Geriatric Considerations
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
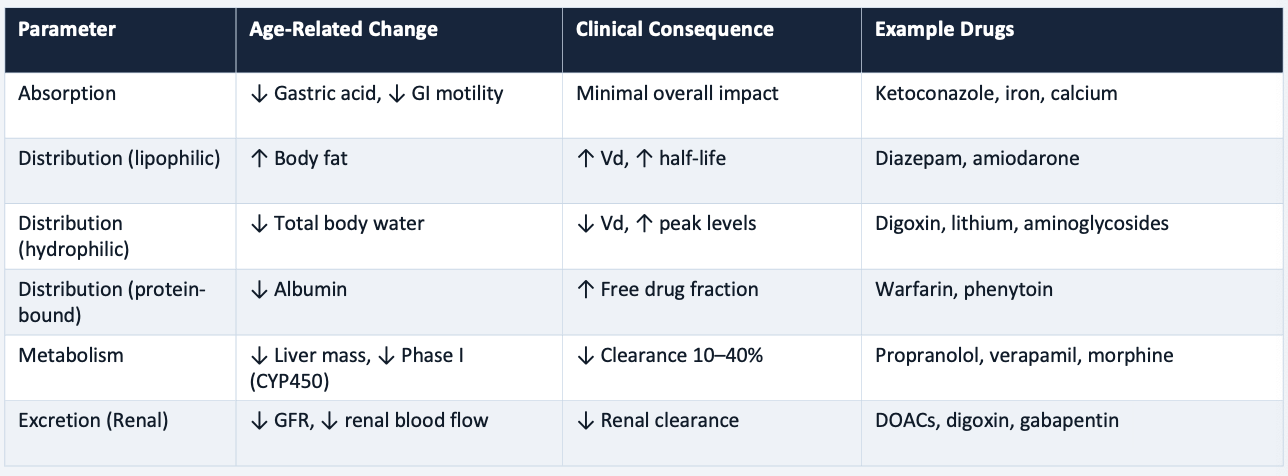
Absorption Changes in Elderly Individuals
Decreased gastric distribution → increase gastric pH
Delayed gastric emptying and decreased GI motility
Decrease splanchnic blood flow
Impact → minimal (most drugs still absorbed well)
Exception: ketoconazole, iron, calcium
Distribution Changes in Elderly Individuals
Increased body fat by 20-40% → lipophilic drugs (diazepam, amiodarone) have prolonged half life (increased Vd)
Decreased total body water by 10-15% → hydrophilic drugs (digoxin, lithium) have increased peak levels (decreased Vd)
Decreased plasma albumin → protein bound drugs (warfarin, phenytoin) have increased free drug fraction
Highly Protein Bound Drugs
Have increased risk with albumin
Warfarin
Phenytoin
Heparin
Amiodarone
Furosemide
Most statins (except pravastatin)
PEARL: decreased albumin → increased free drug → enhanced effect and toxicity risk
Hepatic Metabolism in Elderly Individuals
↓ liver mass (~20–30% by age 80)
↓ hepatic blood flow (20–50%)
↓ phase I metabolism (CYP450): most clinically significant
Phase II (conjugation) relatively preserved
Affected drugs:
Propranolol
Diltiazem
Verapamil
Morphine
Lidocaine
No lab test to quantify hepatic capacity — use clinical judgment
Renal Excretion in Elderly Individuals
MOST CRITICAL
Progressive ↓ GFR (~0.9%/yr after age 20)
↓ renal blood flow + ↓ tubular secretion
⚠ Serum creatinine is UNRELIABLE — low muscle mass masks reduced GFR
Always use validated eGFR equations for dosing decisions
High Risk Renally Cleared Drugs that Require Dose Adjustments
Anticoagulants:
DOACs (apixaban, rivaroxaban, dabigatran)
Enoxaparin
Antibiotics:
Aminoglycosides
Vancomycin
Fluoroquinolones
CV:
Digoxin
Atenolol
Sotalol
ACE inhibitors
Analgesics:
Morphine (M6G)
Gabapentin
Pregabalin
Others:
Metformin
Lithium
Pharmacokinetic Changes Summary
Increased Sensitivity
Benzodiazepines: sedation, falls, cognitive impairment
Opioids: ↑ mu-receptor sensitivity → respiratory depression, delirium
Anticoagulants: greater bleeding risk at therapeutic levels
Anti-diabetics: increased hypoglycemia risk
Psychotropics: anticholinergic effects, EPS
Decreased Sensitivity
Beta-adrenergic agonists/antagonists: reduced receptor density and responsiveness
Some antihypertensives may have blunted efficacy
Impaired Homeostatic Mechanisms
↓ baroreceptor sensitivity → orthostatic hypotension (vasodilators, diuretics, alpha-blockers)
↓ thirst response → dehydration with diuretics
↓ cognitive reserve → drug-induced delirium threshold lower
Double Jeapordy
Higher drug levels (PK) + Greater sensitivity (PD) = compounded risk in older adults
Polypharmacy: Geriatric Syndromes Exacerbated
Falls
Sedatives
Antihypertensives
Anticholinergics
Cognitive impairment/delirium
Anticholinergics
Benzodiazepines
Opioids
Urinary incontinence
Diuretics
Cholinesterase inhibitors
Functional decline and frailty
Non-adherence from complex regimens
Polypharmacy: Risk Factors
Multiple chronic conditions
Multiple prescribers/sub-specialists
Transitions of care
Long term care settings
The Prescribing Cascade
A new medication is prescribed to treat an adverse effect of an existing medication, misinterpreted as a new medical condition
Clinical pearl: before adding any new medication, always ask if the current symptom could be caused by the current medication

High Risk Drug-Drug Combinations
Anticoagulant + anti-platelet → major bleeding risk
Multiple CNS depressants (opioids + benzodiazepines) → respiratory depression
Multiple QT-prolonging agents → torsades de pointes
Warfarin + NSAID → GI bleed / INR elevation
ACE inhibitor + potassium-sparing diuretic → hyperkalemia
CYP inhibition/induction interactions compounded by already-reduced hepatic capacity
High Risk Drug-Drug Interactions
Anticholinergics in dementia → worsened cognition
NSAIDs in CKD → acute kidney injury
NSAIDs in heart failure → fluid retention, decompensation
Beta-blockers in severe COPD → bronchoconstriction
Opioids in fall history → increased fall risk
Benzodiazepines in dementia → delirium, falls
Fluoroquinolones in tendinopathy history → tendon rupture
AGS Beer Criteria
Applies to adults >= 65 y/o (except hospice/palliative care)
1 → avoid in most older adults
Long acting benzodiazepines
First generation antihistamines
Chronic NSAIDs
Sliding scale insulin
2 → avoid with specific diseases
Anticholinergics in dementia
TCAs in fall history
NSAIDs in CKD/HF
3 → use with caution
Aspirin for primary prevention > 70 y/o
Dabigatran in adults > 75 y/o
Tramadol
4 → drug-drug interactions
Opioid and benzodiazepines
Warfarin and NSAIDs
ACE-I and K-sparing diuretic
5 → renal dose adjustment
Medications requiring dose reduction or avoidance based on kidney function → DOACs, metformin
Beers Criteria: Anticholinergics
HIGH RISK
Examples: diphenhydramine, hydroxyzine, antispasmodics, TCAs
Effects:
Confusion
Urinary retention
Falls
Tachycardia
Beers Criteria: Benzodiazepines and Z-drugs
AVOID
Effects
Falls
Cognitive impairment
Dependence
Beers Criteria: antipsychotics
BLACK BOX
Avoid in dementia related behavioral symptoms → increased mortality
Exceptions
Quetiapine, clozapine, pimavanzserin for Parkinson psychosis
Beers Criteria: opioids
CAUTION
Avoid with concurrent BZD use
Avoid in fall history
Tramadol → high risk of SIADH/hyponatremia
Beers Criteria: PPIs
LIMIT
Avoid beyond 8 weeks without clear ongoing indication: can cause C. diff, hypomagnesemia, and/or fractures
Beers Criteria: dabigatran and rivaroxaban
CAUTION
Higher bleeding risk in older adults compared to apixaban
Consider renal function closely
STOPP Criteria
Screening Tool of Older Persons' Prescriptions
65 indicators by physiological system
Addresses DDIs, drug-disease, fall risk, duplication
More sensitive than Beers for detecting ADEs in some studies
European standard (Ireland)
STOPPFrail
Designed for frail older adults with limited life expectancy (≤1 year)
Guides de-prescribing of medications unlikely to provide benefit within remaining lifespan
START Criteria
Screening Tool to Alert to Right Treatment
Identifies beneficial medications that are OMITTED
Statins in CV disease; anticoagulation in A-fib
Osteoporosis treatment after fragility fracture → vaccinations
Addresses under-prescribing — the counterpart to overuse
MAI (Implicit Criteria)
Evaluates each medication across 10 domains
Indication
Effectiveness
Correct dosage
Directions
Drug-drug interactions
Drug-disease interactions
Duplication
Duration
Cost-effectiveness
Highly individualized and time-intensive
Dose Adjustment in Older Adults
Start low, go slow - but don’t stop too low
Begin at 25–50% of standard adult dose
Titrate slowly — longer intervals between changes
Monitor more frequently during titration
Ensure therapeutic targets are achieved
Renal dose adjustment
ALWAYS calculate eGFR before prescribing renally cleared drugs
DO NOT rely on serum creatinine alone
Narrow therapeutic index drugs (digoxin, lithium, aminoglycosides): consider cystatin C-based eGFR
Regular reassessment — eGFR may decline further with acute illness
Hepatic metabolism
No reliable lab test for hepatic metabolic capacity
Prefer Phase II drugs when possible
"LOT" drugs: Lorazepam, Oxazepam, Temazepam over diazepam
Therapeutic drug monitoring when available (digoxin, vancomycin, phenytoin) → check free levels
When to De-prescribe
Time to benefit exceeds life expectancy
Medication was prescribed for a resolved condition
Adverse effects outweigh benefits
No clear indication identifiable
Therapeutic duplication exists
Goals shifted toward comfort-focused care
Common Targets for De-prescribing
Proton pump inhibitors (>8 weeks without indication)
Benzodiazepines and Z-drugs
Antipsychotics in dementia without psychosis
Statins with limited life expectancy and no active CVD
Bis-phosphonates after 3–5 years (drug holiday)
Cholinesterase inhibitors in advanced dementia
Chronic NSAIDs
De-Prescribing Process
Compile complete medication list (including OTC, supplements, herbals)
Identify inappropriate/unnecessary meds (Beers/STOPP)
3. Prioritize (highest risk, least benefit first)
Plan taper when needed (BZDs, opioids, beta-blockers, SSRIs — do NOT stop abruptly)
Monitor for withdrawal/recurrence
Document & communicate
ARMOR Framework for Medication Review
A: Assess
Compile and assess all current medications, including OTC drugs, supplements, and herbals
R: Review
Review each medication for potential adverse effects, toxicity, and drug interactions
M: Minimize
Minimize the total number of medications
Eliminate those without clear indications
O: Optimize
Optimize doses for age related changes
Adjust schedules to simplify regimens
R: Reassess
Reassess at regular intervals → with each visit, acute illness, and care transition
Long Term Care Residents
Highest risk for polypharmacy — multiple chronic conditions and multiple prescribers
Federal regulations require regular medication regimen reviews
Antipsychotic reduction programs are mandated in many facilities
Multiple Sub-Specialites
Each specialist prescribes within their domain without full awareness of the complete medication list
PA's role as primary care provider/care coordinator is critical for reconciliation
Frail Older Adults/Limited Life Expectancy
STOPPFrail criteria guide de-prescribing
Shift from disease-oriented to symptom-oriented prescribing
Beers Criteria explicitly exclude hospice/end-of-life patients
Transitions of Care
Hospital admission, discharge, and transfers = high-risk periods for medication errors
Up to 50% of medication errors occur during transitions of care
Medication reconciliation at EVERY transition is essential
Clinical Pearls in Geriatric Pharmacology
PK changes (especially decreased renal clearance and altered distribution) requires systemic dose adjustment → never rely on serum creatinine alone
Pharmacodynamic sensitivity is increased for many drug classes, creating a double jeopardy effect compounding PK changes
Before adding and new medications, always ask could this symptom be an adverse effect of a current medication (avoid the prescribing cascade)
Apply Beers Criteria and STOPP/START at every medication review — but use clinical judgment as these are guides, not mandates
De-prescribing is a clinical skill requiring the same rigor as prescribing, including monitoring and follow-up
"Start low, go slow — but don't stop too low": individualize therapy to achieve therapeutic goals while minimizing risk
Medication reconciliation at every visit and every transition of care is a patient safety imperative