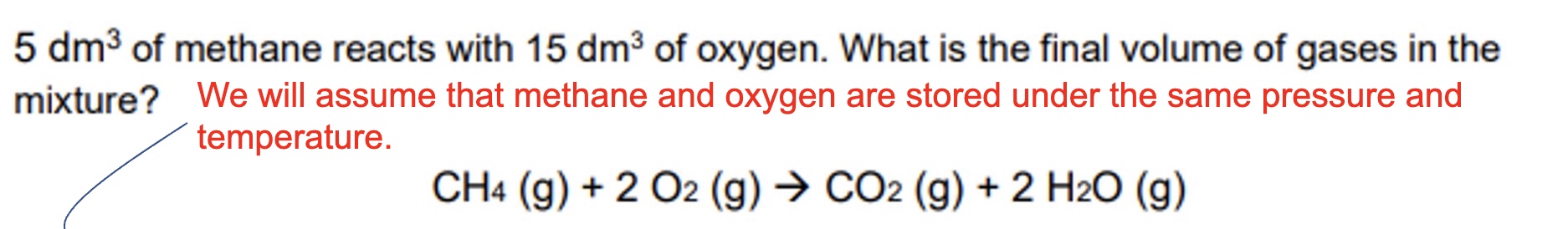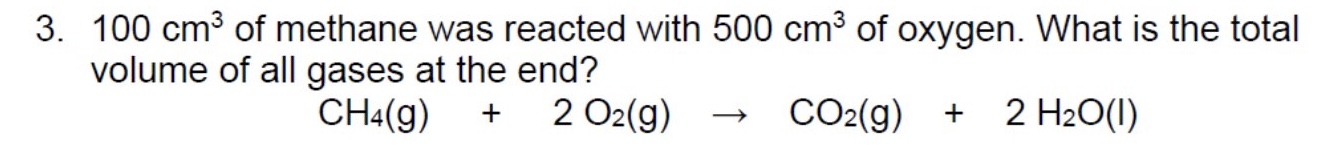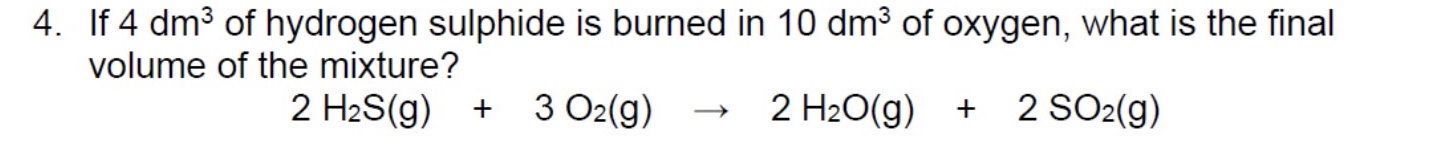3.1.2.3 the ideal gas equation
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
gases also react in ______ amounts, but its much more convenient to consider the ______ of a gas than its mass
molar
volume
give the ideal gas equation
pV = nRT
pressure x volume = moles x gas constant x temp
gives the units of the ideal gas equation

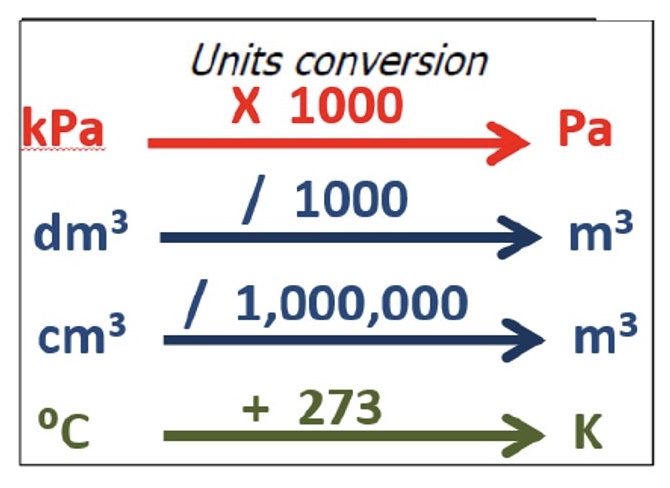
how do you convert between:
a. KPa + Pa
b. dm³ + m³
c. cm³ + m³
d. ℃ + K
what is the ideal gas? give 4 features
the ideal gas:
is made of identical particles in continuous random motion
the particles have position but zero volume
collisions between particles are completely elastic ie no energy is lost
there are no forces acting between the molecules
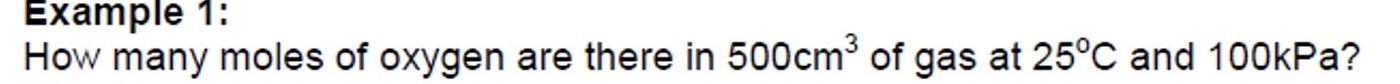
the gas constant is 8.31
make a list of what values you have + what is the unknown
convert units if needed
rearrange ideal gas equation
substitute values
answer = 0.020 mol




the volume of a gas depends on what? + this means what?
on the temperature, pressure + number of moles
this means that under the same conditions of temperature + pressure, the volume of gas will only be affected by the change in moles