01 - Introduction & Basics
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
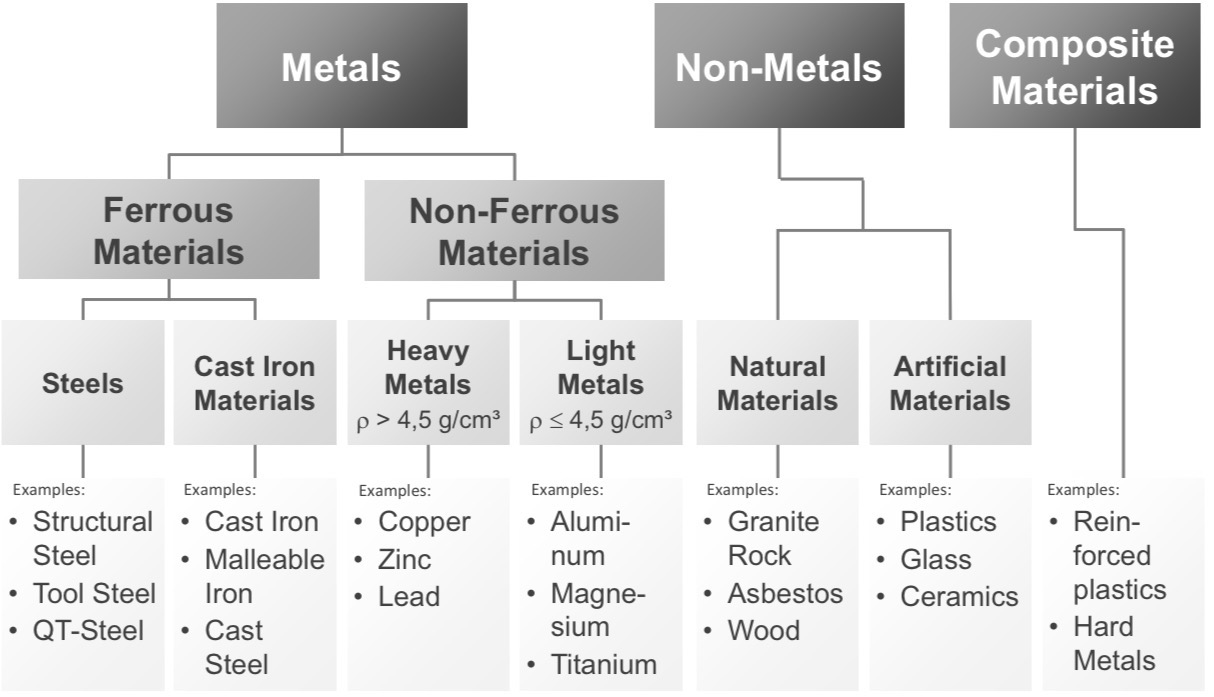
Classify the materials into material groups and give examples
What’s the difference between ferrous and non-ferrous materials?
ferrous materials: contain iron Fe as main element
Non-ferrous: can contain iron Fe but not as the primary element
What’s the density limit between light & heavy metals?
4.5 g/cm3
What are the most important ferrous materials?
Iron & steel (fer & acier)
What’s the definition of cast iron?
Group of iron alloys with high proportion of carbon (>2%) and silicon (1.5%)
What’s steel? (Acier)
Iron-carbon alloy that contains less that 2.06% of carbon (by mass)
What are the properties of metals?
high electrical conductivity
High thermal conductivity
Ductility (formability)
Metallic gloss (mirror finish)
For what reasons do we use non-ferrous metals?
corrosion resistance
High-temperature strength
Scaling resistance (entartrage? Couche d’oxyde)
Low density (light)
Electrical properties
What are the reasons metals have their particular properties?
cohesion of atoms by metallic bonds (atoms held together, electrons shared among many atoms)
Periodic arrangement of the atoms (crystal lattice)
electrons moving freely in the lattice —> electrical & thermal conductivity
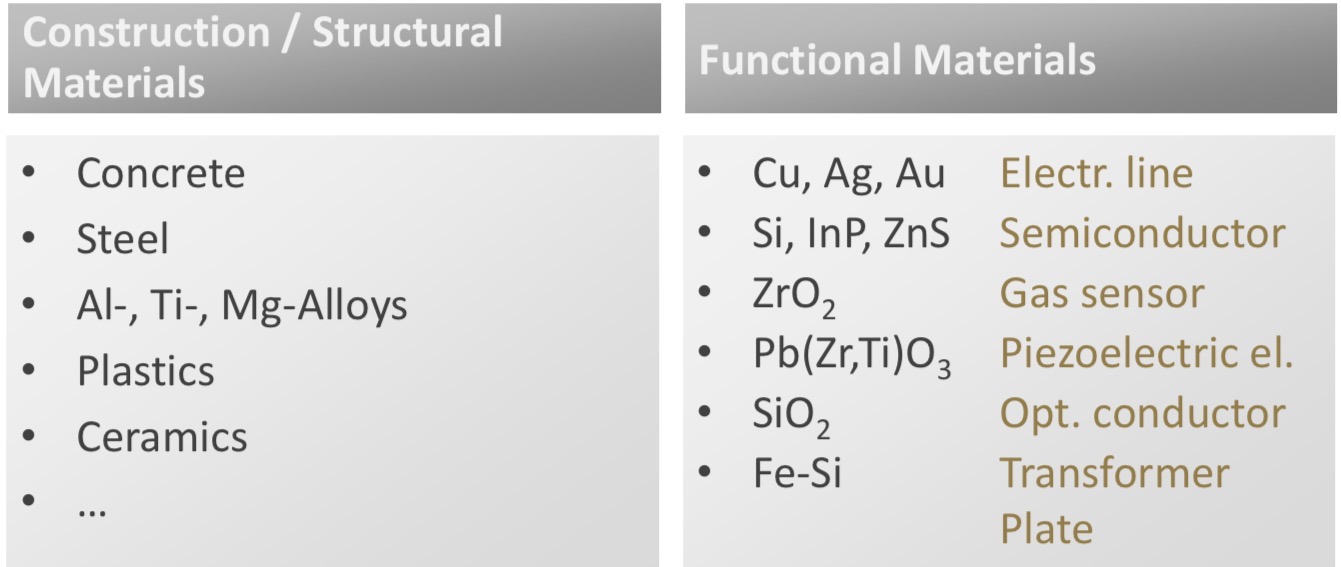
Classify materials according to their use & explain what are the different categories
Construction/structural materials: used for their structural properties, buildings & constructions
Functional materials: respond to external stimuli (light, heat, electricity…)
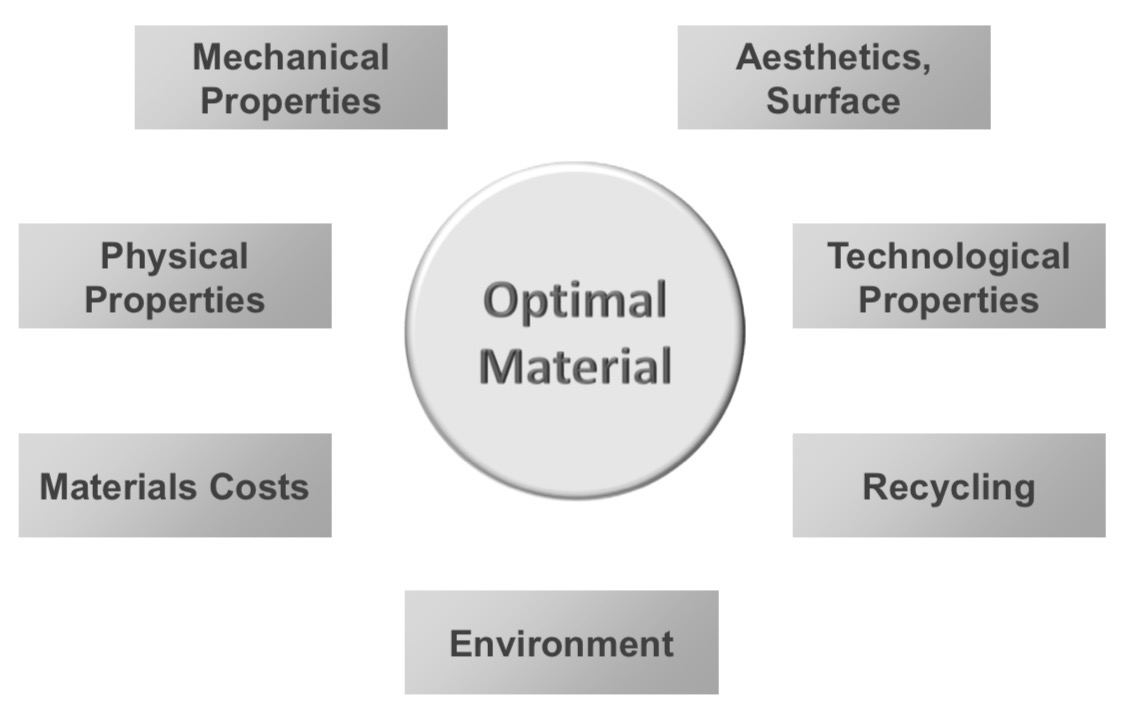
What can we say about “The optimal material”?
THE optimal material doesn’t exist, but there is a material that is optimal for a specific application
What are the different factors to consider when selecting a material?
Why do materials (in general) have different properties?
Properties depend on:
the type of atomic bonds
The atoms arrangement
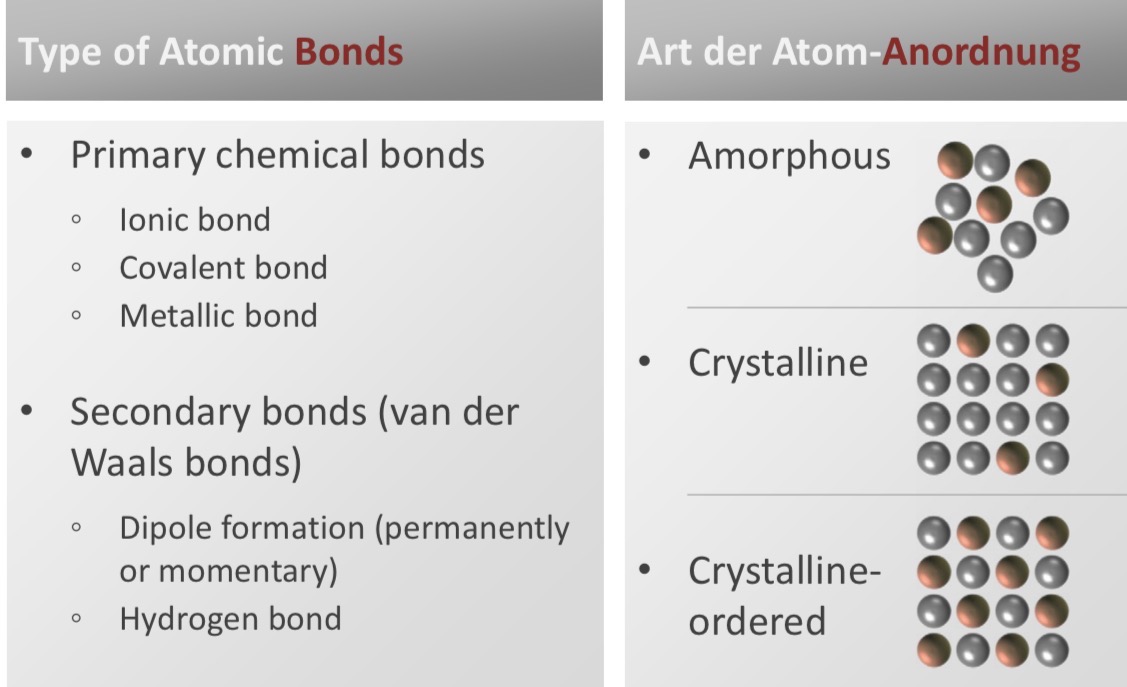
What are the 2 categories of atomic bonds? What types of atomic bonds are there in each?
primary chemical bonds
ionic bonds (e- are transferred)
Covalent bonds (e- are shared)
Metallic bonds (e- are delocalised, free to move)
Secondary bonds (van der Waals)
Dipole formation
Hydrogen bonds
What are the 3 different types of arrangement of the atoms/molecules?
amorphous: randomly arranged, without a repeating pattern
Crystalline: arranged in repeating, periodic pattern (crystal lattice)
Crystalline ordered: same as crystalline + the individual crystalline grains are aligned in a specific orientation
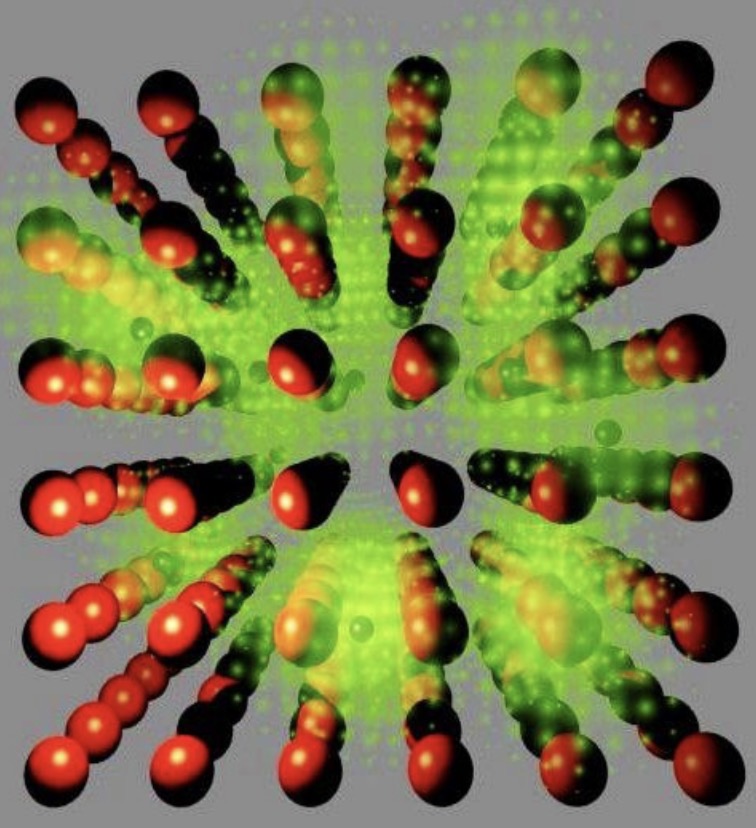
Describe the metallic bond
occurs between metal atoms
All atoms “share” e- that are delocalised and free to move (electron gas model/ sea of e-)
No preferred direction in the bonding
Metallic crystals are closely packed (maximises the #of atoms that can fit into a given space)
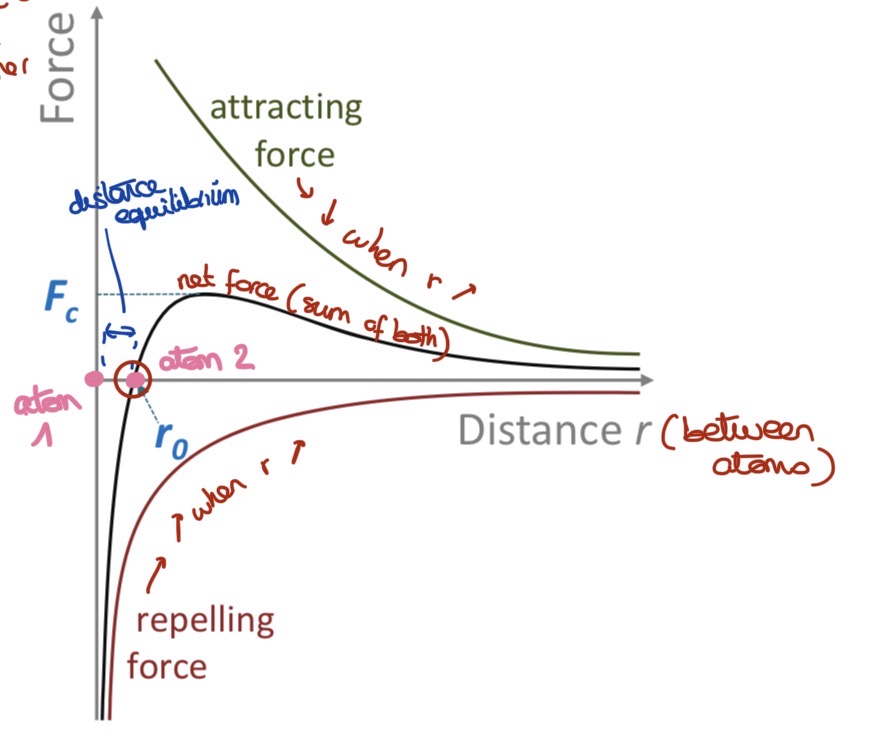
Describe the behavior of atoms according to the bonding forces & the distance between atoms
When atoms bond, their e- rearrange to lower the overall energy of the system & become more stable
The nucleus of the atoms repel each other (+/+) and electrons as well (-/-) —> repulsive forces
Nucleus and e- attract each others —> attractive forces
At equilibrium distance r0, attractive & repulsive forces balance/cancel out & and the system is stable (net force = 0)
What’s the formula of the bonding energy?
E,be = E,isolated - E,bonded
Difference between the energy of an isolated atom, and its energy when bonded
What are the different scales of metals (name & value)?
Macroscopic scale (cm)
Microscopic scale
Grain scale (micrometer)
Atom scale (nanometer)
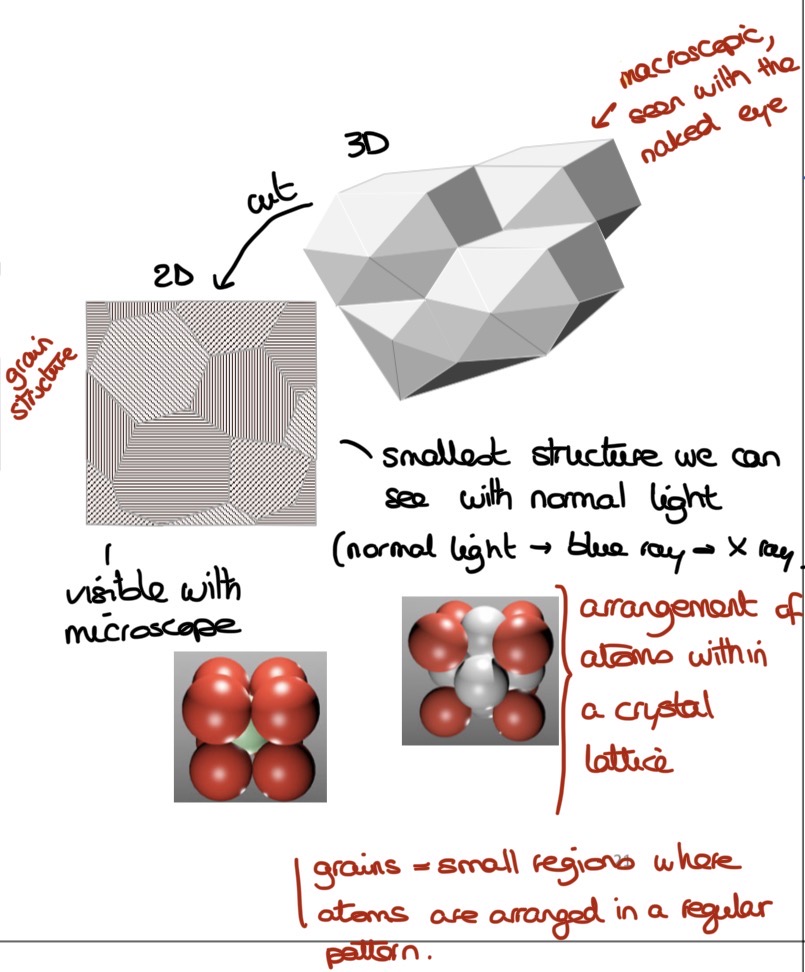
What’s a crystal lattice & how does it form?
Regular, repeating arrangement of atoms in 3D space
Formed when attractive & repulsive forces balance (equilibrium position of atoms)
What’s the unit cell of a crystal lattice?
Smallest repeating unit that creates the lattice when repeated in 3 directions.
Fundamental “building block” of lattice
defines the geometry and symmetry of the lattice
ALWAYS in 3D
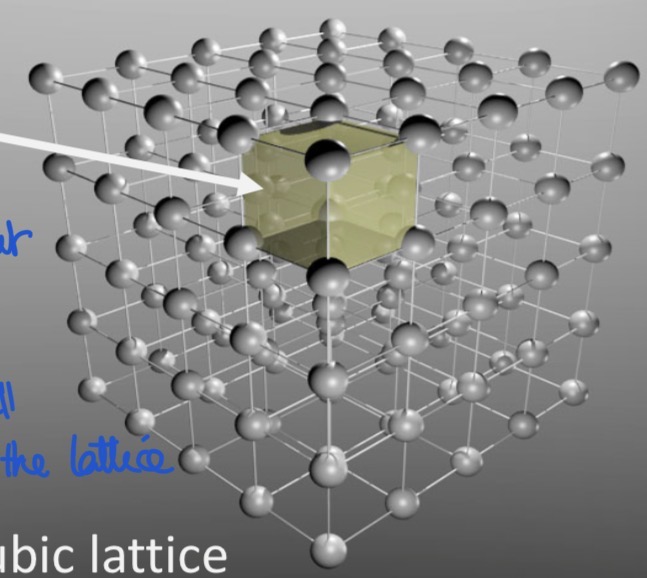
What are the 4 most common cubic lattices?
Which ones are closest sphere packing models?
primitive cubic lattice
Face centered cubic lattice (fcc)
Body centered cubic lattice (bcc)
Hexagonal close packing (hcp)
Closest sphere packing models: fcc, bcc, hcp (atoms are the closest possible)
What’s the problem with the primitive cubic lattice?
Atoms are not optimally arranged
They can easily move (with a small force) to a more stable position
What’s the advantage of a stable arrangement?
The height (vertical distance between atoms layers) is reduced
Improved packing density
Closer = stronger bonds = more stable
What’s an octahedral plane?
Plane where atoms are arranged the most closely together (maximises contact between atoms)
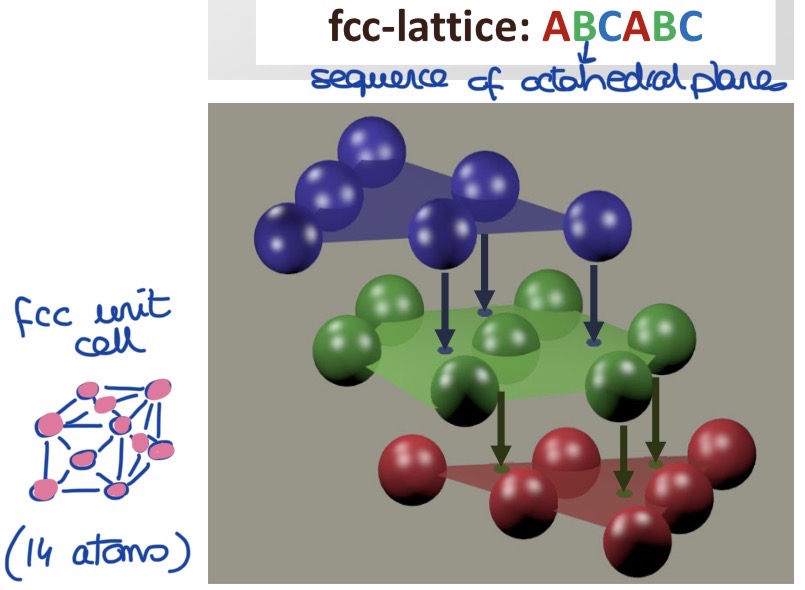
Draw a face centered cubic unit cell.
What proportion of the volume is occupied by atoms?
What’s the sequence of octahedral planes?
74%
(highest packing density for equally sized spheres)
Sequence: ABC ABC ABC
(each new layer fits into the gals of the previous layer but never in the same gaps as the previous or next layer)
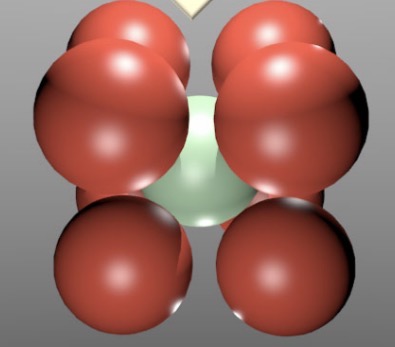
Draw a body centered cubic unit cell.
What’s the packing density? (Volume occupied by atoms)
68% packing density
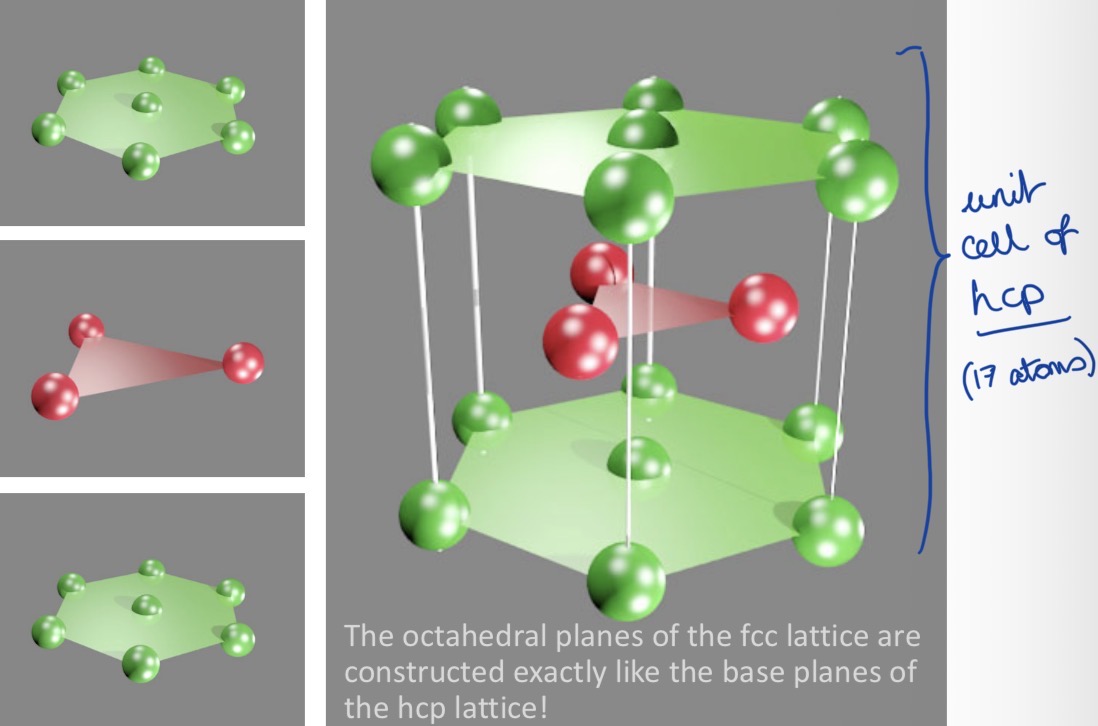
Draw a hexagonal close packing unit cell.
What’s the packing density?
What’s the stacking sequence of octahedral planes?
74%
(highest packing density for equally sized spheres)
AB AB AB
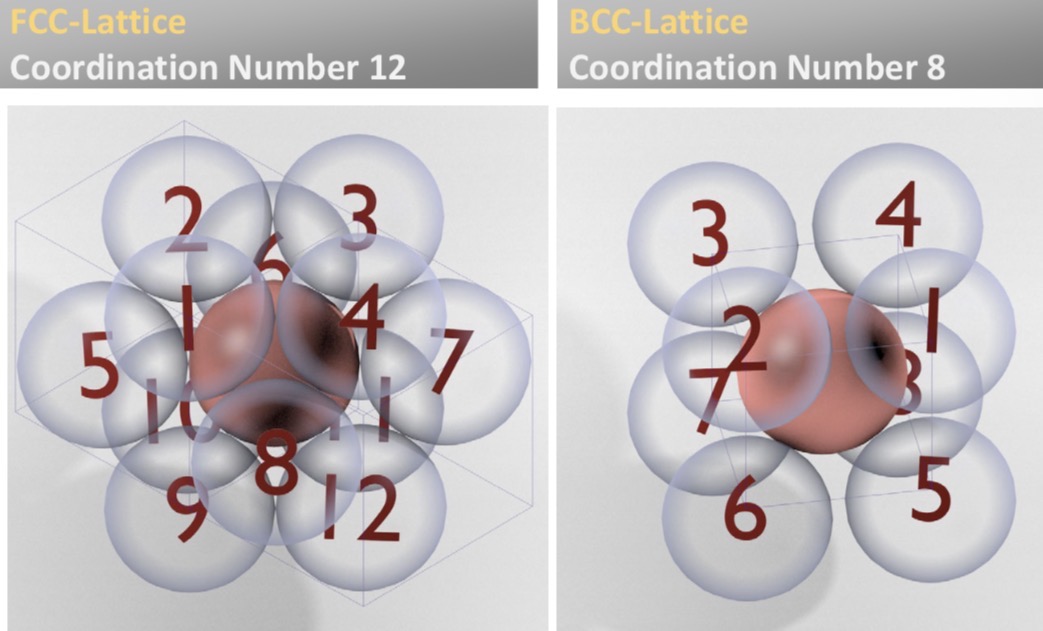
What’s the coordination number?
What is it for fcc? Bcc?
Number of nearest neighbor atoms surrounding a central atom in a crystal lattice
fcc: 12
bcc: 8