Exam 2 O Chem 3 (redone flashcards)
1/92
Earn XP
Description and Tags
Flashcards for exam 2, organic chemistry 3 (5/2/2026)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
result of fisher esterification
carboxylic acid → ester
result of alkylation of carboxylic acids
carboxylic acid → ester
what reagent is used in alkylation of carboxylic acids
diazomethane H2C-N- - -N
result of reacting thionyl chloride with a carboxylic acid
carboxylic acid → acyl chloride
what 2 things are required for synthesis of anhydrides
2 equ carboxylic acid and a dehydrating agent (such as P2O5)
what is needed for synthesis of cyclic anhydrides
only heat because the process is intramolecular
What reagent is used for reduction of carboxylic acids?
LiAlH4
result of carboxylic acid reduction
carboxylic acid → primary alcohol
what is required for decarboxylation to occur?
a ketone at the beta position
what three categories were discussed for decarboxyliation
beta-ketoacids, malonic acids, carbonic acids
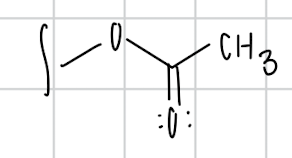
name the substituent
acetoxy
how to name carboxylate salts
name the counter ion first, then name the ion and use the suffix “ate”.
how to name esters
name the substiuent on the carboxylic oxygen first, then name the rest of the chain and use the suffix “oate”
what is a lactone?
a cyclic ester
how to name lactones
use a greek symbol to indicate the position of the ester, then count the number of carbons in the ring and use the suffix “lactone”
how to name acid halides?
name the chain and add the suffix “yl”, then name the halide
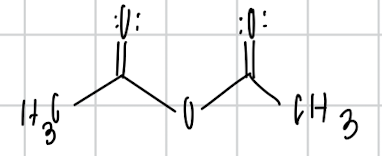
name the compound
acetic anhydride
how to name mixed anhydrides?
name either side of the anhydride (alphabetically) and then anhydride
how to name nitriles?
name the r group, then use the suffix “onitrile”
how to name primary amides
name r group, then add suffic “amide”
how to name secondary amides
use (N) then name the amide substituent, then the r group, then use suffix “amide”
how to name tertiary amides
use (N,N) and name amide substituent groups, then name the R group, then use suffix “amide”
what is a lactam?
a cyclic amide
how to name lactams
use greek letter to indicate position of nitrogen, then count number of carbons in ring and add suffix “lactam”
what functional groups utilize dicarboxylic acid nomenclature?
imides and anhydrides
list the priority of the discussed principal groups
carboxylic acid > anhydride > ester > acyl halide > amide > nitrile > aldehyde > ketone > alcohol > thiol
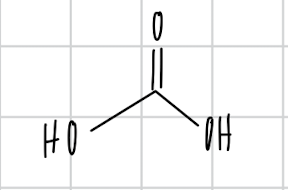
name the compound
carbonic acid
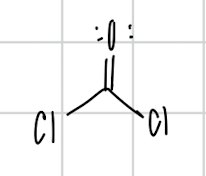
name the compound
phosgene
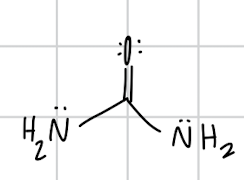
name the compound
urea
key thing to note about amide IR spec
amides exhibit significant C-O single bond character due to resonance stabilization resulting in a weakened bond reading, and therefore a lower IR stretch
orientation of Z-amides
carbonyl oxygen and amide substituents on the same side
orientation of E amides
carbonyl oxygen and amide substituent on opposite sides (steric interaction occurs between substituents)
rank the boiling points of carboxylix acids, amines, esters and ketones
amine > carboxylic acid > ester > ketone
key characteristic of ester IR spectrum
inductive effected strengthen the C=O bond, resulting in a higher than expected IR stretch
are ketones or esters more reactive?
ketones are more reactive than esters
rank the basicity of esters, nitriles, and amides
most basic = amides > ester > nitrile = most acidic
reaction category: carbonyl oxygen
SOCl2 addition
reaction category: carbonyl carbon
acyl substitution
reaction category: reactions are the alpha carbon
halogenation of the alpha carbon
result of saponification of esters
ester → carboxylic acid
what two reactions are the reverse of each other?
fisher esterification and saponification
why is hydrolysis of esters favored under basic conditions (saponification) over acidic conditions
acidic conditions require a strong acid and are very slow
what happens overtime following lactone ring opening?
the ring will collapse again
result of hydrolysis of primary amides
amide → carboxylic acid
result of hydrolysis of secondary amides
amide → aniline
result of nitrile hydrolysis
nitrile → carboxylic acid
requirement for hydrolysis under basic conditions
an additional workup step
stereochemistry of hydrolysis of cyclic anhydrides?
stereochemistry is preserved
reank the reactivity of the discussed functional groups
nitriles < amides < esters/carboxylic acids « anhydrides < acyl chlorides
what explains the reactivity of amides and acid chlorides
resonance structures: amides experience significant C-O single bond character making them less reactive and stabilizing the amide ground state whereas positive charge is poorly stabilized by chlorine in acid chlorides raising the ground state
What is the impact of the stability of the tetrahedral intermediate for amides vs. acyl chlorides
when x = Cl the intermediate is stabilized, when x = N the intermediate is destabilized
relationship between intermediate stability and reaction favorability
the more stable the intermediate, the more favorable a reaction is to occur
result of reacting acid chlorides with a amine
acid chloride → primary amide
why does pyridine act as a catalyst in addition of amines to acyl chlorides but triethyl amine does not
triethyl amine is too bulky to act as a catalyst
what can accelerate the reaction of pyridine addition to acid chlorides?
making the ring electron rich such as DMAP (the N(Me)2 pushes electron density into the ring and can be used as a catalyst)
result of alcohol addition to acid chlorides
acid chloride → ester
result of acid chloride reaction with carboxylate
acid chloride → anhydride
result of anhydride + phenol
anhydride → ester
result of cyclic anhydride + alcohol
cyclic anhydride → acyclic ester
result of anhydride plus amine
anhydride → amide
result of ester plus aniline
ester → amide
result of ester plus alkoxide
ester → ester
result of beta-lactams + alcohol
ring opening (serine example: addition of serine results in covalent linkage of the inhibited enzyme and leads to bacterial death)
what reagent is used to reduce esters, amides, and nitriles
LiAlH4
why is NaBH4 note used to reduce esters?
it is very slow
result of reduction of esters
ester → 2 alcohols
result of reduction of amides
amide → amine
result of reduction of nitriles
nitrile → amine
result of reduction of acid chlorides
acid chloride → aldehyde
what reagent is used for reduction of acid chlorides
LTTBA (lithium tri(tertbutoxide) aluminum hydride
what happens in reduction of acid chlorides if NaBH4 is used instead of LTTBA
is NaBH4 is used, the acid chloride will be reduced to the alcohol instead of the aldehyde
result of grignard addition to esters
ester → tertiary alcohol
result of grignard addition to acid chlorides
acid chloride → ketone
what is added to go from arachidonic acid to anadamide?
adding the enzyme goes to anadamide
what is added to go from anadamide back to arachidonic acid?
FAAH (breaks down amide product)
what is the c-centered enolate?
negative charge on alpha carbon
what is the o centered enolate
negative charge on the oxygen (alpha carbon neutral)
what are alpha, beta unsaturated carbonyls?
carbonyls which contain at least a double bond between the alpha and beta carbon
do ester or ketone enolates have a lower pka?
Ketone enolates have a lower pka because they are more stable through resonance when deprotonated. The conjugate base of esters is destabilized by the donation of an electron pair from the alpha oxygen creating repulsive negative charges in the c-centered enolate
what carbons are deuterated in alpha H/D exchange with dioxane and D2O?
all alpha carbons
exceptions where the enol form is favored over the ketone form and why
phenols (resonance stabilization) and beta-dicarbonyls (intramolecular hydrogen bonding and conjugation) are the exceptions
Kequ when the enol vs keto form is favored
enol favord: Kequ > 1. keto favored: Kequ < 1.
result of beta halogenation in acidic conditions
ketone → beta-haloketone
result of beta-halogenation in basic conditions
ketone → carboxylic acid
rate determining step in acidic beta-halogenation
concentration of acid
rate law for acidic beta-halogenation
rate = k[ketone][H3O+] (Br2 is not in the rate law and therefore does not effect rate of reaction)
what can alpha halogenation reaction be used for?
synthesis of ylides (wittig reagent)
are SN2 reactions faster with or without carbonyl chemistry?
SN2 reactions are faster with a carbonyl carbon
what product is formed during an aldol addition reaction
the beta-hydroxycarbonyl
the betahydroxy carbonyl product indicate what reaction occured
the aldol addition reaction
the alpha,beta-unsaturated carbonyl indicates what kind of reaction occured?
an aldol condensation reaction
what product is formed during an aldol condensation reaction
an alpha,beta-unsaturated carbonyl
What does E1CB stand for
elimination unimolecular conjugate base