IB Chemistry Stuff (Regurgitation)
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
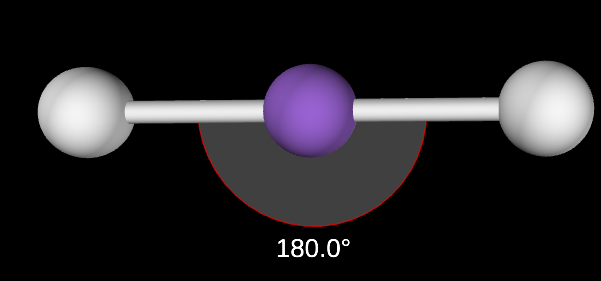
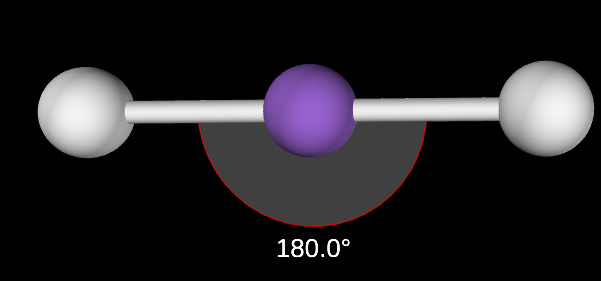
Linear Angle
180
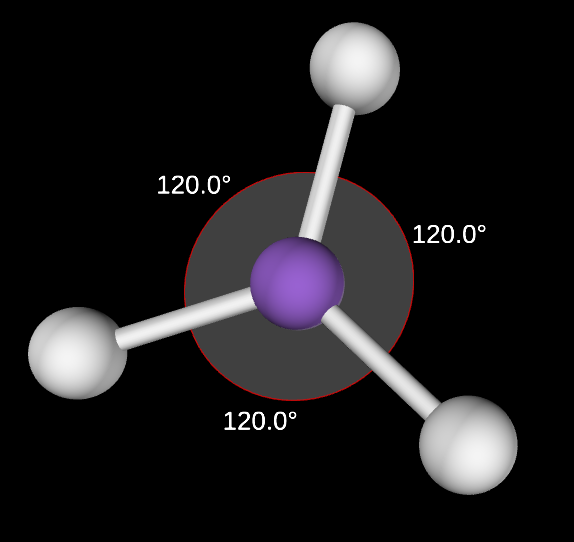
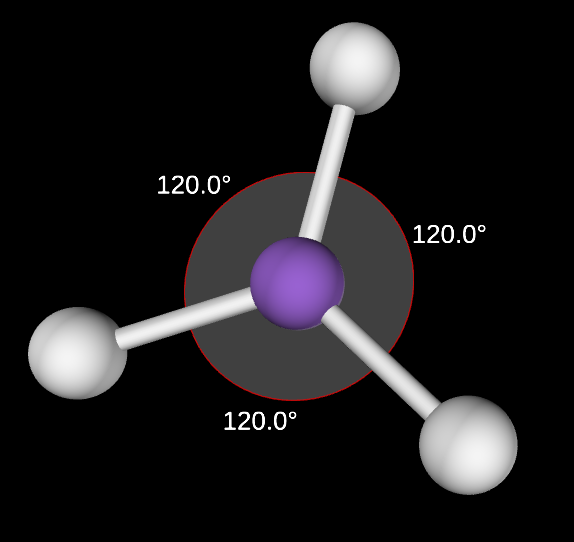
Trigonal Planar Angle
120
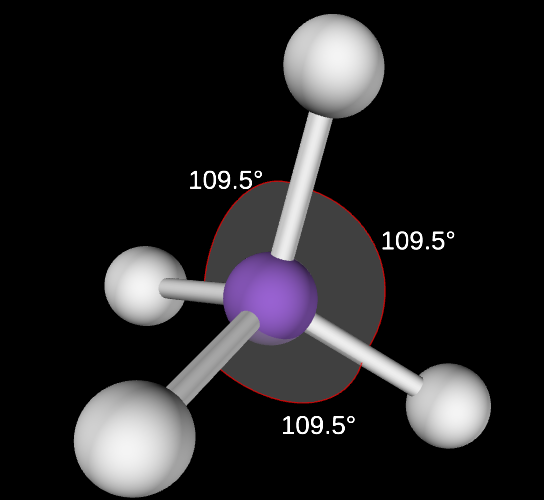
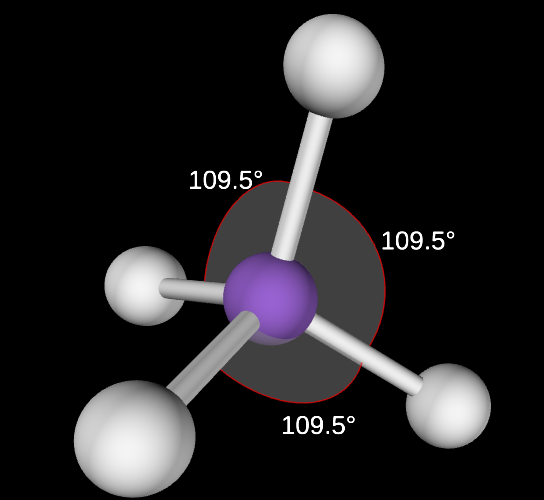
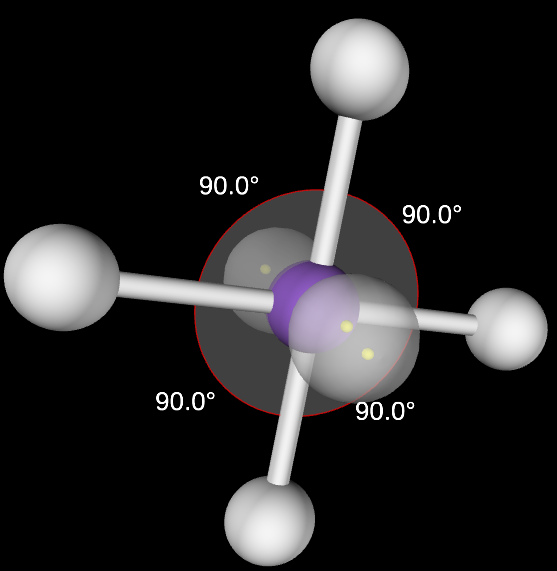
Tetrahedral Angle
109.5
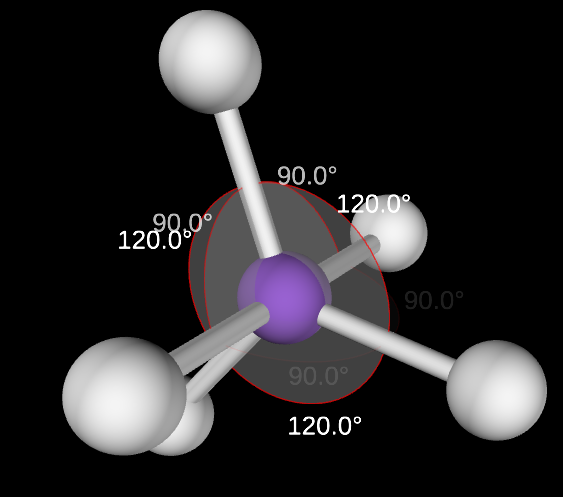
Trigonal Bipyramidal Angle
90 & 120
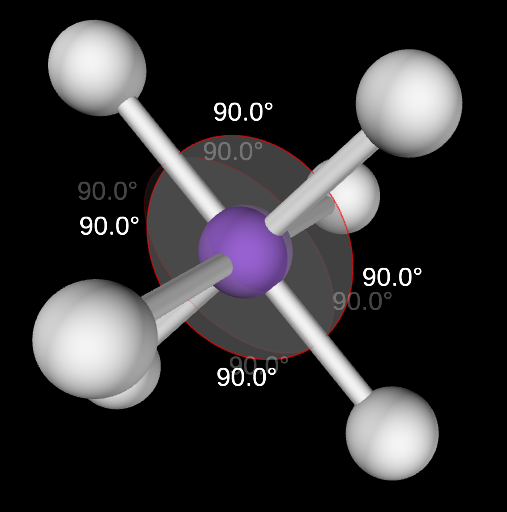
Octahedral Angle
90
Linear electron domain
2
Trigonal Planar electron domain
3
Tetrahedral electron domain
4
Trigonal Bipyramidal electron domain
5
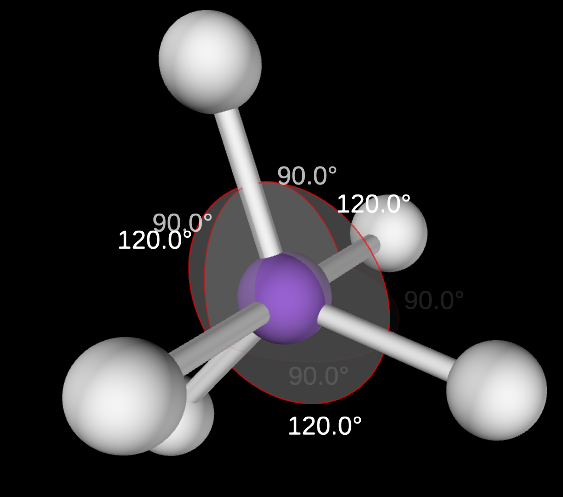
Octahedral electron domain
6
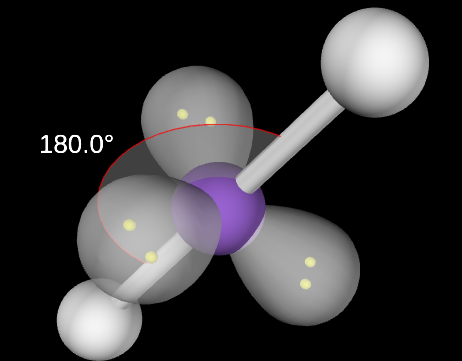
Linear Molecular Geometry
2,0 or 2,3
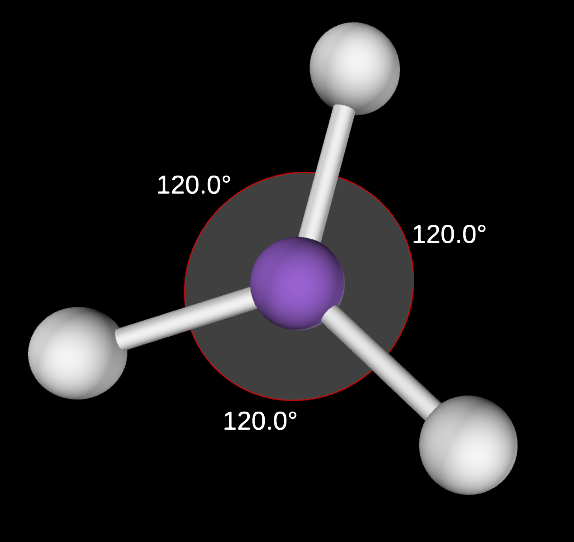
Trigonal Planar Molecular Geometry
3,0
Bent Molecular Geometry
2,1 or 2,2
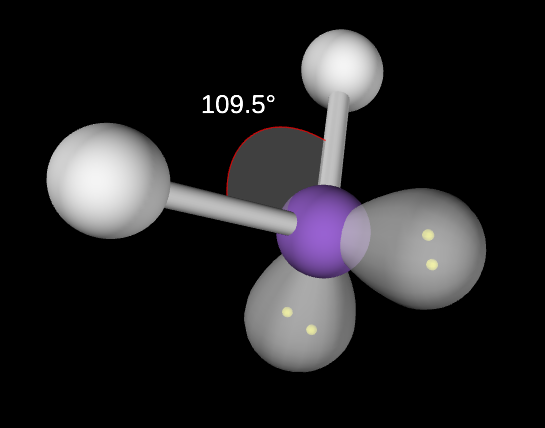
Tetrahedral Molecular Geometry
4,0
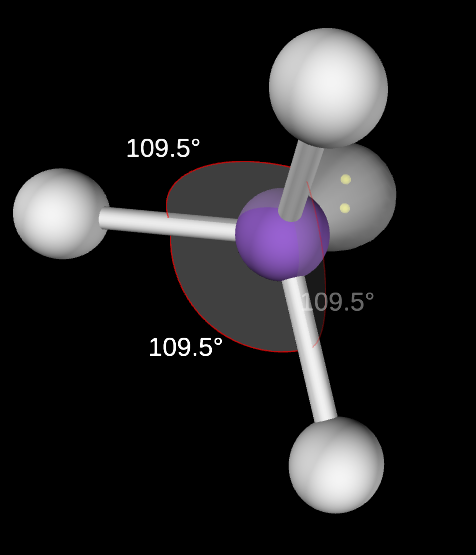
Trigonal Pyramidal Molecular Geometry
3,1
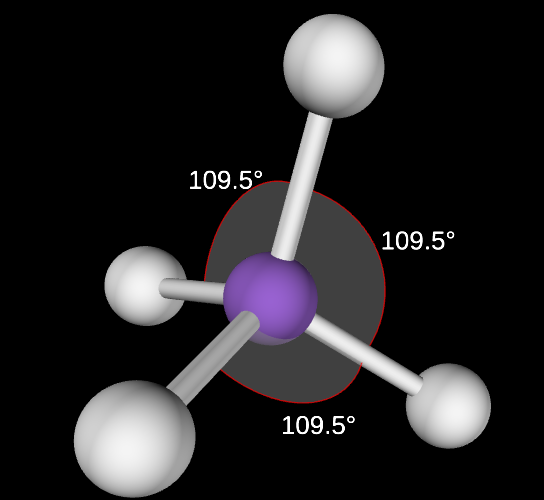
Trigonal Bipyramidal Molecular Geometry
5,0
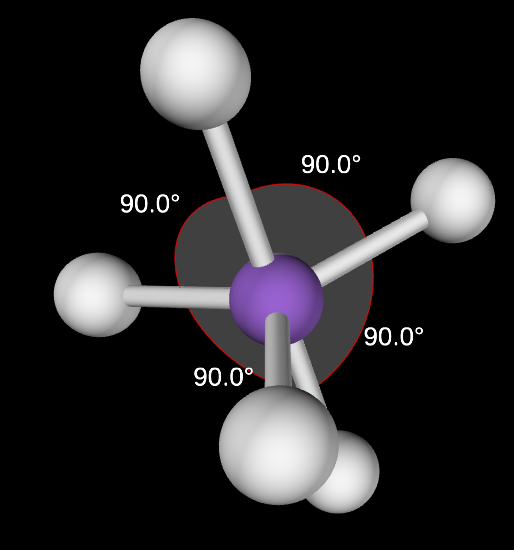
Seesaw Molecular Geometry
4,1
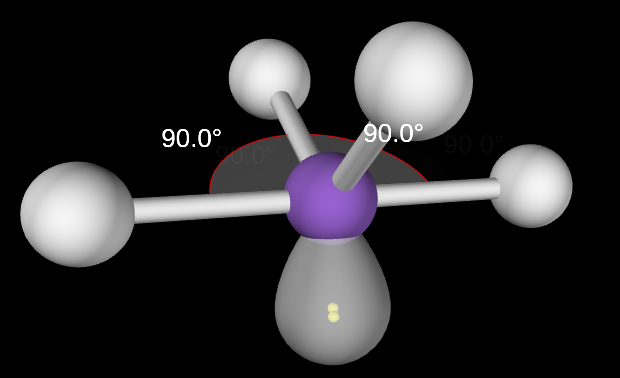
T-Shaped Molecular Geometry
3,2
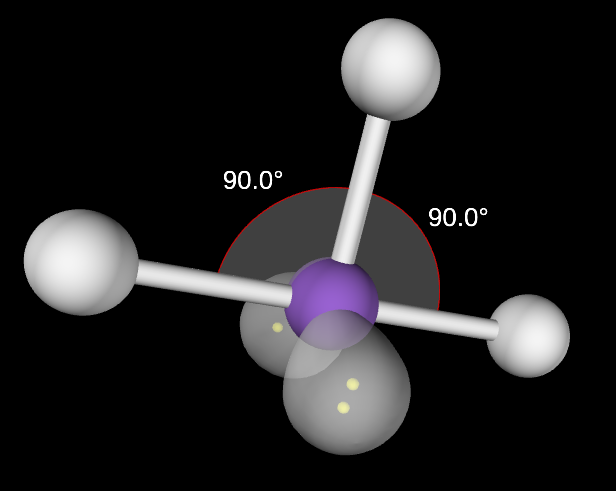
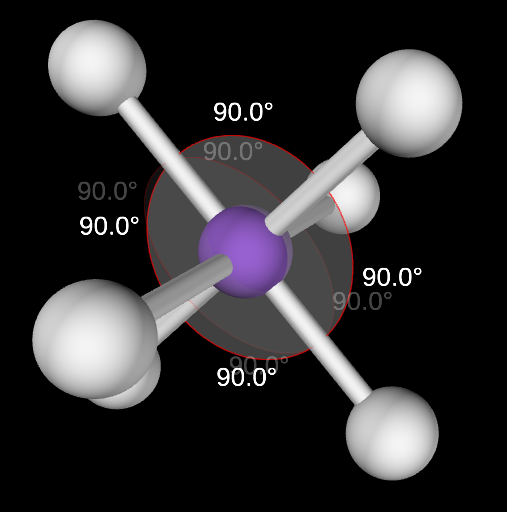
Octahedral Molecular Geometry
6,0
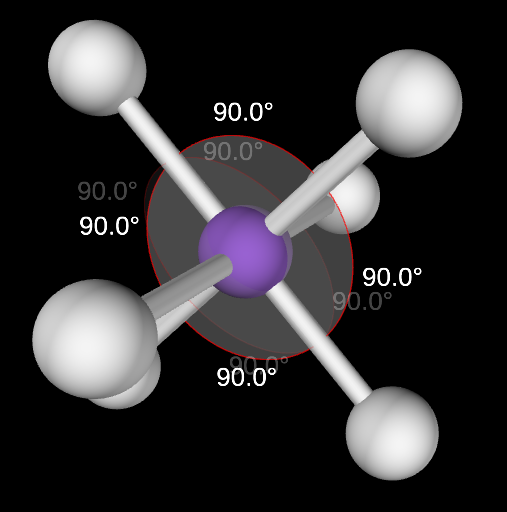
Square Pyramidal Molecular Geometry
5,1
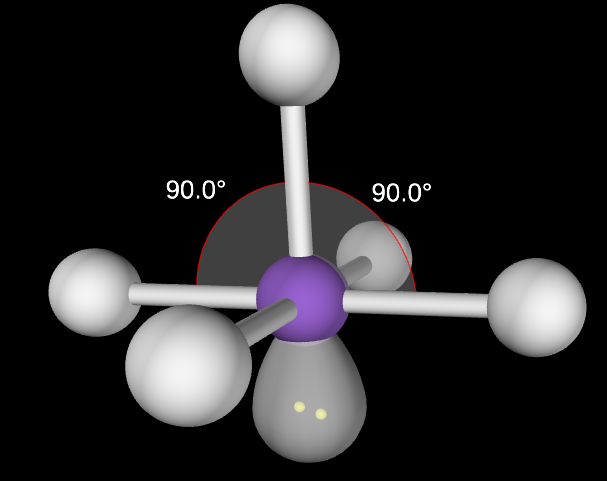
Square Planar Molecular Geometry
4,2
Alkane
end in -ane
General formula CnH2n+2
Alkene
Alkenyl Functional Group
end in -ene
General formula CnH2n
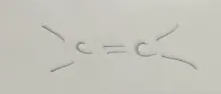
Alkyne
Alkynel Functional Group
end in -yne
General formula CnH2n-2
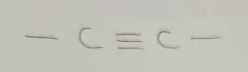
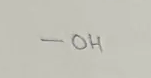
Alcohol
Hydroxyl Functional Group
end in -anol
General formula CnH2n+1OH
Ether
Alkoxy Functional Group
end in -oxyalkane
General formula CnH2n+2O (R-O-R’)
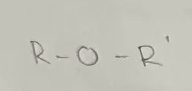
Aldehyde
Carbonyl (Aldehyde) Functional Group
end in -anal
General formula CnH2nO (R-CHO)
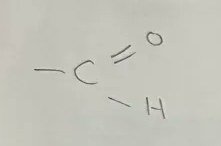
Ketone
Carbonyl (Ketone) Functional Group
end in -anone
General formula CnH2nO (R-CO-R’)
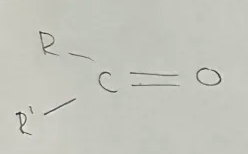
Carboxylic Acid
Carboxyl (Acid) Functional Group
end in -anoic acid
General formula CnH2n+1COOH
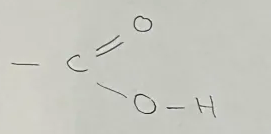
Ester
Carboxyl (Ester) Functional Group
end in -anoate
General formula CnH2nO2 (R-COO-R’)
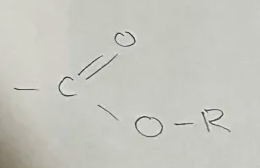
Amide
Amido Functional Group
end in -anamide
General formula CnH2n+1CONH2
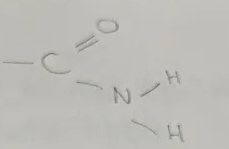
Amine
Amino Functional Group
end in -anamine
General Formula CnH2n+1NH2

Halogenoalkene
Halogeno Functional Group
BEGIN with Fluoro-, Bromo-, Chloro-, or Iodo-
General Formula CnH2n+1X, where X is F, Br, Cl, or I
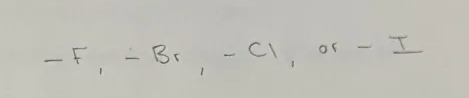
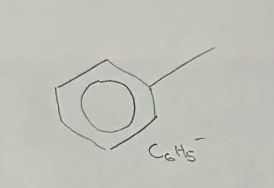
Arene
Phenyl Functional Group
end with - benzene
C6H5-
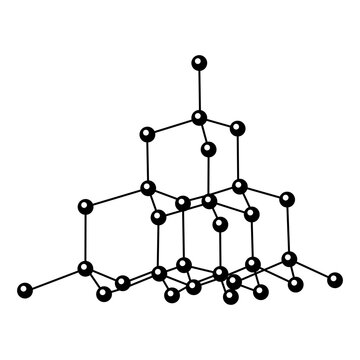
Diamond Structure
sp3 hybridized carbon atoms; covalent bonds to 4 carbon each; tetrahedral, all bond angles are 109.5 degrees
Diamond Electrical Conductivity
Not conductive; all electrons are bonded (non-mobile)
Diamond Thermal Conductivity
Very good at conducting heat (better than metals)
Diamond Appearance
Highly transparent, lustrous crystal
Diamond Physical and Chemical Properties
Hardest known natural substance; incapable of being scratched; brittle; high melting point
Diamond Uses
Jewelry, Ornamentation, Tools and Machinery to Grind or Cut Glass
Graphite Structure
sp2 hybridized carbons that are covalently bonded to 3 others (creates hexagons)
parallel layers with bond angles of 120 degrees
remaining valence electron on each carbon is delocalized— can move around freely within the layer
layers can slide over each other bc held together by LDF only
Graphite Electrical Conductivity
Good electrical conductivity bc of free moving valence electrons within each layer
Graphite Heat Conductivity
Not good at conducting heat unless the heat is forced to conduct parallel to the layers
Graphite appearance
Non-lustrous, gray crystalline solid
Graphite Physical/Chemical Properties
Soft/slippery— layers slip over each other due to weak LDFS
brittle, very high melting point, most stable carbon allotrope
Graphite uses
Dry lubricant, pencils, electrode rods in electrolysis
Graphene Structure
Same as Graphite but only 1 layer — still has one delocalized valence electron for each carbon
“honeycomb” structure
Graphene Appearance
Almost transparent
Graphene Physical/Chemical Properties
Thickness of 1 atom => thinnest material to ever exist
100 times stronger than steel
Very flexible
very high melting point
Graphene Uses
TEM (transmission electron microscopy) grids
photo-voltaic cells
touch screens
high-performance electronics
applications are still being developed
C60 (Fullerene or Buckyball) Structure
sp2 hybridized carbons that are bonded into a sphere — 12 pentagons and 20 hexagons
Spherical cage where each carbon is bonded to three others
NOT a giant molecule because it has a formula
Fullerene electrical conductivity
Poor electrical conductors; individual molecules have delocalized electrons, but there is little movement between the molecules
Fullerene Thermal Conductivity
Very Low Thermal Conductivity
Graphene Electrical Conductivity
Very good electrical conductor due to one delocalized electron per atom giving electron mobility across layers
Graphene Thermal Conductivity
Best thermal conductivity known, better than diamond
Fullerene Appearance
Black Powder
Fullerence Chemical/Physical Properties
Very light and strong;
Reacts with potassium to make superconducting crystalline material
low melting point
Fullerene Uses
Lubricants, medical, and industrial devices for binding specific target molecules
Related forms for nanotubes and nanobuds as well as catalysts
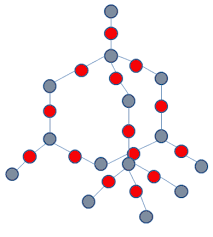
SiO2 Structure
Tetrahedral positions with all 4 silicon valence electrons involved
Each silicon is bonded to 4 oxygen, which are each bonded to 2 silicon
SiO2 Properties
Strong, Insoluble in water, high melting point, not electrically conductive
(Think about sand/glass/quartz)