Crystalline vs. Amorphous Solid
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
According to the kinetic theory of matter; All matter is composed of particles that have a certain amount of energy to make them move at different speeds depending on?
temperature
It is the average kinetic energy of particles in
a system
Temperature
This state of matter, the intermolecular forces
between neighboring particles are
held together by strong covalent
or ionic bonding
Solid
These are noncompressible due to lack of
spaces between them
Solids
Solids have?
definite shape and volume
This state of matter can appear in two different forms;
Amorphous
Crystalline
Solids
These are solids in which the atoms,
ions, or molecules are
arranged in a definite
repeating pattern
Crystalline Solids
This type of solid is held together by uniform,
strong intermolecular forces.
Crystalline Solids
Galena, Quartza and Pyrite are all examples of?
Crystalline solids
It is the small repeating pattern in crystalline solids, made up of unique arrangement of atoms
Unit cell
It represents the structure of solids and the crystal structure can be built by piling the unit over and over
Unit cell
from the Greek words for
“without form”
this type of solid lacks the order found in
crystalline solids.
Amorphous Solids
structures of this type of solid at the atomic level is similar to the structures of liquids
Amorphous Solids
atoms, ions, or molecules
have little freedom to move
unlike in liquids, and they do not have well-defined shapes of a crystal. State which type of solid is this?
Amorphous Solids
examples: obsidian (volcanic
glass) and rubber
Amorphous Solids
It is the temperature at which a solid
loses it definite shape and is
converted to a liquid
Melting Point
It is the temperature at which liquid
changes to solid
Freezing Point
at this temperature, solid and liquid forms of a
substance are in equilibrium with each other
Melting/Freezing Point
These type of solids have a precise melting point.
crystalline solids
These types of solids melt over a wide range of temperature.
amorphous solids
molecules with a stronger interaction has a?
higher melting point
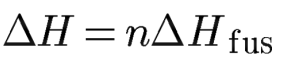
It is the quantity of heat necessary to melt a solid
Heat of Fusion
solids with stronger intermolecular forces: also has?
higher values of heat of fusion
What is the heat of fusion for crystalline solids?
heat of fusion is fixed and definite
What is the heat of fusion for amorphous solids
no precise value
It is the process of direct passage from solid to vapor phase, bypassing the liquid state
Sublimation
It is the reverse process of sublimation (vapor to solid
phase)
Deposition
It is the quantity of heat to convert a solid to vapor
Enthalpy of Sublimation
Ice exhibits an appreciable
sublimation pressure of?
Example:
● ice - solid with significant
sublimation pressure
● temperature may not rise above
0 OC, but snow may disappear.
(4.58 mm Hg).
It is the ability of solid to undergo
compressive stress without
breaking it
Malleability
these materials are highly malleable
○ shaped through forging,
rolling, extrusion, and
indenting.
Metals
It is the ability of solid to undergo
tensile stress and it can be can be measured.
Ductility
It describes the extent to which
the solid can be stretched
without fracture
Ductility
This type of metal is known to be the most
malleable and ductile.
Gold
Most of the time, malleability
and ductility properties ______
Some examples: silver and gold
coexist
It is the measurement of the ability of
atoms, molecules, or ions to transfer
electrons from one to another
Electrical Conductivity
These are solids that do not conduct electricity
electrical insulators
What type of chemical bonding has it easy for
electrons to move?
metallic bond
What type of chemical bonding has it hard to conduct electricity?
ionic or covalent bond
the measurement of the ability of atoms,
molecules, or ions to move and collide
with its neighboring particles.
Thermal Conductivity
What type of chemical bonding is a good heat conductor due to the nondirectional nature of the bonds.
Mettalic bond
What type of chemical bonding has a low thermal conductivity because of the rigidity between atoms?.
ionic/covalent bonds
These solids have a highly ordered
and repeating arrangement of particles,
forming a well-defined geometric
structure, such as in quartz and salt.
Crystalline solids
These solids lack a regular
arrangement of particles, resulting in a
disordered structure, as seen in
materials like glass and rubber.
Amorphous solids