS causes inadequate Ca buffering in large neurons
How does ALS lead to Ca toxicity
Ca channels and glutamate elevate Ca currents leading to high intracellular Ca which causes ROS development leading to excitotoxicity
What are some possible origins of sporadic ALS? (insane card, probably don’t worry)
Toxic event triggers excess glutamate receptor activity, and Ca toxicity via NMDA and AMPA receptors, and Ca channels
Poor fast Ca buffering (like parvalbumin) in motoneurons leads to toxic damage to mitochondria, which take up excess Ca, and this leads to release of reactive oxidative species (ROS, O2-) and lack of ATP, and eventually apoptosis via mPTP and caspase
Excess Ca also activates calpain indiscriminately, which are the ‘scissors’ of cells, chopping proteins. This damages channels, transporters, glutamate uptake, all of which amplify Ca toxicity
ADAR2 that edits RNA is also damaged, leading to more constitutively active AMPA and Ca channels
Proteins like TDP43 get mislocalized to cytoplasm because of damage to nuclear transport and protein tagging, due to ROS and calpain
Lack of ATP prevents proteosomes from pulling apart and destroying misfolded proteins
Misfolded proteins clog up the cytoplasm in clumps, and calpain chops these clumps up into bits that seed other healthy proteins to misfold in prion-like way
TDP43 and SOD1 aggregates (clumps) are released at synapses or via exosomes to spread misfolded proteins to nearby cells, and lead to a prion-like spread of misfolded proteins that eventually causes death
Potential Treatments for ALS
- Riluzole: a sodium channel blocker
- tofersen: antisense oligonucleotide of SOD1 mRNA reducing SOD1 protein synthesis
What is hSOD1?
Potential cure for familial ALS that involves viral injections that block mutant SOD1 or TDP43 gene actions
What type of disease is spinal muscular atrophy (SMA)
autosomal recessive disease
What causes SMA
genetic defect in SMN1 gene → loss of function mutation
Function of SMN1
development of motoneurons
What does genetic defect of SMN1 lead to
diminished abundance of proteins resulting in death of motoneurons in ventral horn
What is the most common cause of infant death?
SMA
Symptoms of SMA
- weakened muscle tone in limb and trunk
- respiratory problems leading to impaired breathing
- scoliosis
- difficulty walking and sitting due to feeble movement of arm and legs
- swallowing difficulties due to weak sucking reflex
5 physical symptoms to diagnose SMA
– poor muscle tone in the limbs and trunk
– feeble movements of the arms and legs
– swallowing difficulties
– a weak sucking reflex
– impaired breathing
Describe type 1 SMA
- called werdnig-hoffman disease
- most severe
- children never move or breathe independently
60% of all SMA patients
Describe type 2 SMA
- resp and feeding not as severe as type 1
- can sit but not walk
- can live into adulthood
- happens before 18 months
Describe type 3 SMA
- called kugelber-welander or juvenile SMA
- happens between 18 months and adulthood
- difficulty walking, muscle weakness, prone to infections
- can live into adulthood
Describe type 4 SMA
- adult form - less common
- affect walking
- symptoms emerge after 35
What decreases severity of SMA
if backup genes like SMN2 is present
Drawback of SMN2 gene
makes truncated ineffective SMN
How to make SMN2 more effective
- use antisense oligonucleotides to alter pre-mRNA splicing to make full length SMN proteins
- use virus to deliver replacement of SMN1 gene
Example of SMN2 antisense oligonucleotides
Nusinersen
What is Duchenne muscular dystrophy
- muscle wasting
- defect in dystrophin gene on X chromosome that causes loss of function
Cause of duchenne muscular dystrophy
Genetic origin → defect in dystrophin gene (loss of function)
Function of dystrophin
creates link between contractile machinery and extracellular matrix
Symptoms of Duchenne Muscular Dystrophy
- child does not run
- pseudohypertrophy
- in wheel chair by 12
no treatment
How to detect Duschenne Muscular Dystrophy
genetic studies performed during pregnancy
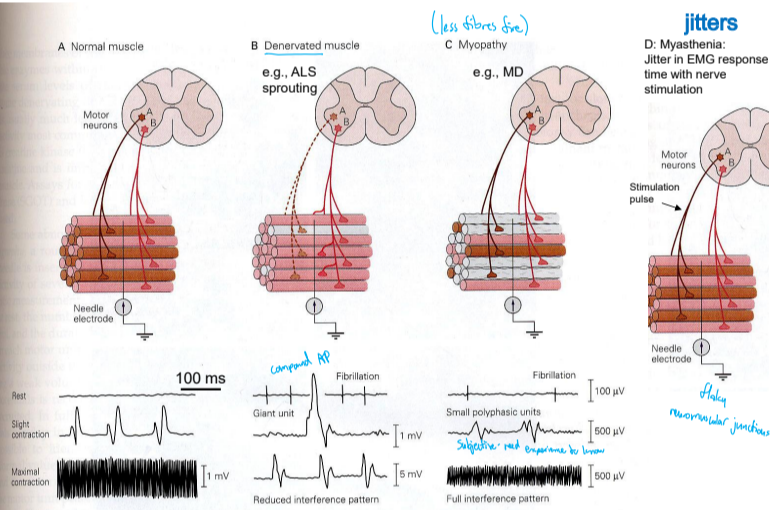
What is EMG
extracellular recording of action potentials travelling down muscle fibres
EMG diagnostic criteria for difference diseases
What caused increased incidence of ALS among native Guamanians
eating bats infested with BMAA toxin that was created by cyanobacteria in cycad seeds