BIO 300 Lecture3.CellCycleCancerIntro Review
1/55
Earn XP
Description and Tags
BIO 300 Pathophysiology
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
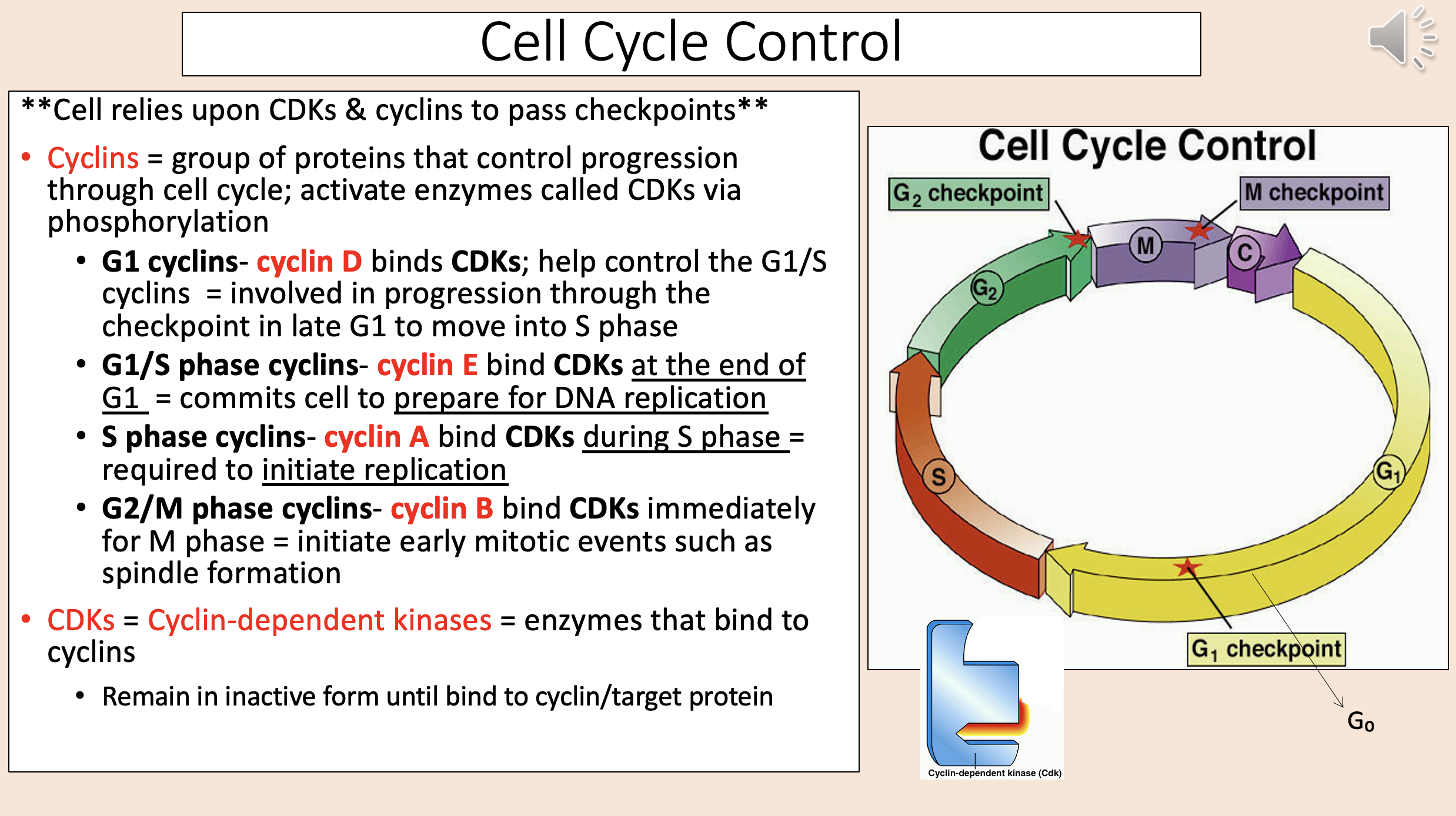
Orderly sequence of events that occur as a cell duplicates its contents & divides
cell cycle
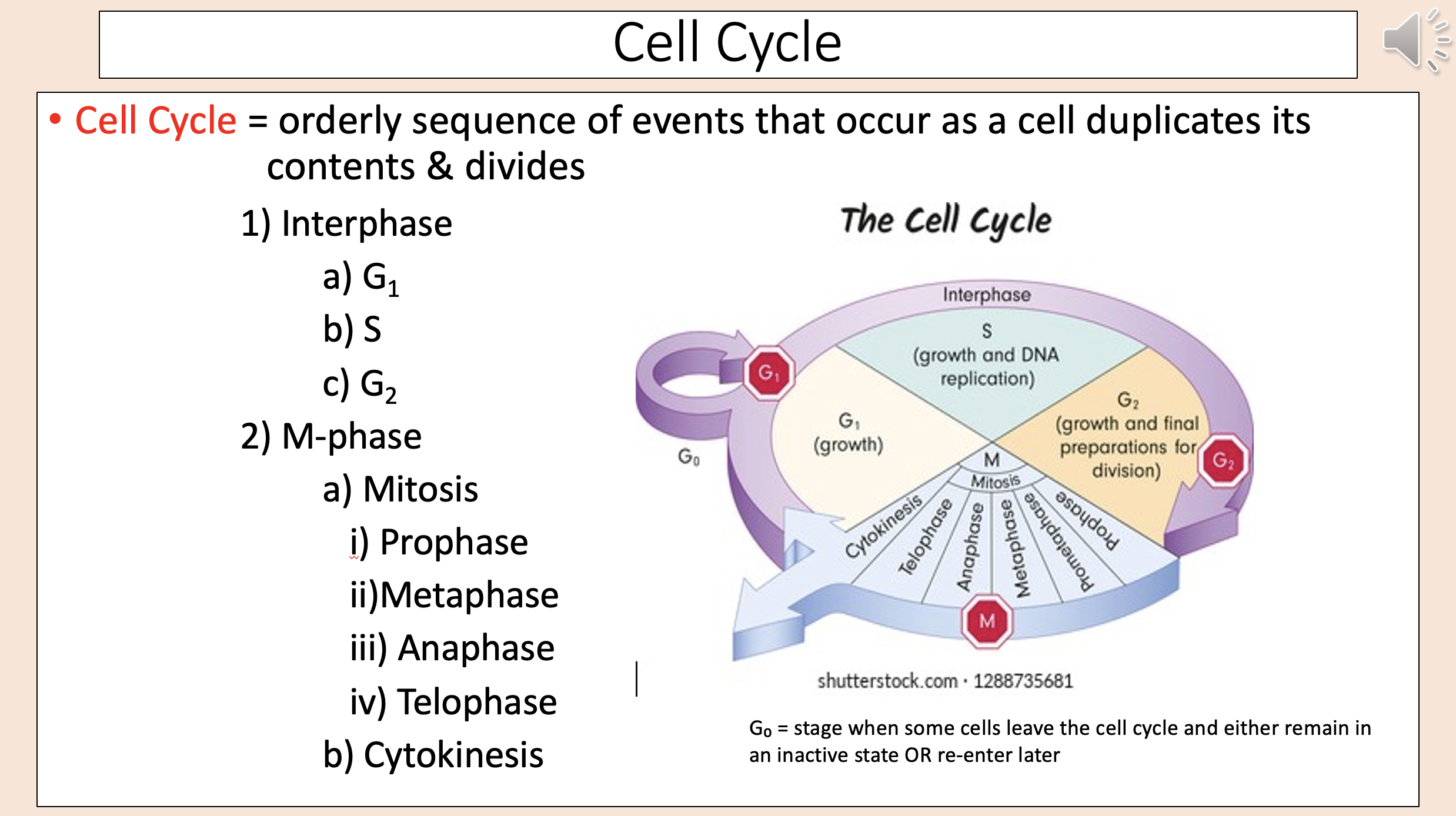
Cell Cycle:
interphase
G1
S
G2
M-phase
mitosis
prophase
metaphase
anaphase
telophase
cytokinesis
Interphase
G1
S
G2
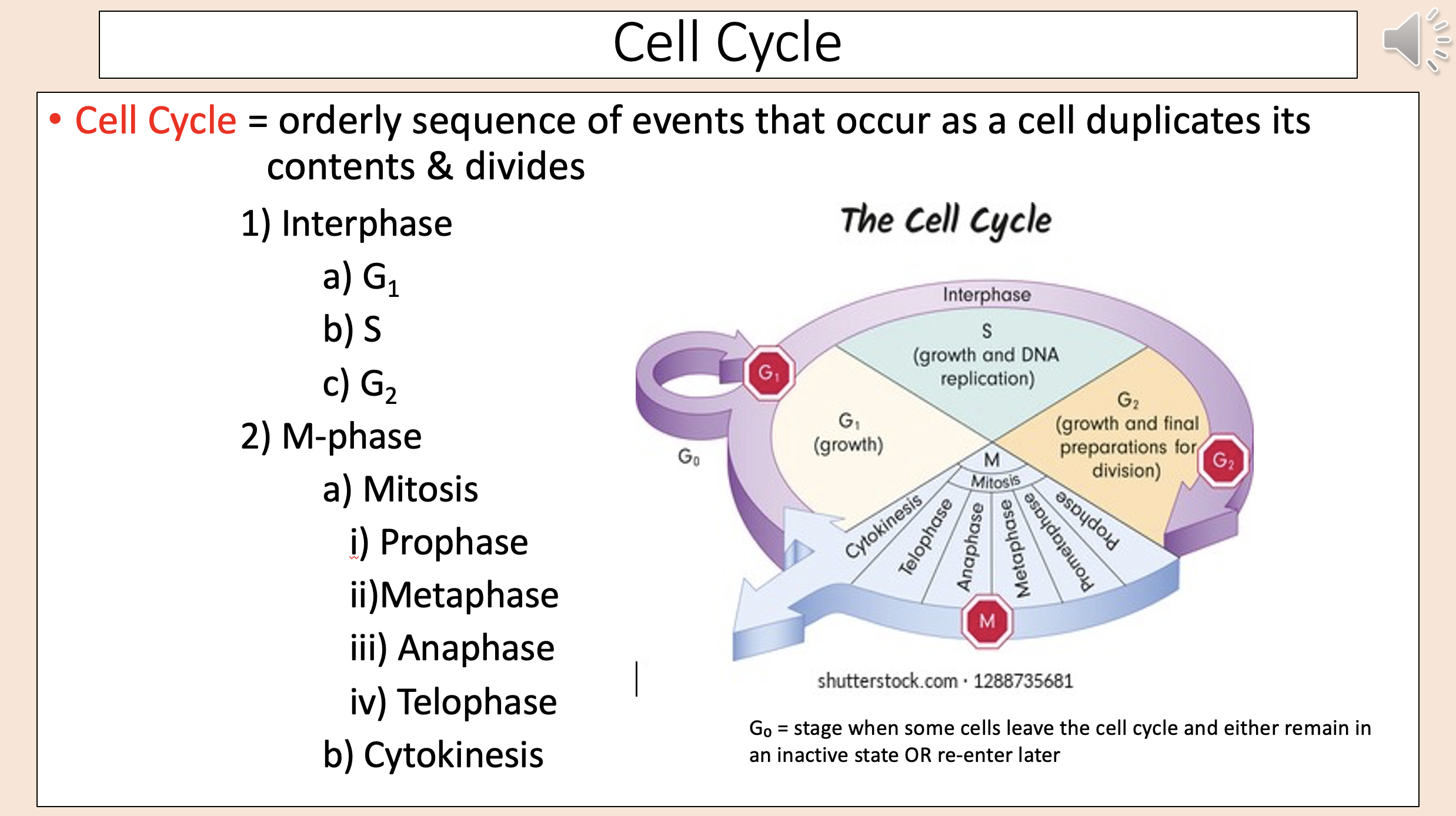
M-phase
mitosis
prophase
metaphase
anaphase
telophase
cytokinesis
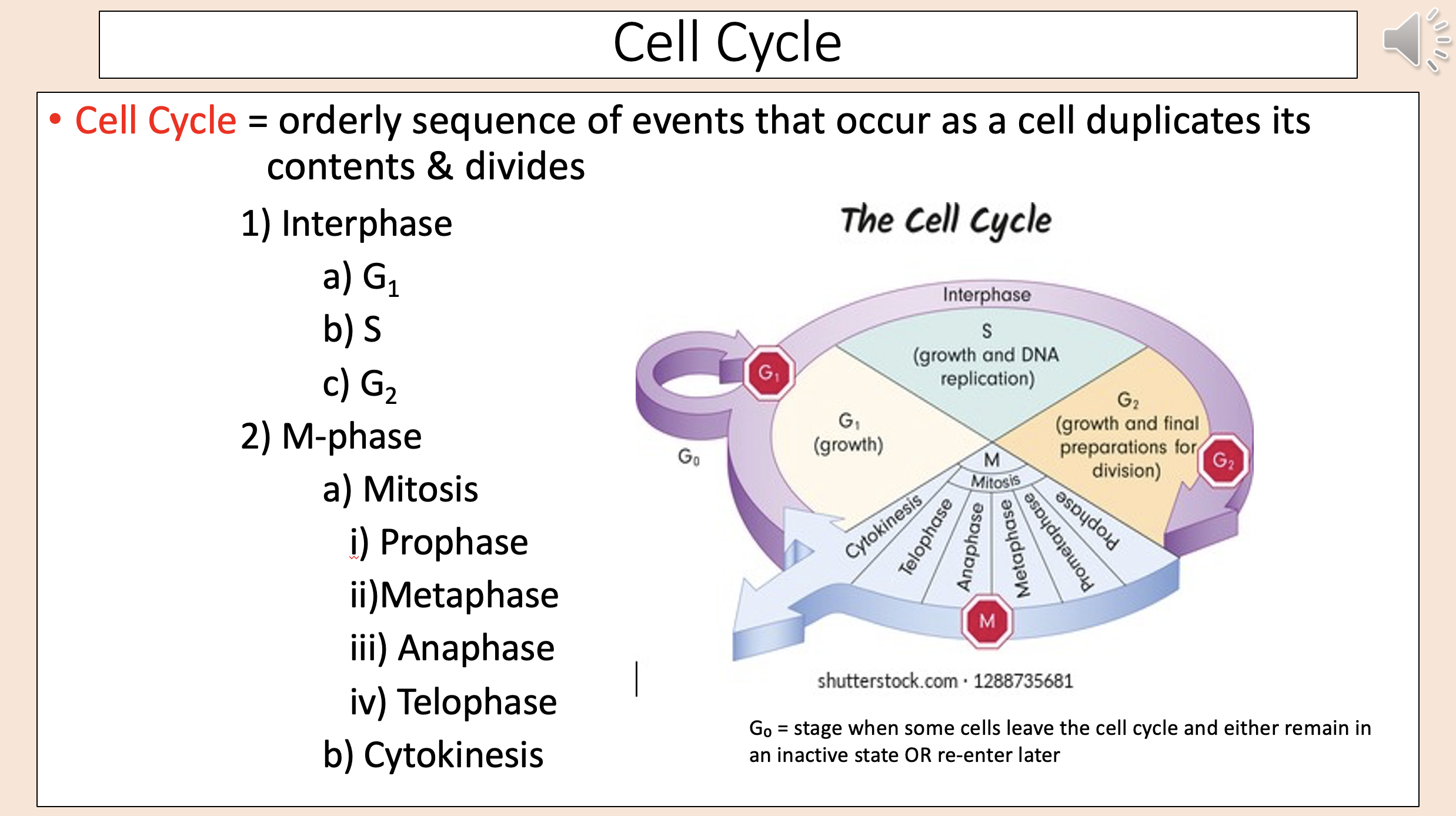
Stage when some cells leave the cell cycle and either remain in an inactive state OR re-enter later
G0
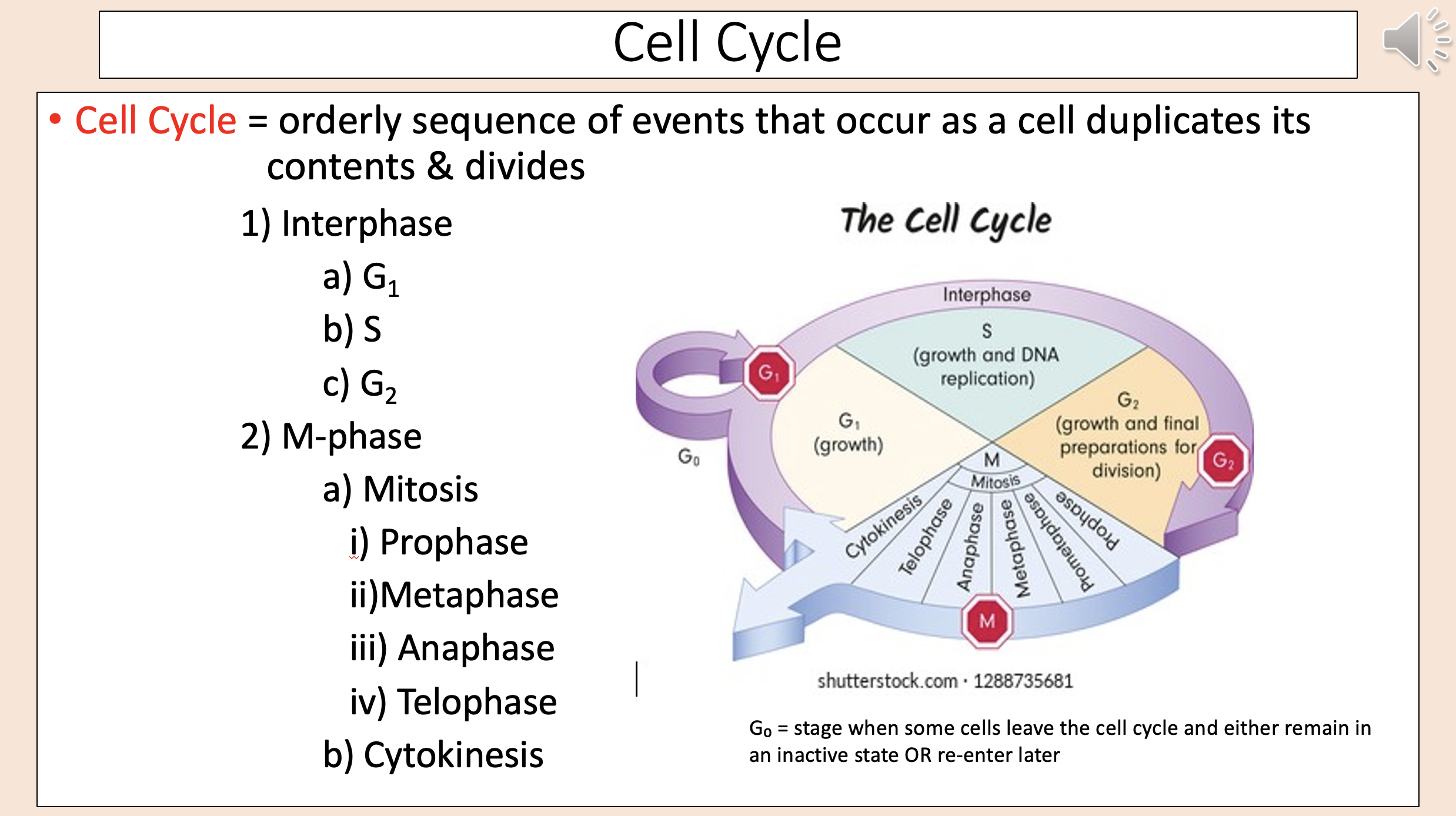
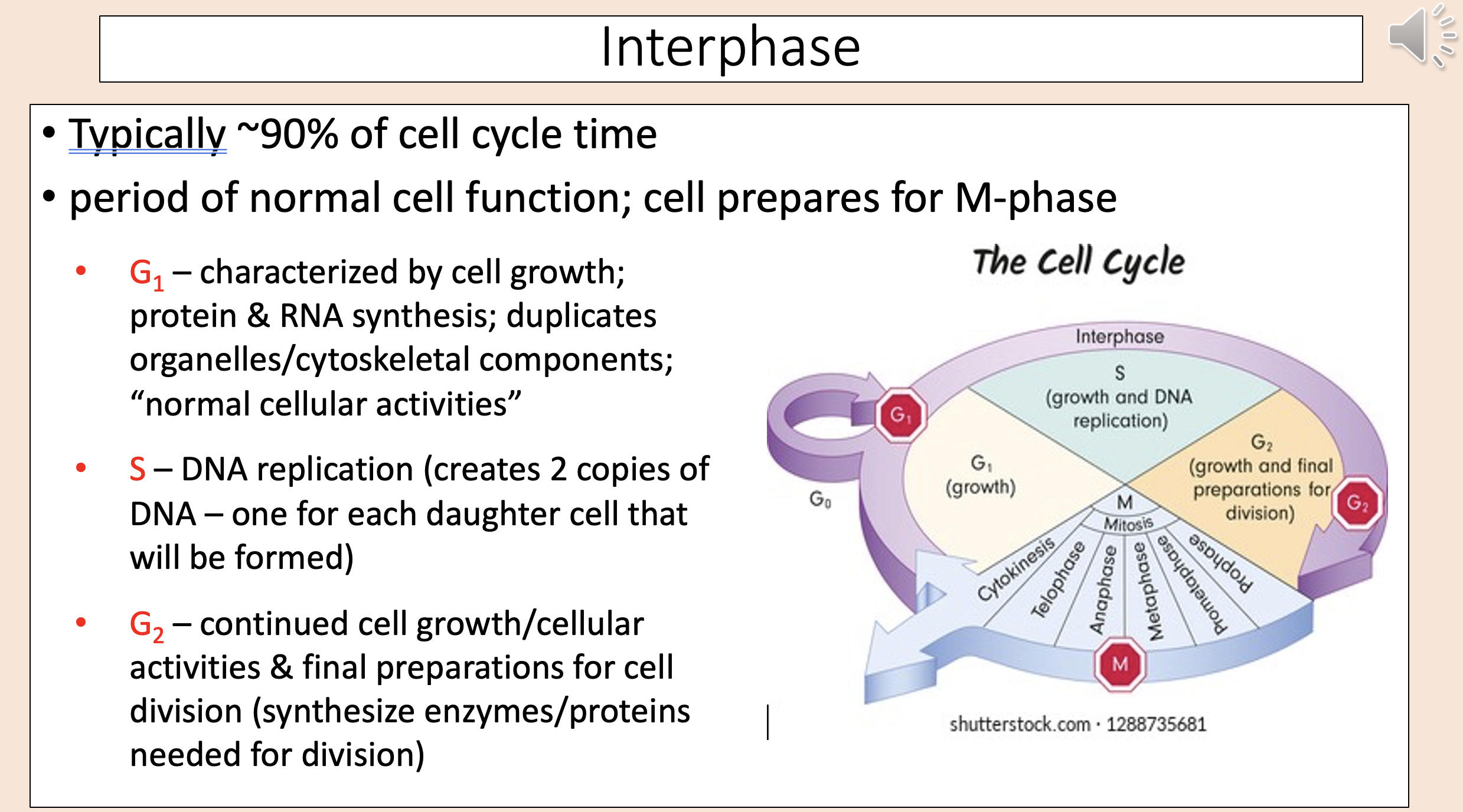
Typically ~90% of cell cycle time.
Period of normal cell function; cell prepares for M-phase.
interphase
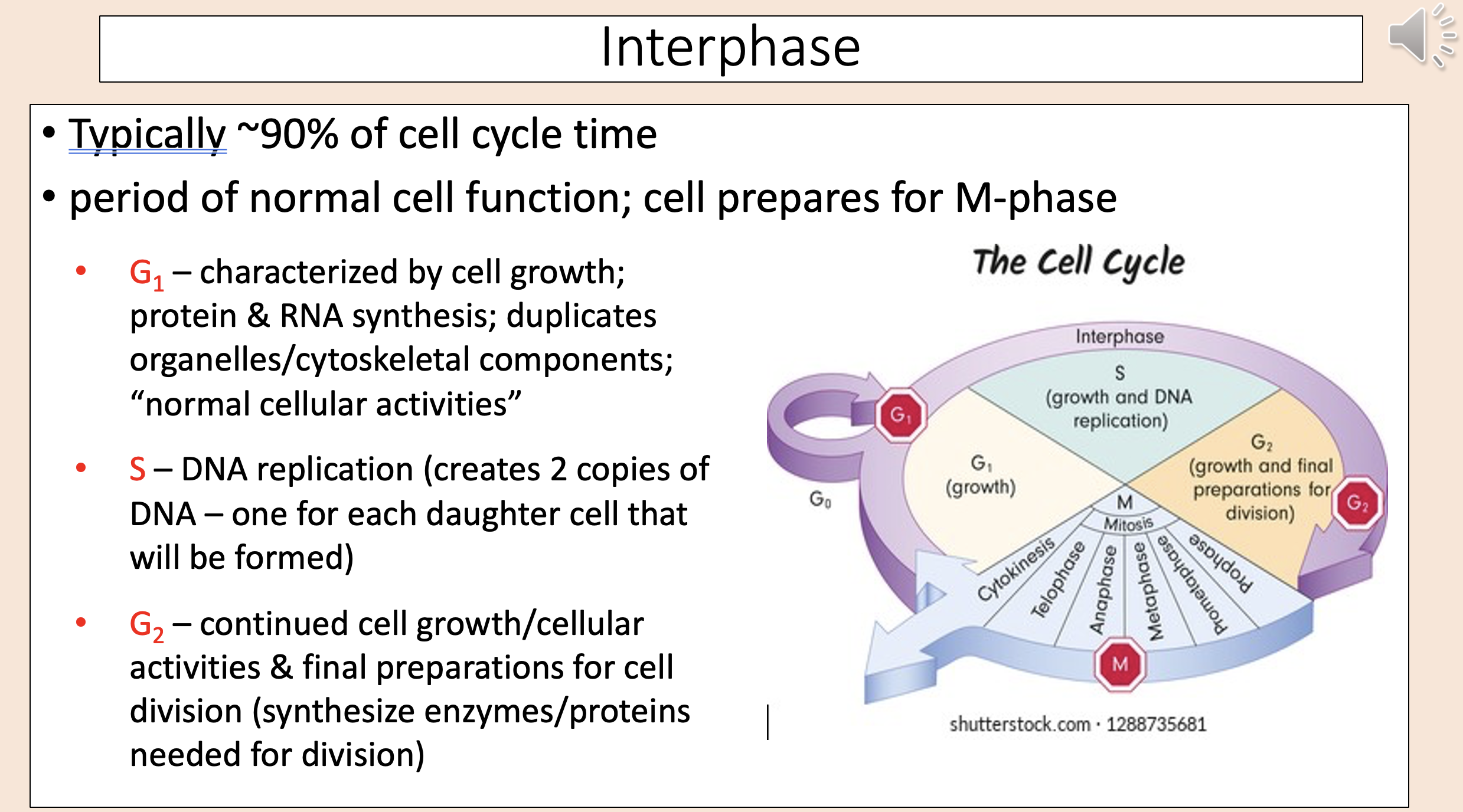
Characterized by cell growth; protein & RNA synthesis; duplicates organelles/cytoskeletal components; “normal cellular activities”
G1
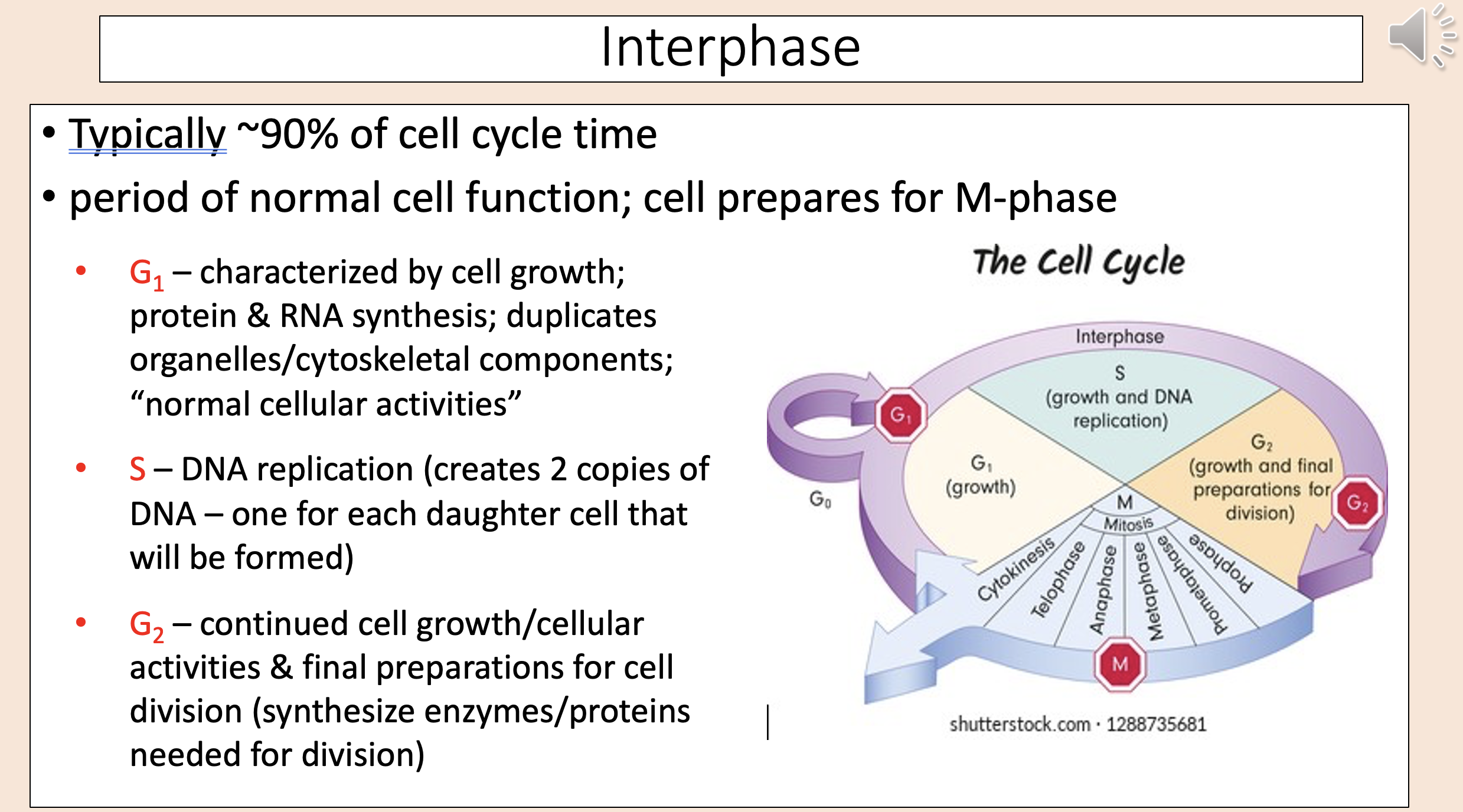
DNA replication (creates 2 copies of DNA – one for each daughter cell that will be formed)
S
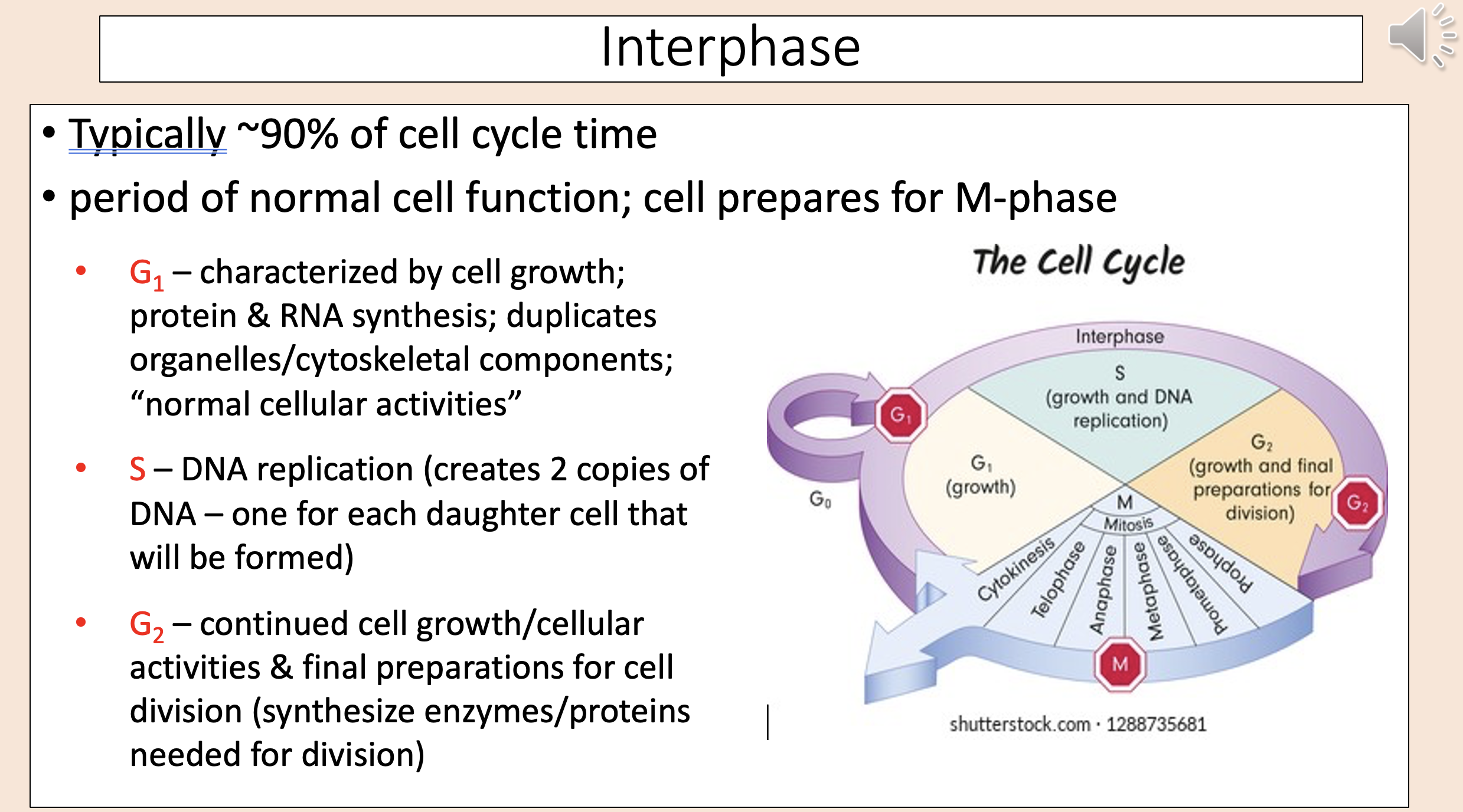
Continued cell growth/cellular activities & final preparations for cell division (synthesize enzymes/proteins needed for division)
G2
A long molecule of DNA that is tightly coiled together with several proteins
chromosome
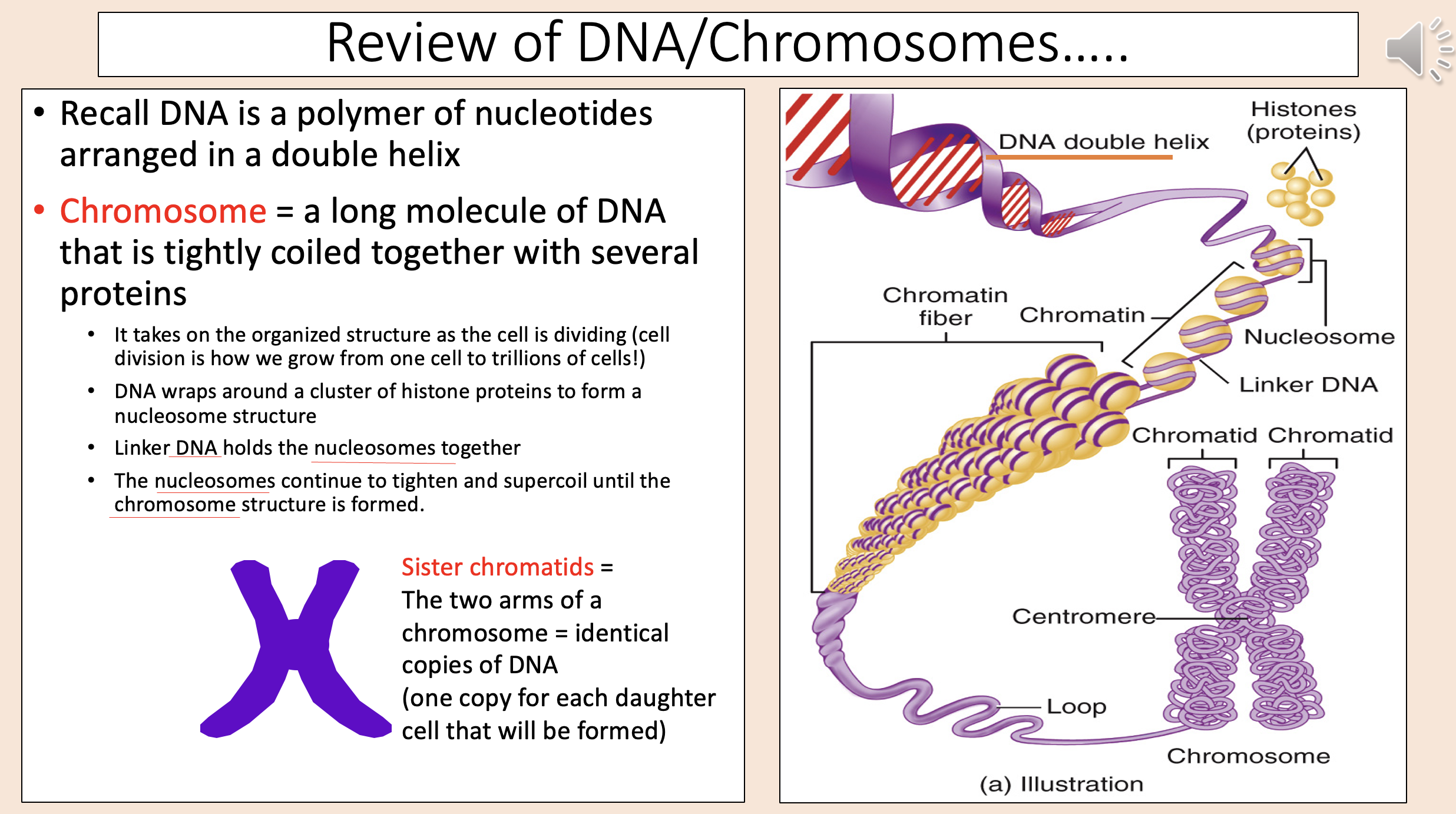
The two arms of a chromosome = identical copies of DNA (one copy for each daughter cell that will be formed)
sister chromatids
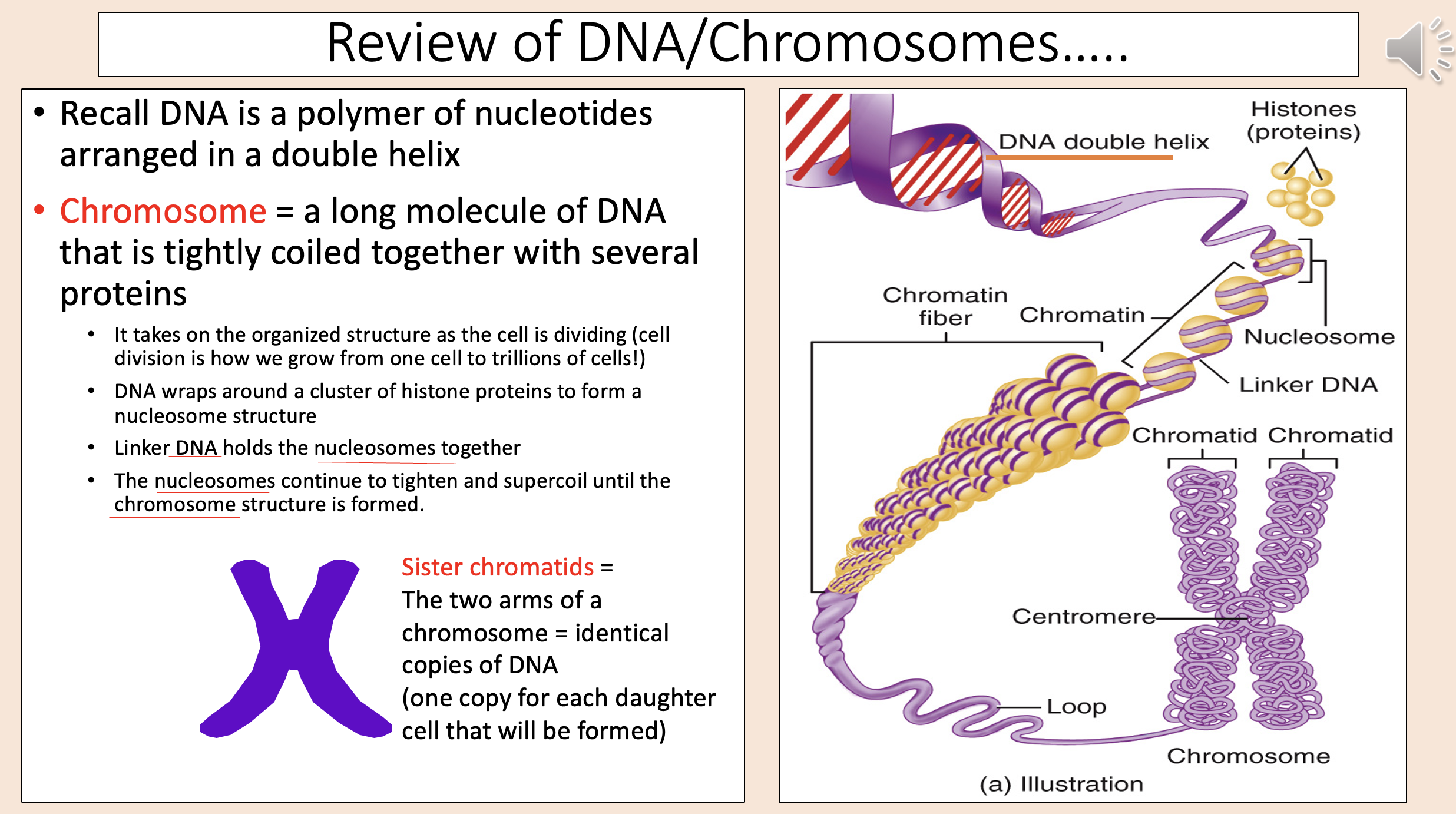
Spindle begins to form in centrosome area (centrioles are located in this area).
Chromatin condenses into chromosomes.
Nucleoli disappear.
Nuclear envelope dissolves.
Centrosomes/centrioles migrate to opposite poles of cell.
Spindle spreads across cell.
Chromosomes migrate; their kinetochore region attaches to kinetochore microtubules in spindle, allowing them to move.
prophase
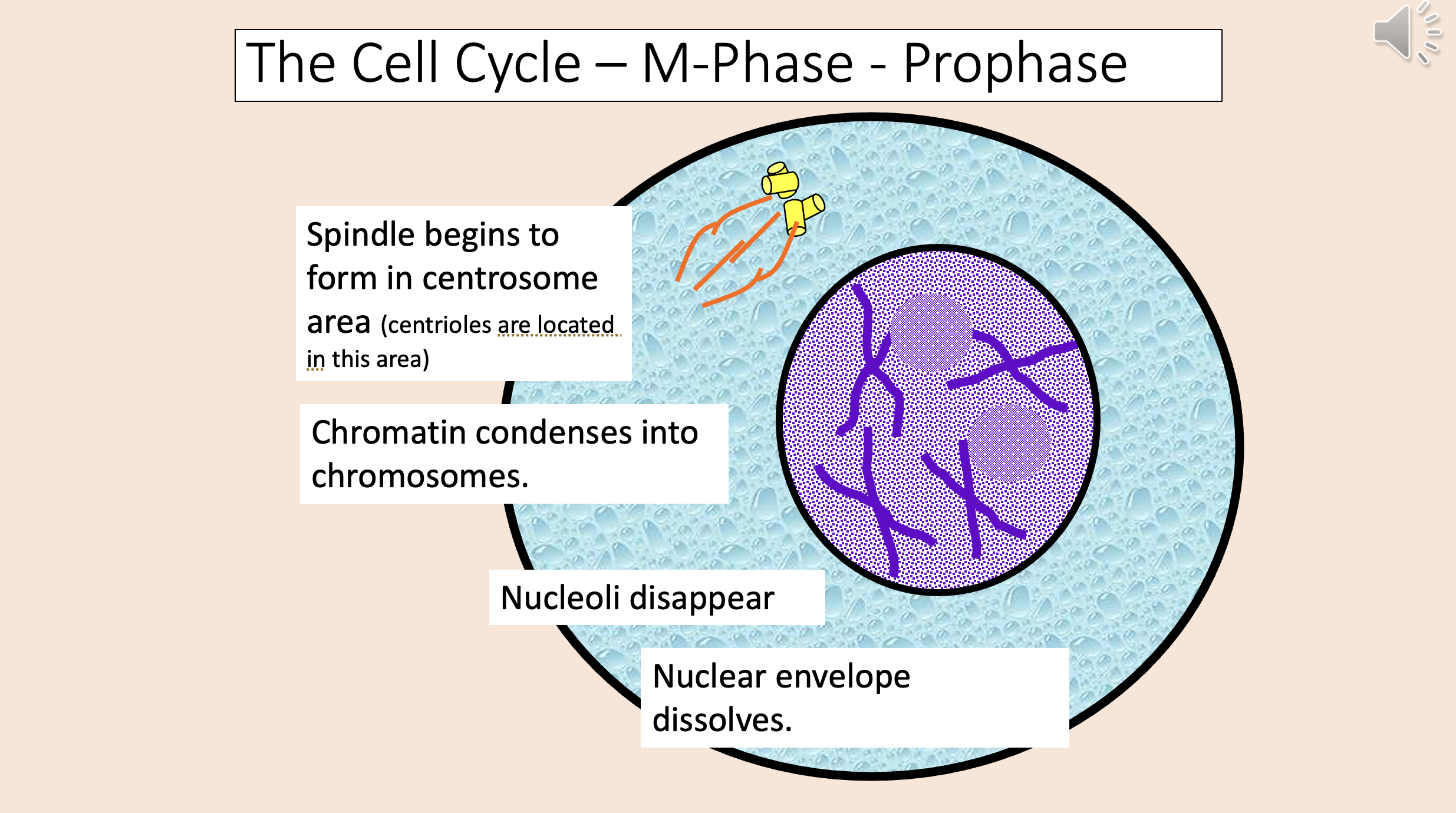
Chromosomes aligned by kinetochore microtubules along equator (aka metaphase plate)
metaphase
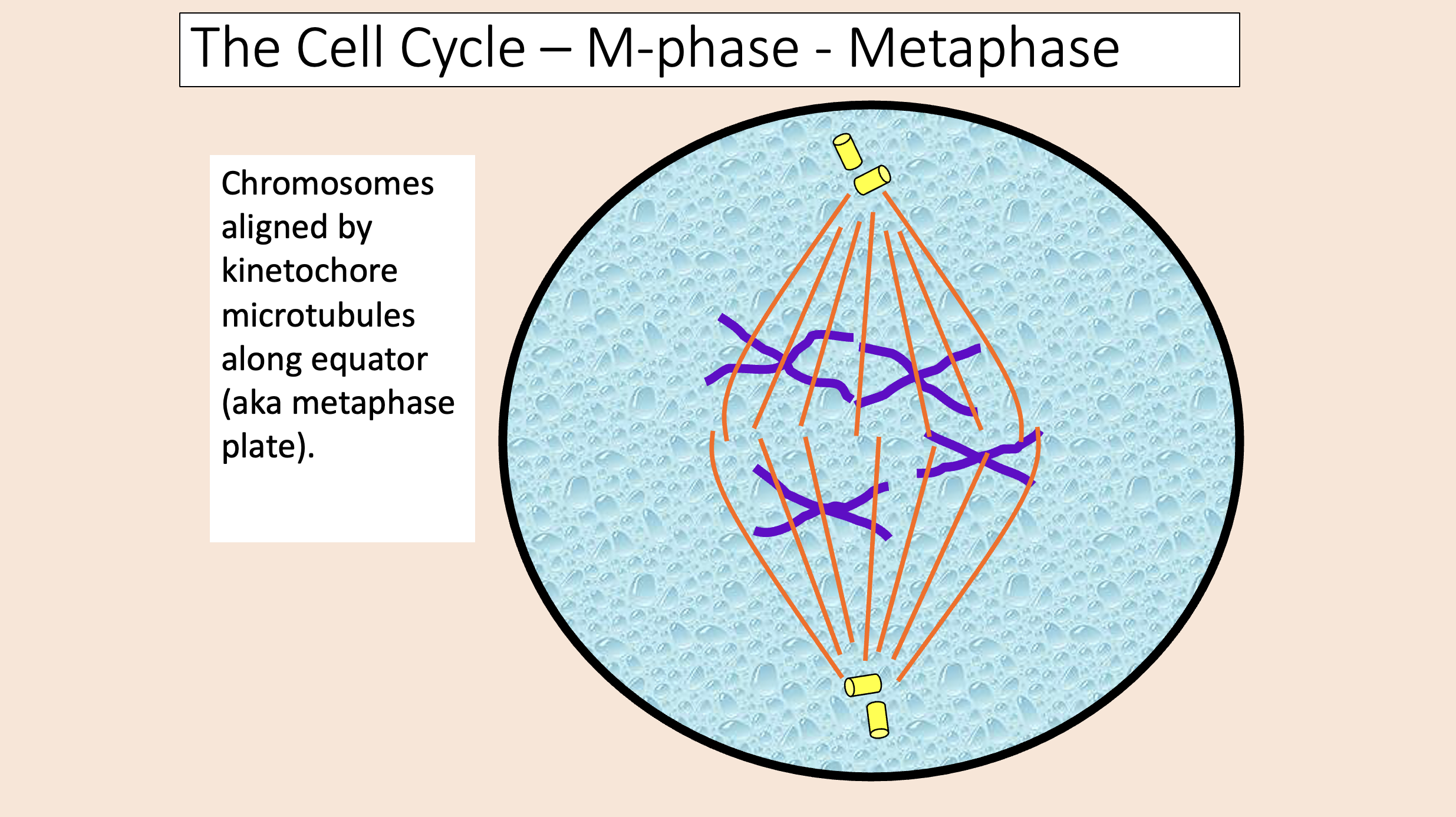
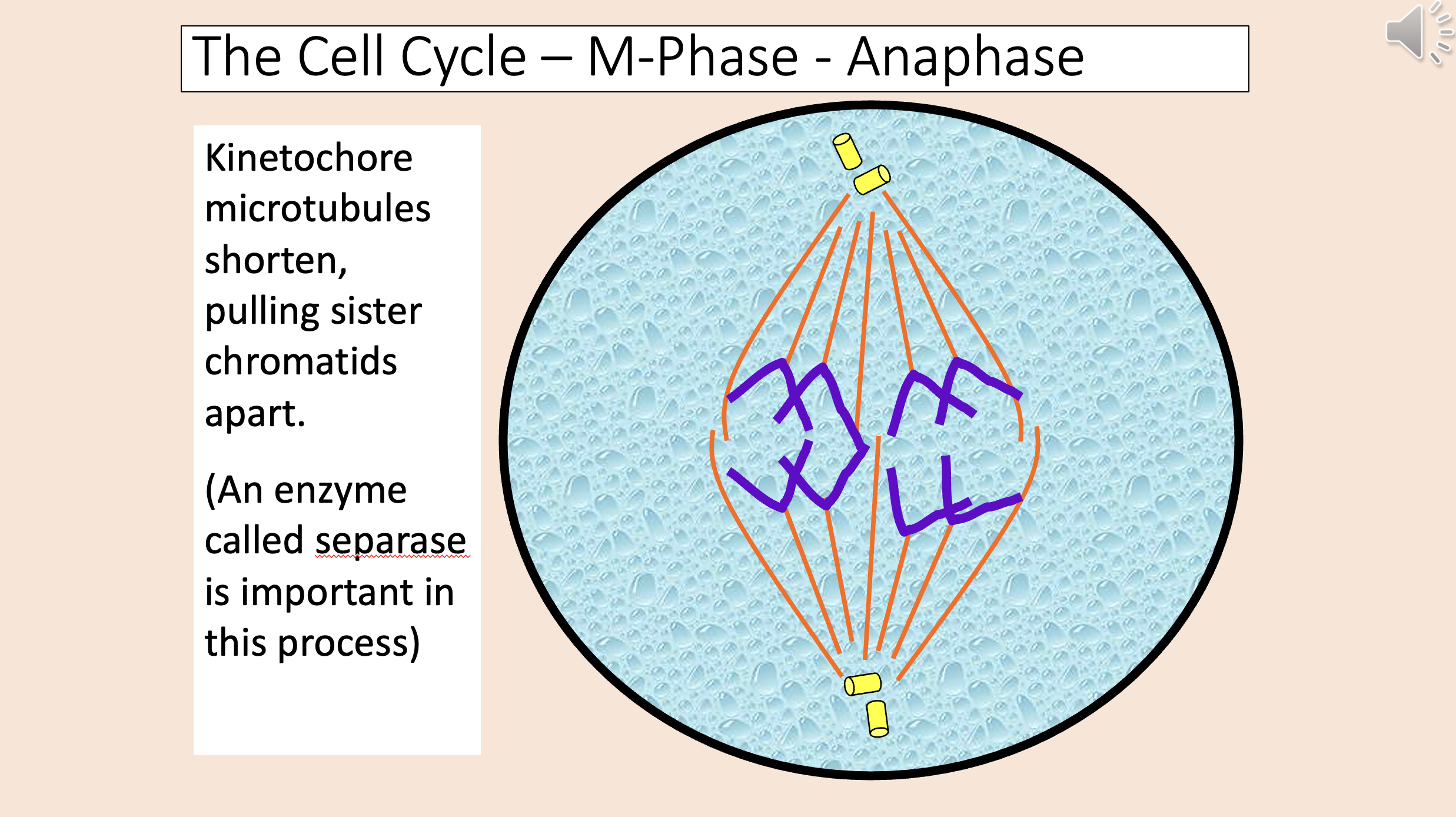
Kinetochore microtubules shorten, pulling sister chromatids apart.
An enzyme called separase is important in this process.
Chromatids migrate toward centrioles.
Polar microtubules lengthen, pushing poles apart.
anaphase
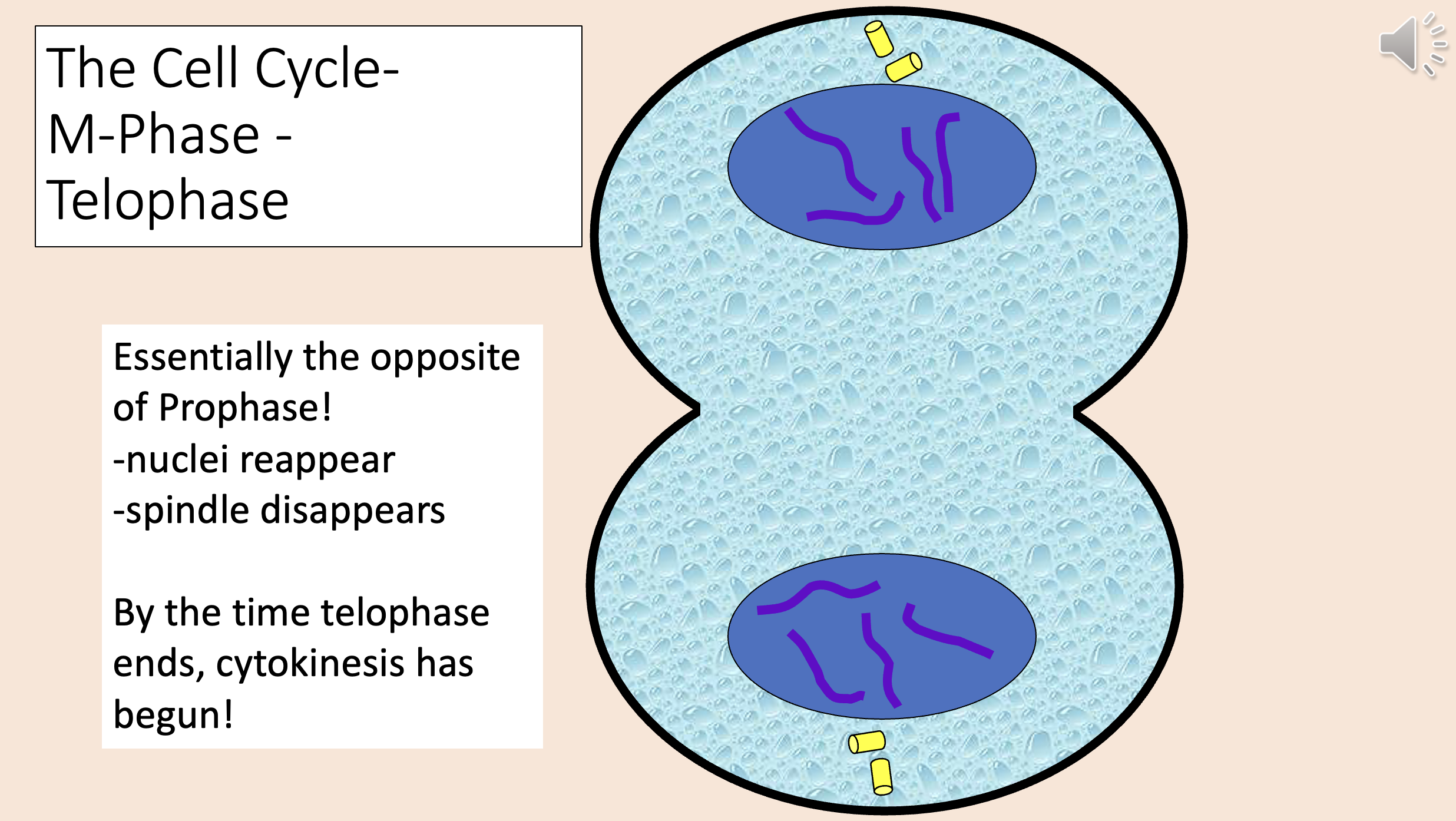
The opposite of Prophase!
Nuclei reappear.
Spindle disappears.
By the time telophase ends, cytokinesis has begun!
telophase
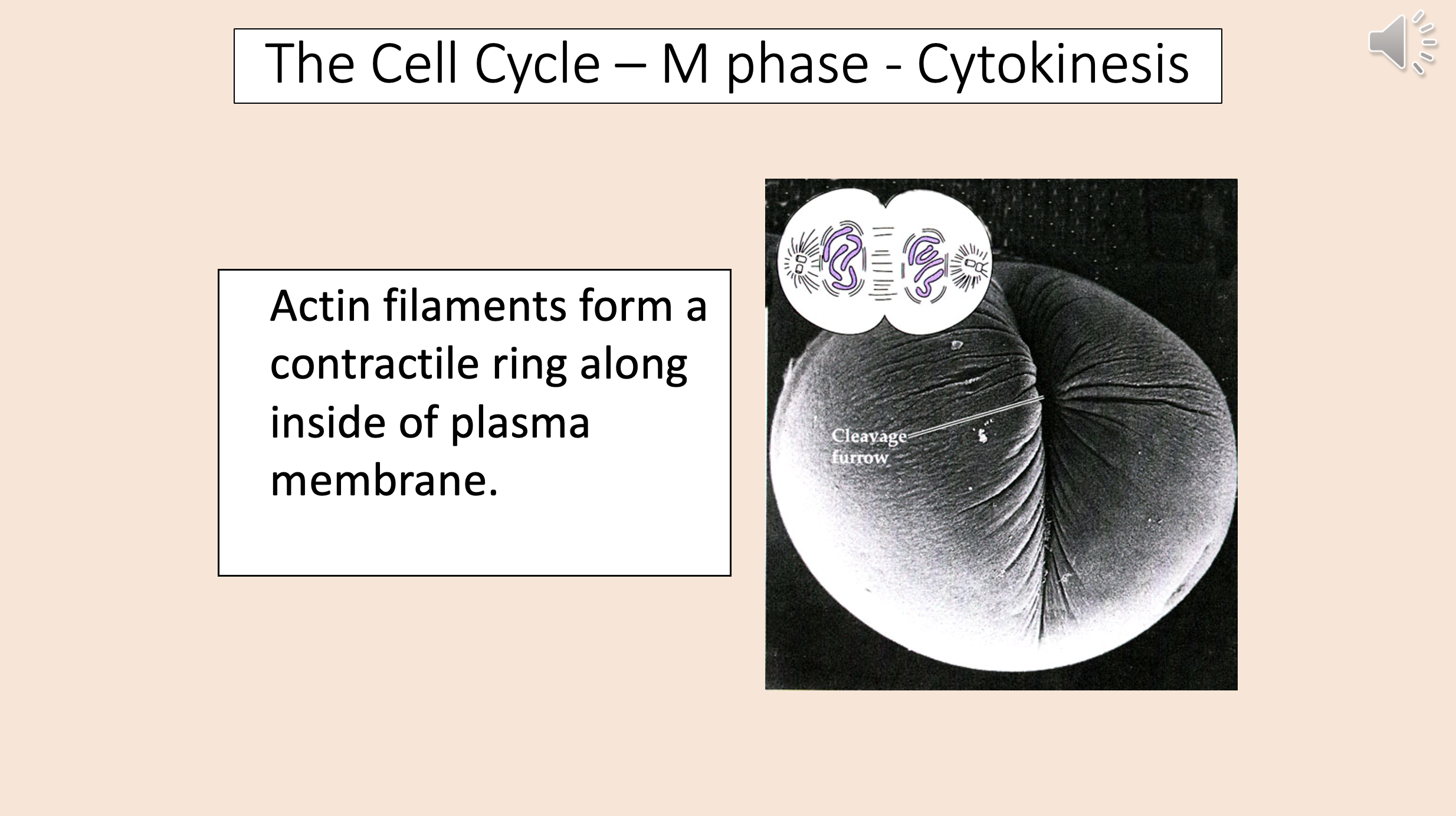
Actin filaments form a contractile ring along inside of plasma membrane.
End result of cell cycle:.
2 genetically identical daughter cells.
cytokinesis
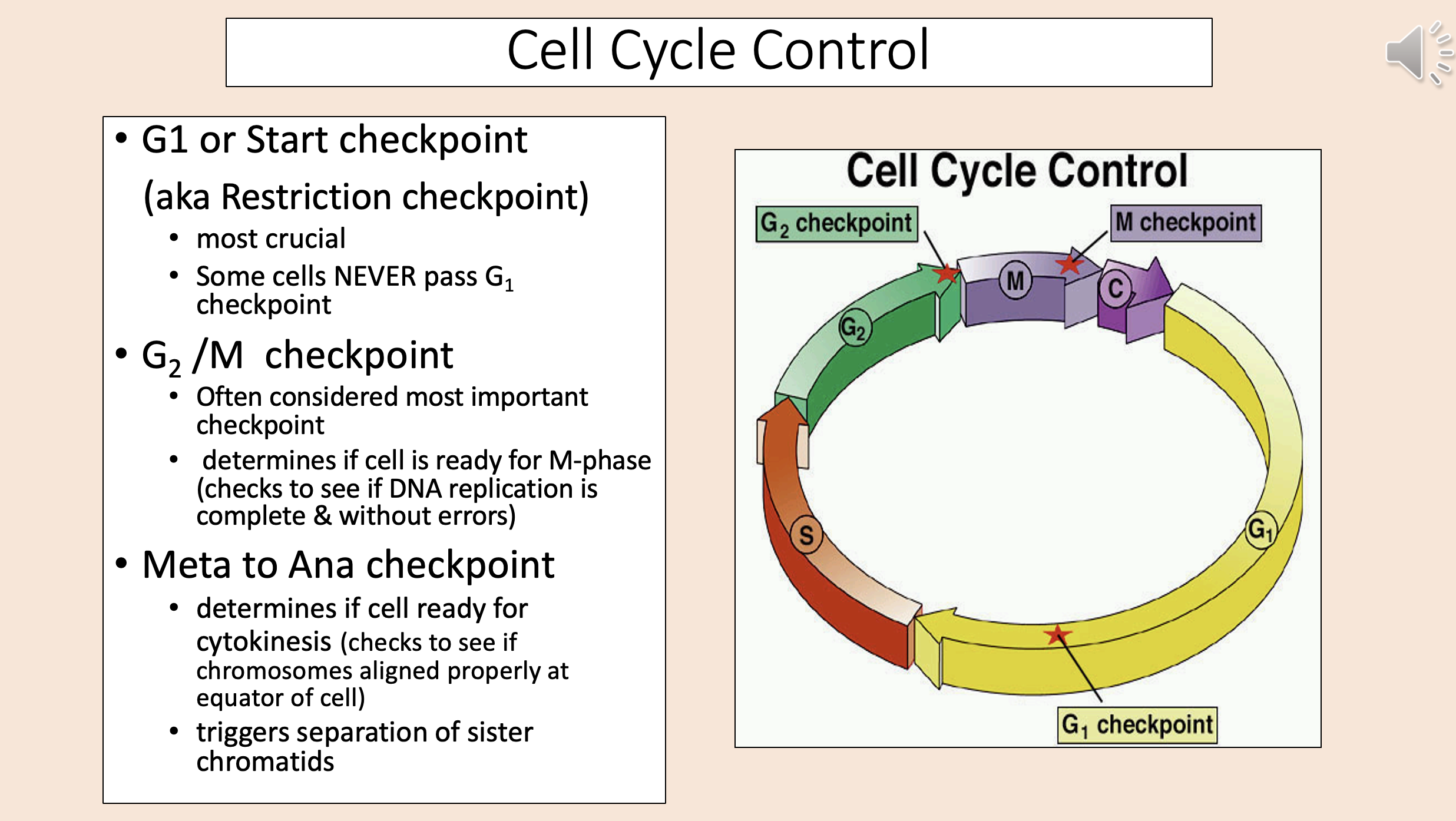
aka Restriction checkpoint
G1
start checkpoint
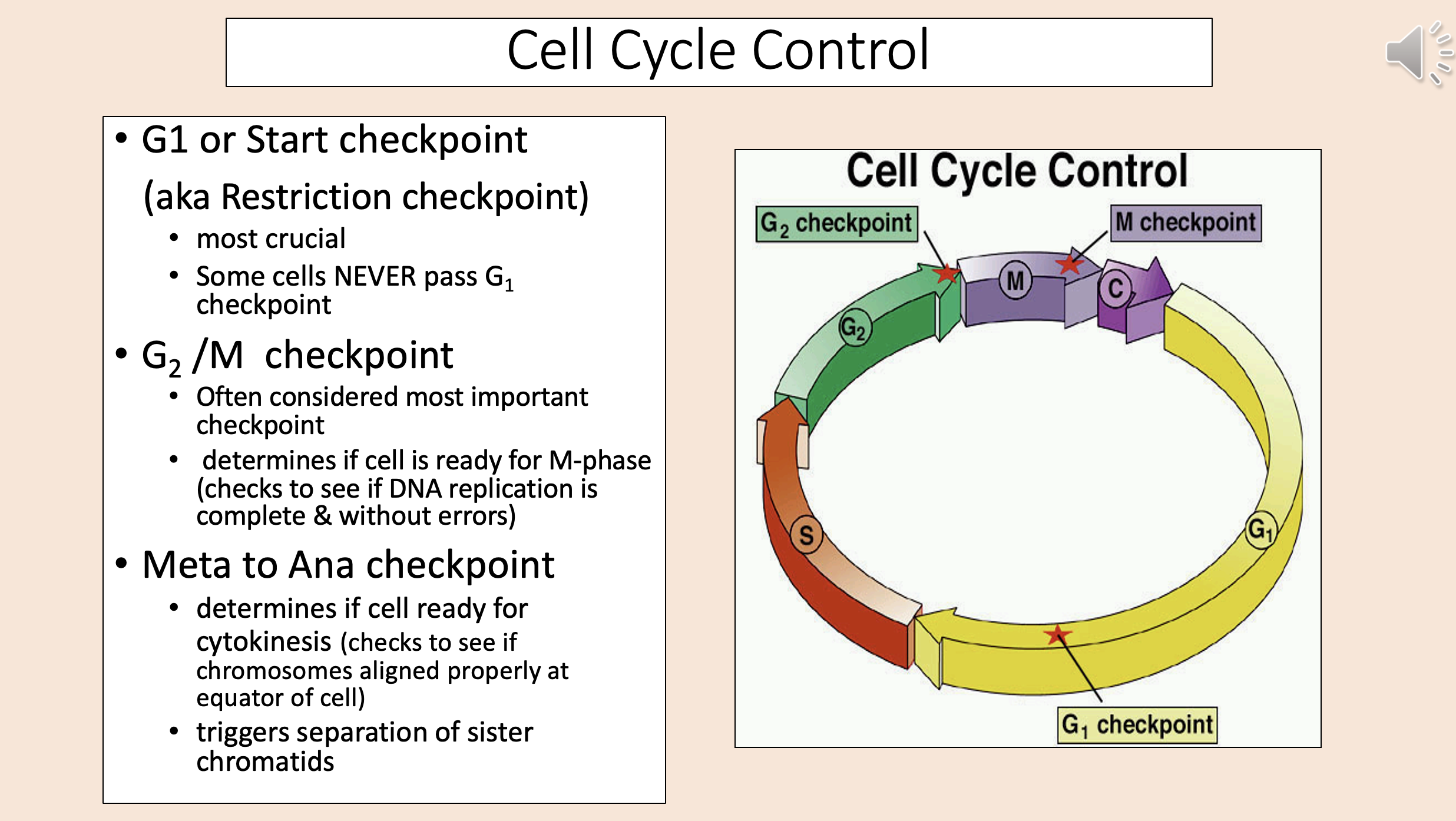
Most crucial.
Some cells NEVER pass G1 checkpoint.
G1
start checkpoint
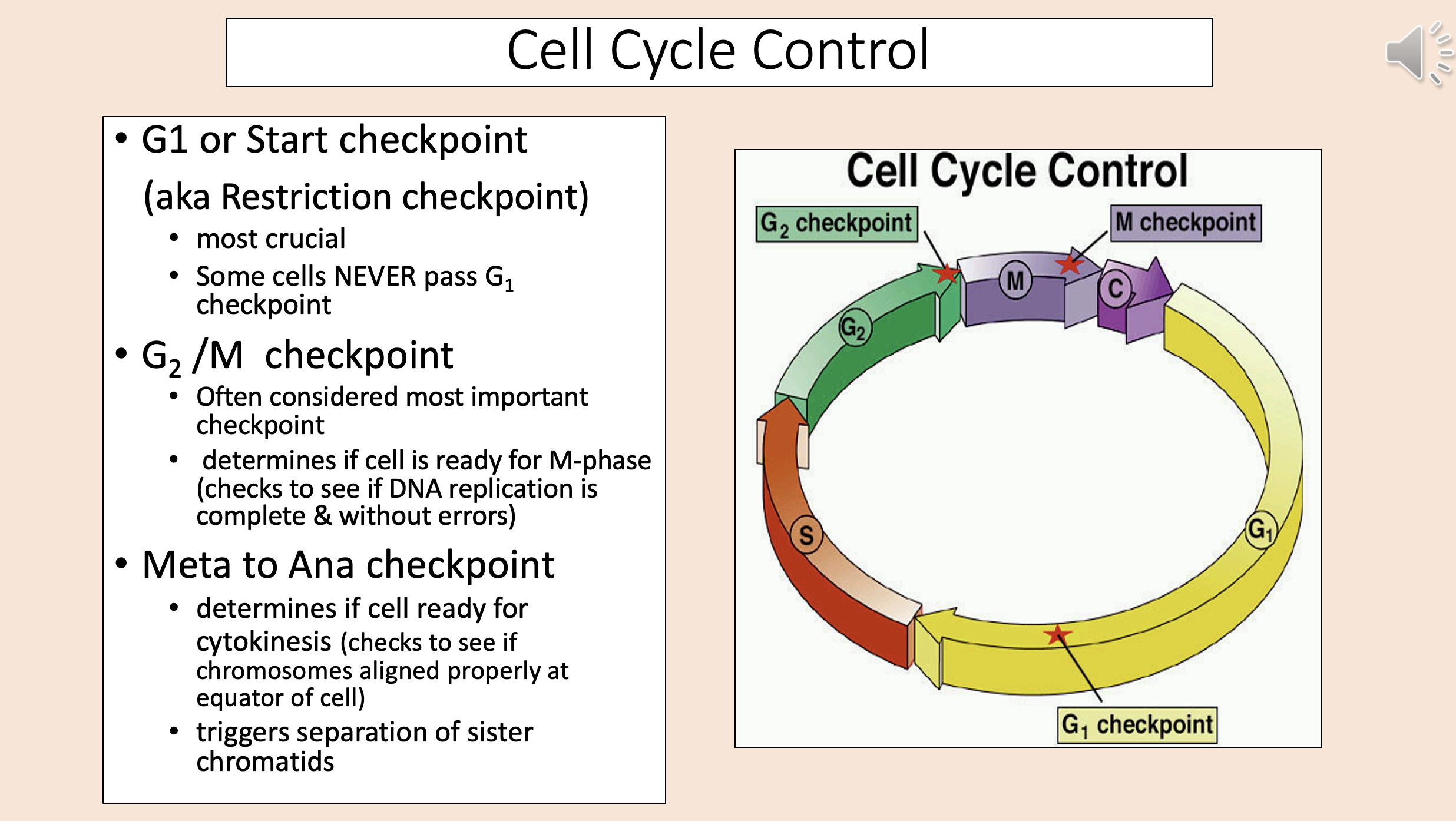
Often considered most important checkpoint.
Determines if cell is ready for M-phase (checks to see if DNA replication is complete & without errors).
G2 M checkpoint
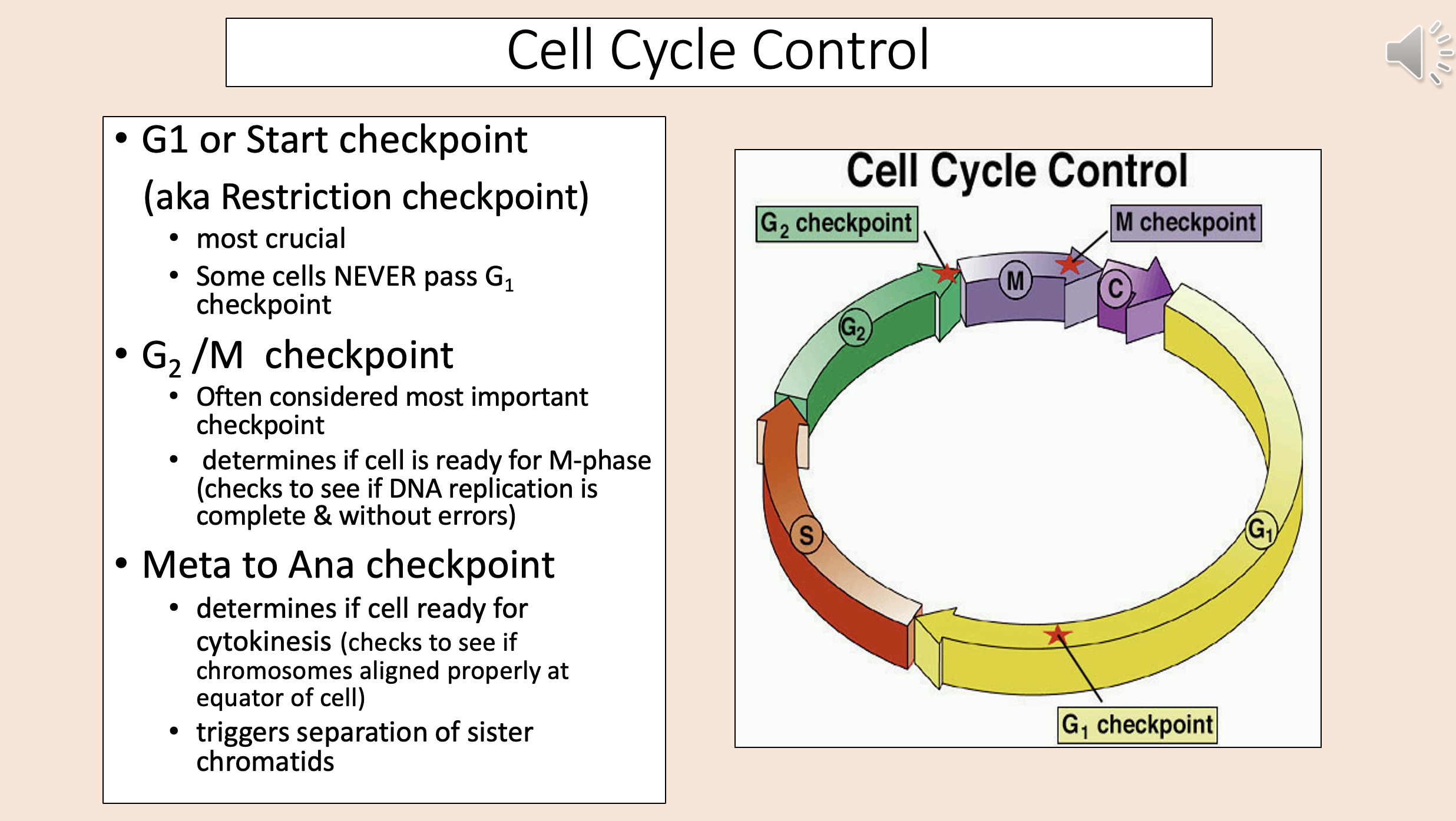
Determines if cell ready for cytokinesis (checks to see if chromosomes aligned properly at equator of cell).
Triggers separation of sister chromatids.
meta to ana checkpoint
Group of proteins that control progression through cell cycle; activate enzymes called CDKs via phosphorylation
cyclins
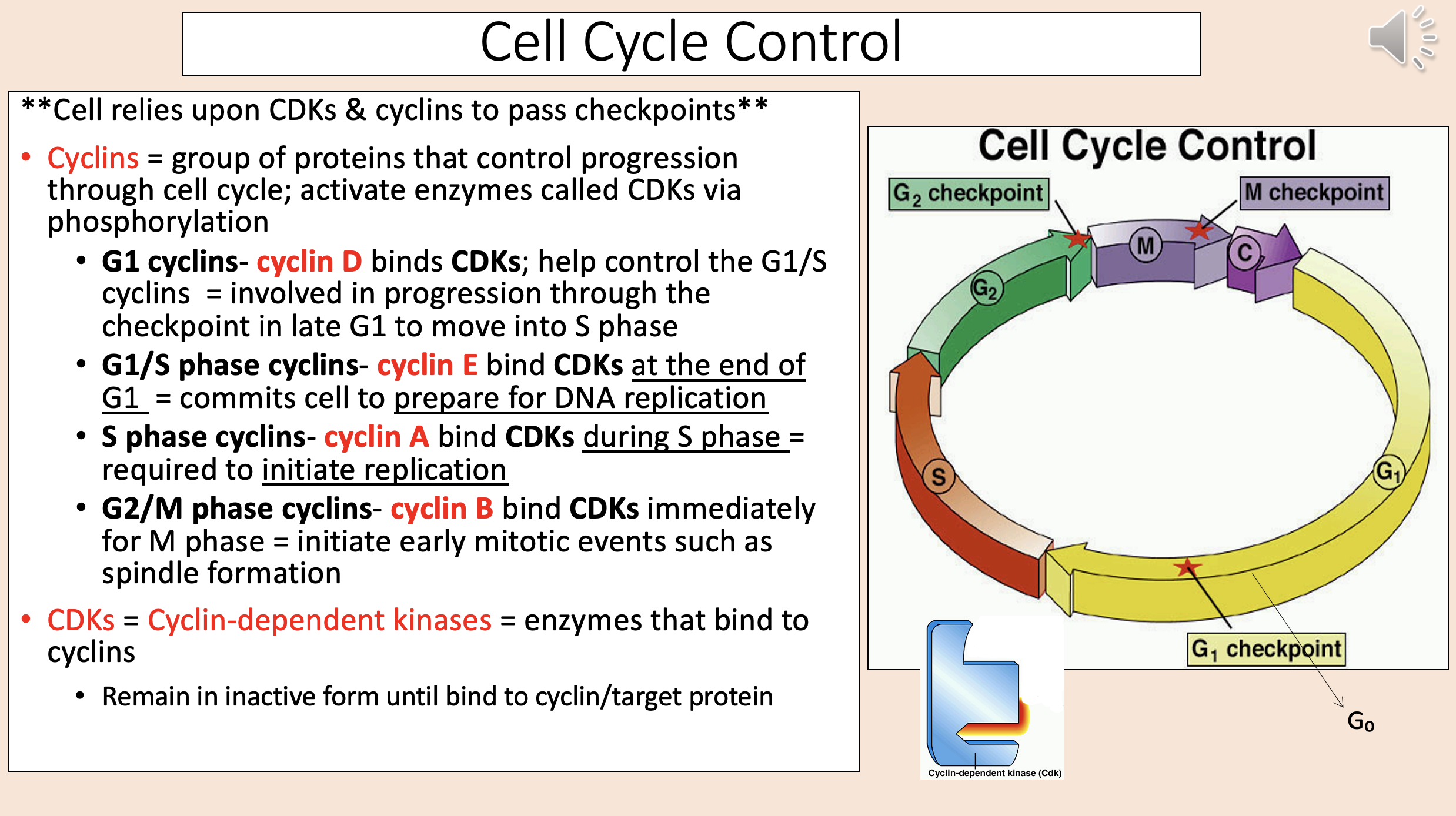
Binds CDKs; help control the G1/S cyclins.
Involved in progression through the checkpoint in late G1 to move into S phase.
G1 cyclins
cyclin D
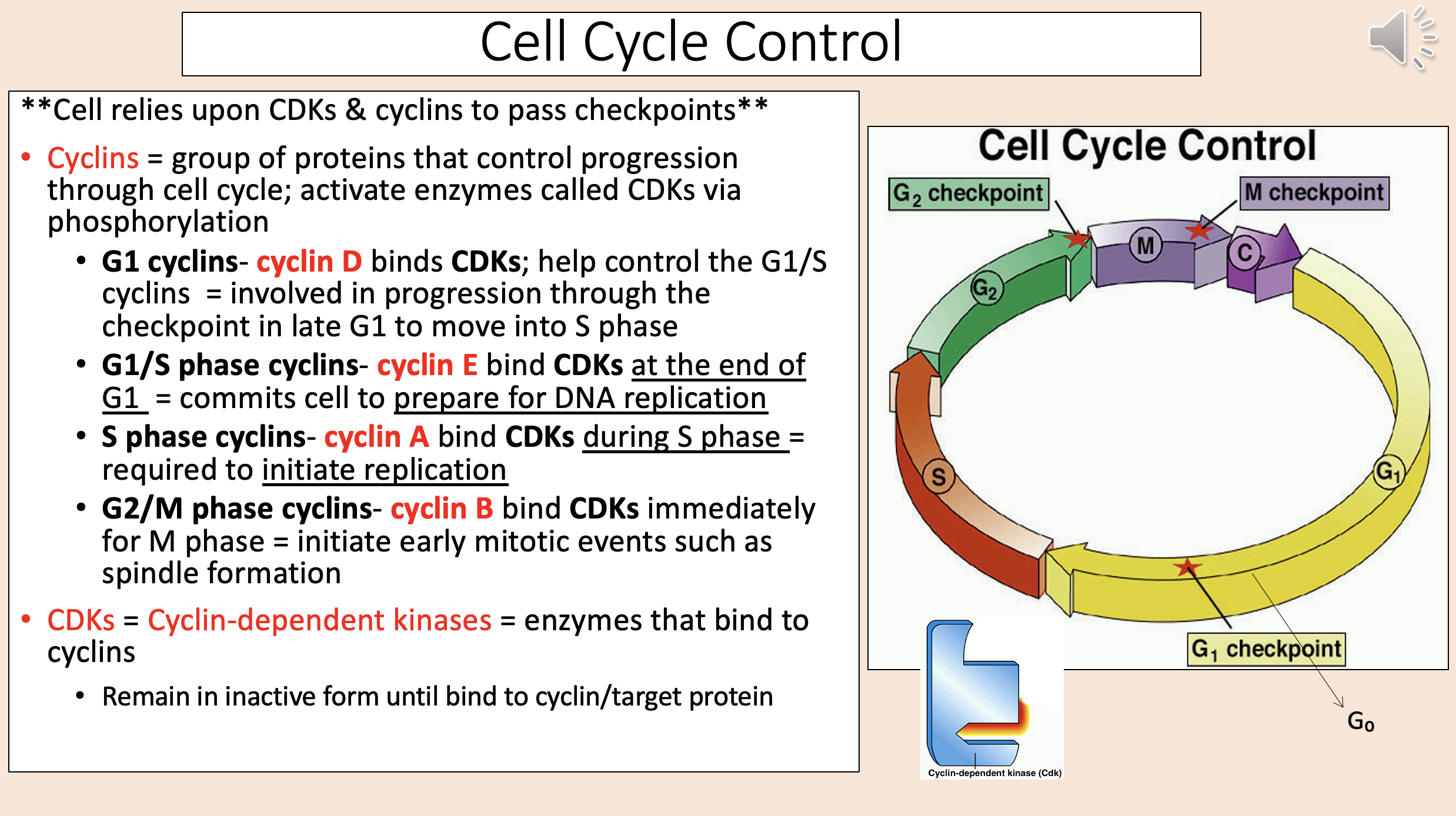
Bind CDKs at the end of G1.
Commits cell to prepare for DNA replication.
G1 S phase cyclins
cyclin E
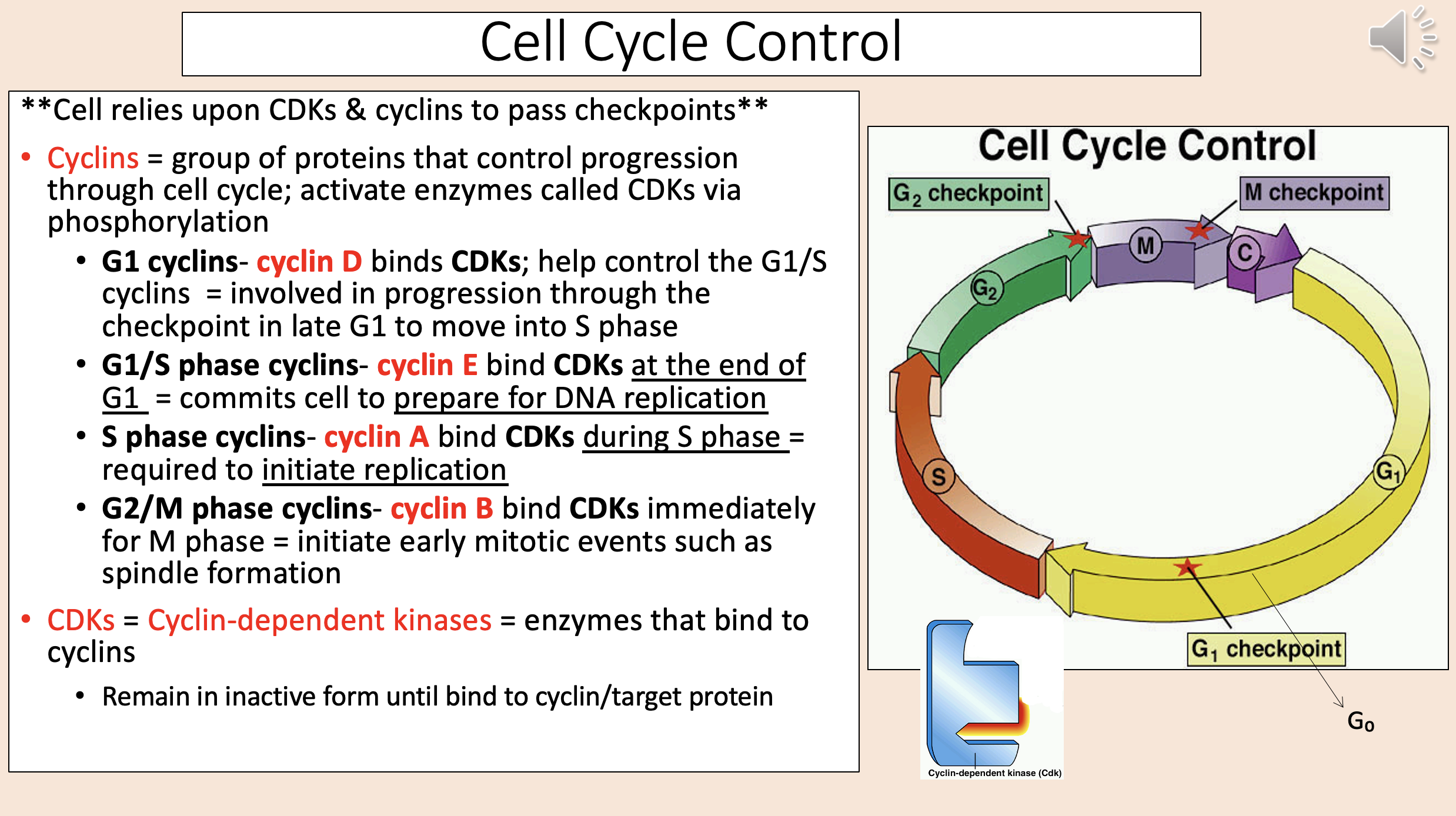
Bind CDKs during S phase.
Required to initiate replication.
S phase cyclins
cyclin A
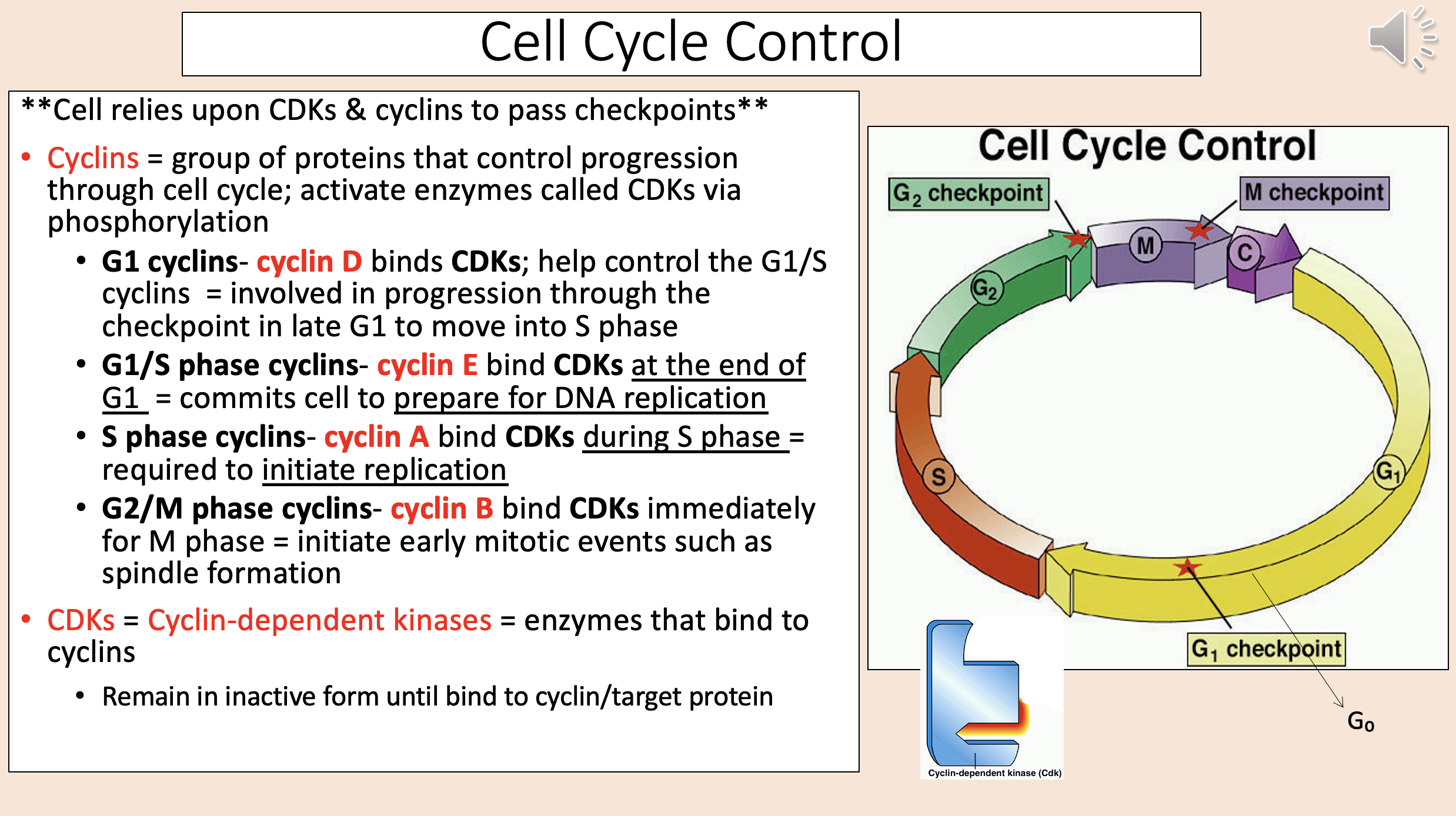
Bind CDKs immediately for M phase.
Initiate early mitotic events such as spindle formation.
G2 M phase cyclins
cyclin B
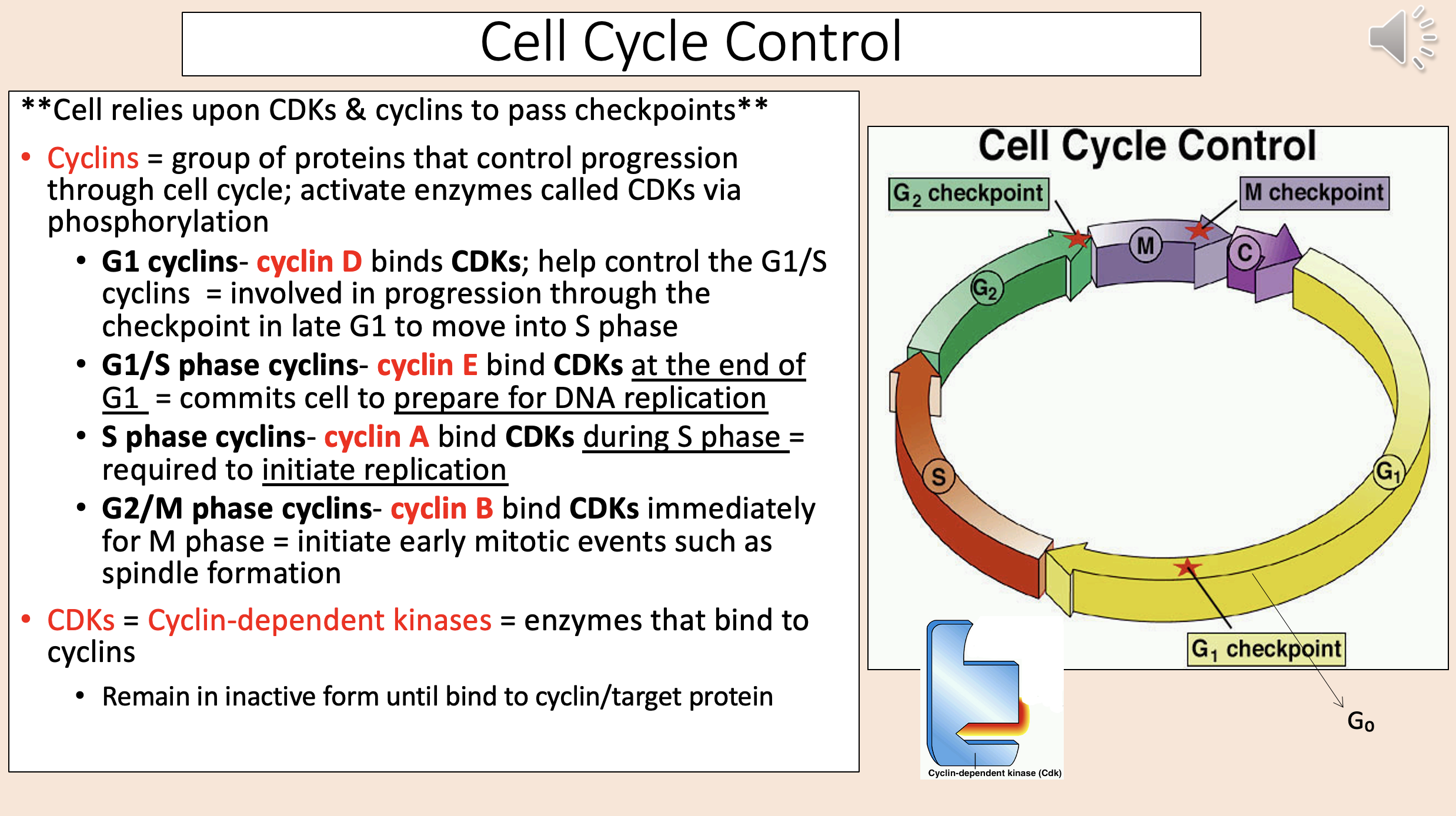
Enzymes that bind to cyclins
CDKs = Cyclin-dependent kinases.
Process of increasing cell numbers by mitotic cell divisions.
Regulated so that the cells produced = cells dying/shed.
Rate varies among the >200 cell types.
cell proliferation
Detailed process by which proliferating cells become specialized cells.
The new cells formed acquire the structure & function of those they are replacing!
cell differentiation
A form of programmed cell death.
Eliminates senescent cells, cells with DNA damage, unwanted cells.
apoptosis

Disorder of abnormal cell proliferation & differentiation.
Division/growth lacks normal regulatory controls over the cell cycle.
cancer

The growth/mass associated with cancer
neoplasm

A swelling that can be caused by numerous conditions
tumor

Tumor Two classifications:
benign
malignant

Well-differentiated; resemble cells of the healthy tissue/organ.
Slow; often comes to standstill.
Expands WITHOUT invading neighboring tissues; usually encapsulated.
Does NOT spread/infiltrate/invade.
Usually does NOT cause death.
benign

Undifferentiated with anaplasia & atypical structure; little resemblance to healthy tissue/organ.
Variable; often rapid.
Invasive – infiltrates surrounding tissues.
Spreads to other areas of body – gains access to blood/lymph channels to spread.
Often causes death.
malignant

Loss of differentiation/characteristics of mature cells
anaplasia

Variation in size & shape of cells (b/c of loss of differentiation).
Nuclei, nucleoli, and chromatin are even abnormal.
The degree of pleomorphism is used to grade neoplasms on a scale of 1-4.
pleomorphism

Well-differentiated/minimal anaplasia
1

Poorly differentiated/marked anaplasia
4

Mutation phenotypes/high rate of mutations in cells.
Often see aneuploidy, deletions, insertions, point mutations, and amplifications (aka chromosome errors).
genetic instability

Can divide without the influence of growth factors (molecules that regulate cell division)
growth factor independence
aka contact inhibition
loss of cell density-dependent inhibition

Most cells cease dividing when the population reaches a certain density – not cancer cells
loss of cell density-dependent inhibition

Most cells use cadherins to link to/stick to each other & to link intracellularly to the cytoskeleton with the aid of other proteins like catenins.
Amounts of E-cadherin is reduced in cancer cells.
ß-catenin accumulates inside cancer cells.
HYPOTHESIS = in the absence of cadherin, the ß-catenin binds to another protein (actinin-4) and that is what shuts off the adhesion process and allows cancer cells to shed/metastasize.
loss of cell cohesiveness adhesion

Can remain functional without anchoring to other cells or the extracellular matrix
anchorage dependence

Abnormal cytoskeletal components (actin, intermediate filaments, microtubules)
cytoskeletal changes

Cancer cells express different several surface molecules (antigens).
They often are like those expressed during embryonic/fetal development instead of mature/differentiated normal cells.
antigen expression

Cancer cells are immortal – unlimited divisions so unlimited life span!
Normal cells have a limited number of divisions before they senesce.
Telomeres.
life span

Short, repetitive sequences on ends of chromosome arms.
They shorten with each cell division; eventually chromosomes reach a critically short length & can no longer replicate = loss of cell division ability.
telomeres

Enzyme that prevents telomere shortening; levels are high in cancerous cells.
Molecular structures that cap the ends of chromosomes.
telomeres

The chromosomes become damaged and genetically unstable to the point that the cells can’t divide any more — a state called senescence.
The cells trigger apoptosis, ending the life of the cell.
after number of divisions

Direct invasion & extension.
Seeding of cancer cells in body cavities.
Metastatic spread through vascular/lymphatic pathways.
spread of cancer three pathways

Cancer cells secrete enzymes that break down proteins & allow for infiltration of neighboring cells/tissue
direct invasion extension

A tumor sheds cells into a cavity (ex: peritoneal, pleural, pericardial, joints).
Often occurs after surgical removal of a cancer.
seeding of cancer cells in body cavities

Sentinel node.
Hematologic spread is often related to the vascular drainage of the primary tumor.
metastatic spread through vascular lymphatic pathways

The initial lymph node to which a primary tumor drains
sentinel node
