biochemistry final (new material)
1/140
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
141 Terms
describe the differences between fatty acid catabolism and anabolism
catabolism
produces acetyl-CoA
produces reducing power (NADH, FADH2)
takes place in the mitochondria
anabolism
requires acetyl-CoA and malonyl-CoA
requires reducing power from NADPH
takes place where NADPH levels are high (PPP) - in cytosol
where does anabolism of fatty acids occur
cytosol
NADPH levels are high from PPP
why does anabolism of fatty acids require NADPH instead of NADH
because fatty acid synthesis takes place in the cytosol
[NADPH] higher in cytosol
[NADH] low in cytosol, high in mitochondria
fatty acids are built in several passes, processing one acetate unit at a time. where does each acetate unit come from?
acetate comes from activated malonate in the form of malonyl-CoA
each pass involves reduction of a carbonyl carbon to a methylene carbon
describe the general formation reaction for malonyl-CoA
acetyl CoA + HCO3- + ATP → malonyl-CoA + ADP + Pi
enzyme: acetyl CoA carboxylase
cofactor: biotin
what two enzymes are similar to acetyl-CoA carboxylase?
acetyl CoA carboxylase uses a two step reaction similar to carboxylations catalyzed by
pyruvate carboxylase
gluconeogenesis
propionyl CoA carboxylase
odd chain FA break down
describe the mechanism of the acetyl-CoA carboxylase reaction
use energy from ATP to add CO2 to biotin in the biotin carboxylase site
CO2 is activated by attachment to N in ring of biotin
enzyme undergoes conformational change to carry CO2 to transcarboxylase site
swinging arm
CO2 attaches to acetyl CoA and leaves active site
malonyl CoA is produced
what is the overall goal of fatty acid synthesis
attach acetate unit (2 carbon) from malonyl-CoA to a growing chain and then reduce it
what are the four steps of FA synthesis
condensation
reduction
dehydration
reduction
describe step 1 of fatty acid synthesis
condensation of the growing chain with activated acetate
make a C-C bond and release CO2
describe step 2 of fatty acid synthesis
reduction of carbonyl to hydroxyl
need oxidizing agent → uses NADPH, form NADP+
describe step 3 of fatty acid synthesis
dehydration of alcohol to trans-alkene
remove H2O from trans double bond between 𝛼 and 𝛽 carbons
describe step 4 of fatty acid synthesis
reduction of alkene to alkane
reduce db again using NADPH
move lengthened acyl chain to KS so that malonyl CoA can bind ACP
how is the growing chain attached to the fatty acid synthase?
the growing chain is initially attached to the enzyme via a thioester linkage
what enzyme catalyzes fatty acid synthesis
fatty acid synthase (FAS)
what is the function of acyl carrier protein (ACP)
ACP serves as a shuttle in fatty acid synthesis. binds malonyl CoA
contains a covalently attached prosthetic group
flexible arm to tether acyl chain while carrying intermediates from one enzyme subunit to the next
describe the structure of the ACP prosthetic group
prosthetic group contains pantothenic acid and an -SH group at the end where malonyl groups can bind
describe the function of the KS domain of FAS
make C-C bond, condensation
acetyl-CoA/growing chain attaches here
describe the function of the MAT domain of FAS
transferase, transfer growing chain to KS
describe the function of the DH domain of FAS
dehydration
describe the function of the ER domain of FAS
reduction (reductase)
describe the function of the KR domain of FAS
reduction (reductase)
write out the stoichiometry of synthesis of palmitate (16:0)
7 acetyl-CoAs are carboxylated to make 7 malonyl-CoAs
7 acetyl-CoA + 7 CO2 + 7 ATP → 7 malonyl-CoA + 7 ADP + 7 Pi
seven cycles of condensation, reduction, dehydration, and reduction
acetyl-CoA + 7 malonyl-CoA + 14 NADPH + 14H+ → palmitate + 7 CO2 + 8 CoA + 14 NADP+ + 7 H2O
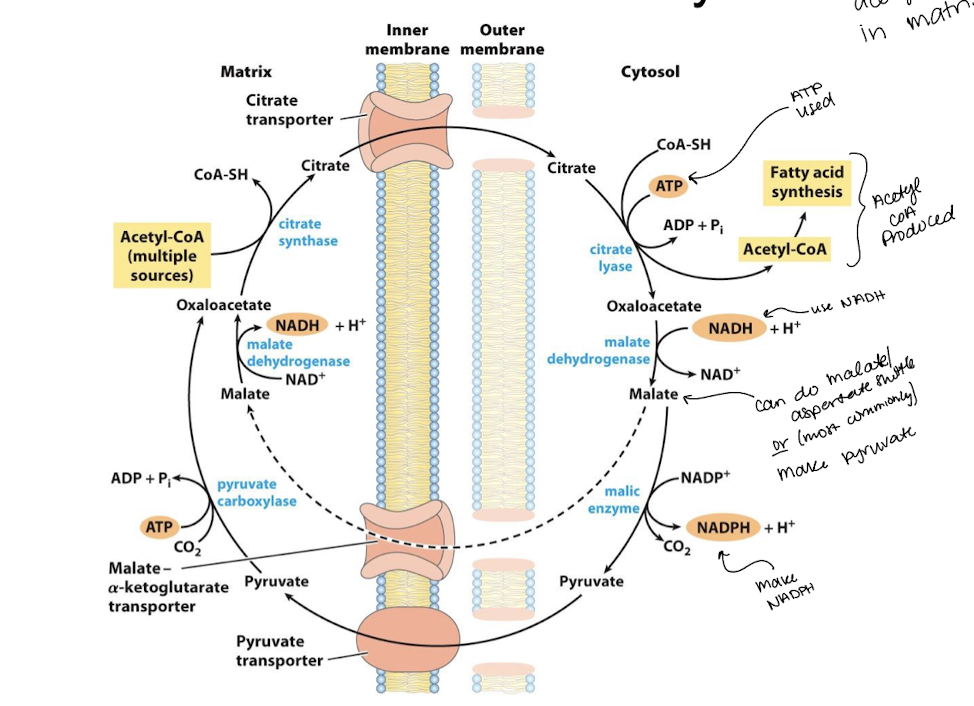
why do acetyl groups need a shuttle?
acetyl groups are synthesized in the mitochondrial matrix. they are needed for fatty acid synthesis in the cytosol
describe the two shuttle systems that acetyl groups can use to get from mitochondria to cytosol
use citrate transporter
citrate + CoASH + ATP → oxaloacetate + ADP + P + acetyl CoA
acetyl-CoA goes to FA synthesis
oxaloacetate + NADH + H → malate + NAD+
malate can do malate-aspartate shuttle BUT most commonly it makes pyruvate
malate + NADP+ → NADPH + H + CO2 + pyruvate
what enzyme is regulated in fatty acid synthesis
acetyl-CoA carboxylase (ACC)
ACC catalyzes the rate limiting step
how is the ACC regulated?
allosterically regulated
feedback-inhibited by palmitoyl-CoA
activated by citrate
regulated by covalent modification
inhibited when energy is needed
glucagon and epinephrine
how do glucagon and epinephrine regulate fatty acid synthesis?
low energy → do not consume lipids, phosphorylation cascade
lead to phosphorylation and inactivation of ACC
ACC is inactive as phosphorylated monomers
phosphorylate PKA to activate glycolysis
when dephosphorylated, ACC polymerizes into long, active filaments
forms large active complex with lots of polymers
describe how fatty acid synthesis is regulated by phosphorylation
glucagon and epinephrine trigger phosphorylation
phosphorylate ACC to inhibit fatty acid synthesis
phosphorylate PKA to activate glycolysis
how does citrate regulate fatty acid synthesis
ACC is activated by citrate
used citrate to shuttle acetyl CoA
citrate in cytosol means high energy in cell to make fats
how does palmitoyl-CoA regulate fatty acid synthesis
feedback-inhibited by palmitoyl-CoA
downstream inhibitor
describe reciprocal regulation of fatty acid metabolism using insulin
high blood sugar, increase insulin
dephosphorylate ACC to activate
perform FA synthesis, synthesize malonyl CoA
malonyl CoA inhibits fatty acid import into mitochondria
FA cannot be broken down bc they are being made
one of many ways to ensure that fat synthesis and oxidation don’t occur simultaneously
describe FA metabolism when blood sugar is low
low BS, high glucagon
ACC is phosphorylated and inactive
low malonyl CoA allows entry of FA into matrix for break down
how do we store fats for energy
as triacylglycerols
what two molecules contain a glycerol backbone
TAGs and phospholipids
describe the structure of a TAG
3 fatty acids attached to a glycerol backbone
describe the structure of a phospholipid
2 fatty acid and a phosphate group attached to a glycerol backbone
what is the first step in making TAGs and phospholipids
make glycerol-3-phosphate first
why does triacylglycerol and phospholipid synthesis start the same way?
making glycerol-3-phosphate first tags it for TAG or phospholipid synthesis
describe the synthesis of the backbone of TAGs and phospholipids
most glycerol-3-phosphate comes from siphoning off dihydroxyacetone phosphate from glycolysis
via glycerol-3-phosphate dehydrogenase (same enzyme from glycerol-3-P shuttle)
use NADH
some glycerol-3-phosphate is made from glycerol
via glycerol kinase
use ATP
what is the precursor to TAGs and phospholipids
phosphatidic acid
what is the advantage of making phosphatidic acid
can be made into triacylglycerol or phospholipid
how is phosphatidic acid made
start with glycerol-3-phosphate
2 fatty acids attached by acyl transferases
releases CoA
describe how phosphatidic acid can be modified to form TAGs
phosphatidic acid phosphatase (lipin) removes the 3-phosphate from the phosphatidic acid
yields 1,2-diacylglycerol (DAG)
the third carbon is then acetylated with a third fatty acid
uses acyl transferase
yields triacylglycerol
describe how phosphatidic acid can be modified to form phospholipids
attachment of head group (serine, choline, ethanolamine, etc.) to the phosphate group. yields glycerophospholipid
describe the steps of triacylglycerol synthesis
DHAP + NADH + H+ → glycerol-3-phosphate + NAD+
glycerol-3-phosphate dehydrogenase
glycerol-3-phosphate + 2 fatty acids → phosphatidic acid + 2 CoASH
acyl transferase
phosphatidic acid → diacylglycerol
phosphatidic acid phosphatase
diacylglycerol + fatty acid → triacylglycerol + CoASH
acyl transferase
describe the steps of phospholipid synthesis
DHAP + NADH + H+ → glycerol-3-phosphate + NAD+
glycerol-3-phosphate dehydrogenase
glycerol-3-phosphate + 2 fatty acids → phosphatidic acid + 2 CoASH
acyl transferase
phosphatidic acid + head group → glycerophospholipid
how does insulin regulate triacylglycerol synthesis
insulin results in stimulation of triacylglycerol synthesis
promotes synthesis is acetyl CoA and fatty acids
what does lack of insulin result in (lipid biosynthesis chapter)?
increased lipolysis
increased fatty acid oxidation
sometimes to ketones if citric acid cycle intermediates (OAA) that react with acetyl CoA are depleted
failure to synthesize fatty acids
what is an eicosanoid?
potent, short-range hormones made from arachidonate
prostaglandins, leukotrienes, thromboxanes
cause inflammation, pain, increased body temp
released in response to injury
what are eicosaniods made from
made from arachidonate in the phospholipids of membranes
what enzyme is activated to release arachidonate
phospholipase A2
what is PGH2 synthase
a cyclooxygenase/peroxidase (COX) enzyme
describe eicosanoid synthesis
phospholipid containing arachidonate → arachidonate + phospholipid
enzyme: phospholipase A2
arachidonate + 2O2 → PGG2
cyclooxygenase activity of COX
PGG2 → PGH2
peroxidase activity of COX
PGH2 → prostaglandins/thromboxanes
what do NSAIDs do
they inhibit cyclooxygenase (COX) activity
how does aspirin work?
aspirin (acetylsalicylate) is an irreversible inhibitor
acetylates a serine in active site
blocks active site in COX enzymes
how do ibuprofen and naproxen work
ibuprofen and naproxen are competitive inhibitors
resemble substrate
blocks active site in COX enzymes
what is cholesterol made from
made strictly from acetate units (starts as acetyl CoA)
provide the general steps for cholesterol synthesis
acetate (2C) → mevalonate (6C) → activated isoprene (5C) → squalene (30C) → cholesterol (27C)
describe the structure and function of mevalonate
composed of 3 acetate molecules
important intermediate in cholesterol synthesis
how are activated isoprenes created? what are they turned into?
reduce carbon on mevalonate and phosphorylate to make activated isoprene (5C)
6 isoprenes are needed to create squalene
what is squalene
the unfused (30C) intermediate of cholesterol. made from 6 isoprene molecules
what enzyme makes mevalonate? what is significant about this enzyme?
HMG-CoA reductase
first committed step of cholesterol synthesis, highly regulated
how do ATP levels regulate HMG-CoA reductase?
low [ATP] → high [AMP] → active AMPK → inhibit HMG-CoA reductase
how do insulin and glucagon regulate HMG-CoA reductase
insulin activates, glucagon inhibits
how does cholesterol regulate HMG-CoA reductase
high cholesterol → oxysterol → proteolysis of HMG-CoA reductase → inhibition
how does cholesterol travel through the body?
cholesterol and other lipids are carried on lipoprotein particles
describe the structure of lipoproteins
surface: protein (called apoliporotein) and phospholipid monolayer
interior: cholesterol, TAGs
rank the four classes of lipoprotein particles based on protein content
lowest: chylomicron
VLDL
LDL
highest: HDL
what does “density” refer to when talking about lipoprotein classes?
density refers to protein content
ex. HDL has more protein than LDL
what is the function of VLDL
transport TAGs to tissues
what is the function of LDL
transport cholesterol to tissues
how is cholesterol transported into tissue cells
apolipoproteins bind to LDL receptor
lipoproteins taken up by endocytosis
internalized apolipoproteins are degraded in lysosomes, while cholesterol is used for biological function
note: number of receptors increase sin response to a need for cholesterol or steroid hormones
can cholesterol be metabolically degraded?
no! cholesterol cannot be metabolically degraded
excess is returned to liver for ultimate excretion as bile to the intestine
what is the function of HDL
can participate in reverse cholesterol transport
pick up cholesterol in extrahepatic tissues and carry to the liver for metabolism or excretion
describe the structure of HDL
more protein than lipid, contain relatively little cholesterol
describe how cardiovascular disease is multifactorial
very high LDL-cholesterol levels tend to correlate with atherosclerosis
although many heart attack victims have normal cholesterol, and many people with high cholesterol do not have heart attacks
low HDL-cholesterol levels are associated with heart disease
what is familial hypercholesterolemia
due to genetic mutation in LDL receptor
impairs receptor-mediated uptake of cholesterol from LDL
cholesterol accumulates in the blood/arteries
rare, homozygous individuals can experience severe CVD/heart attacks as youths
how do statins work?
inhibit HMG-CoA reductase to lower cholesterol synthesis
statins resemble mevalonate → competitive inhibitors of HMG-CoA reductase
how many amino acids can bacteria synthesize
can synthesize all 20
name the nonessential amino acids
glutamate
glutamine
proline
arginine (conditionally essential)
serine
glycine
cysteine
aspartate
asparagine
alanine
tyrosine
name the essential amino acids
valine
histidine
methionine
isoleucine
leucine
lysine
phenylalanine
threonine
tryptophan
what does it mean to be essential?
need to get it from the diet
what is the nitrogen source for amino acid synthesis
glutamine or glutamate
2 most plentiful amino acids
where do the carbon skeletons come from in amino acid synthesis
carbon skeletons derived from intermediates
glycolysis
citric acid cycle
pentose phosphate pathway (bacteria only)
all amino acids in humans derive from one of four precursors. what are these precursors?
citric acid cycle
𝛼-ketoglutarate
oxaloacetate
glycolysis
pyruvate
3-phosphoglycerate
what amino acids are derived from 𝛼-ketoglutarate?
glutamate, glutamine, proline, arginine
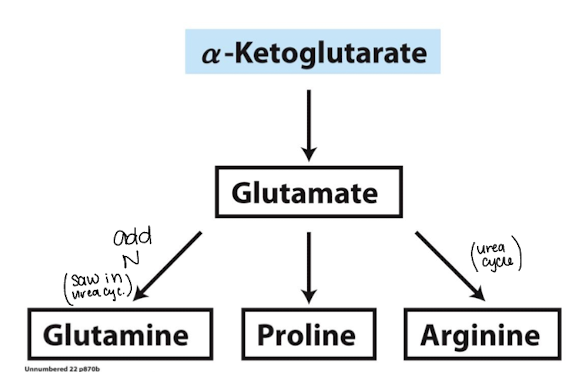
how is glutamate synthesized
glutamate is derived from transamination of 𝛼-ketoglutarate
need transaminase
how is arginine synthesized
synthesized from glutamate (via ornithine) in the urea cycle
add guanidinium to ornithine, get nitrogen from glutamate
how is proline synthesized
proline can be synthesized from glutamate or from arginine
ornithine is derived from glutamate, the urea cycle, or degradation of arginine
ornithine aminotransferase converts ornithine to glutamate semialdehyde that cyclizes and converts to proline
deaminate ornithine to make something similar to glutamate
what amino acids are derived from 3-phosphoglycerate?
serine, glycine, cysteine
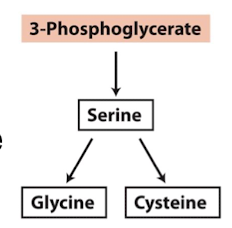
how is serine synthesized
oxidation → transamination → dephosphorylation
3 phosphoglycerate is oxidized
need reducing agent, NAD+
add nitrogen from glutamate
remove phosphate to yield serine
how is glycine synthesized
3-phosphoglycerate → serine → glycine
remove hydroxymethyl from serine to produce glycine
how is cysteine synthesized
3-phosphoglycerate → serine → cysteine
what amino acids are derived from oxaloacetate
aspartate and asparagine
how is aspartate synthesized
aspartate is formed from transamination of oxaloacetate
how is asparagine synthesized
oxaloacetate → asparatate → asparagine
asparagine is formed from transamidation of aspartate
what amino acids are derived from pyruvate
alanine
how is alanine synthesized
transamination of pyruvate forms alanine
pyruvate + glutamate → 𝛼-ketoglutarate + alanine
how does consuming carbs lead to greater alanine production
metabolizing carbs: make pyruvate
then pyruvate → alanine using glutamate as a nitrogen source