A Level Chemistry 1.12 Acid and Bases
1/27
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
35.0 cm3 of 0.150 mol dm–3 aqueous sodium hydroxide are mixed with 20.0 cm3 of a 0.100 mol dm–3 solution of hydrochloric acid.
The temperature of the solution formed is 40 °C
Calculate the pH of the solution formed. Give your answer to 2 decimal places. (5)
nOH- = 5.25 × 10-3 and nH+ = 2.00 × 10-3 mol (1)
excess OH- = 3.25 × 10-3 mol (1)
[OH-]=3.25×10-3 /55.0×10-3 = 0.0591 mol dm-3 (1)
[H+] = 2.92×10-14 /0.0591=4.94×10-13 (1)
12.31 (1)
Suggest why a student doing an experiment to produce the curve in the figure above would add the sodium hydroxide solution dropwise around the equivalence point. (1)
As there is a large pH change (for a small addition of alkali) (1)
When sodium hydroxide solution is added to aqueous propanoic acid, the solution formed acts as a buffer when between 5 cm3 and 15 cm3 have been added.
Explain why the pH stays approximately constant during this part of the experiment. (2)
OH- reacts with propanoic acid OR reacts with H+(1)
ratio of [CH3CH2COOH] to [CH3CH2COO- ] remains almost constant OR Equilibrium for dissociation of CH3CH2COOH moves right to maintain [H+] /replace H+ (1)
Methyl orange and universal indicator are not suitable indicators for the titration of solutions of propanoic acid with sodium hydroxide.
State the reason why each indicator is not suitable (2)
Methyl orange - would not change colour at the equivalence point/pH range does not match rapid pH change (1)
Universal indicator - idea of range of colours during titration/no distinct colour change (at equivalence/end-point) (1)
Give an expression for Ka for propanoic acid (CH3CH2COOH). Use this expression to show that pH = pKa when half of the propanoic acid has reacted with sodium hydroxide.(3)
See image (1)
[CH3CH2COOH] = [CH3CH2COO- ] (1)
Ka=[H+] (1)
![<p>See image (1) </p><p>[CH<sub>3</sub>CH<sub>2</sub>COOH] = [CH<sub>3</sub>CH<sub>2</sub>COO<sup>-</sup> ] (1) </p><p>K<sub>a</sub>=[H<sup>+</sup>] (1) </p>](https://knowt-user-attachments.s3.amazonaws.com/bbdf9c02-1dc3-4d92-aee6-b2a87061f184.png)
The final mixture contains a saturated solution of Ca(OH)2 at 293 K
At 293 K
• the solubility of Ca(OH)2 in this solution is 0.400 g dm–3
• Kw = 6.80 × 10–15 mol2dm–6
Calculate the pH of this solution. Give your answer to two decimal places.(5)
0.400/74.1(=0.00540mol dm-3) (1)
[OH-]=0.00540×2=0.0108mol dm-3(1)
[H+]=6.80×10-15 /0.0108 (=6.30×10-13mol dm-3) (1)
pH=-log10[H+] (1)
-log10(6.30×10-13)(= 12.20)(1)
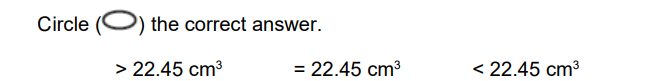
The pH of a barium hydroxide solution is lower at 50 °C than at 10 °C
At 50 °C a 25 cm3 sample of this barium hydroxide solution was neutralised by 22.45 cm3 of hydrochloric acid added from a burette.
Deduce the volume of this hydrochloric acid that should be added from a burette to neutralise another 25 cm ( sample of this barium hydroxide solution at 10 °C Explain your answer (2)
22.45 or same (1)
same [OH–] or amount/number of OH– ions (1)
State how a buffer solution can be made from solutions of potassium hydroxide and ethanoic acid.
Give an equation for the reaction between potassium hydroxide and ethanoic acid. State how this buffer solution resists changes in pH when a small amount of acid is added. (3)
add excess ethanoic acid to KOH (1)
KOH + CH3COOH ⟶ CH3COOK + H2O (1)
CH3COO– (from salt) reacts with (added) acid/H+ (1)
A buffer solution is made by adding 2.00 g of sodium hydroxide to 500 cm3 of 1.00 mol dm–3 ethanoic acid solution.
Calculate the pH of this buffer solution at 25 °C Give your answer to 2 decimal places. For ethanoic acid, Ka = 1.74 x 10–5 mol dm–3 at 25 °C (5)
n(NaOH) =2/40=0.05 mol and n(CH3COOH) =500×1/1000=0.5 mol (1)
(after adding NaOH) n(CH3COOH) = (0.50 − 0.05) = 0.45 (mol) (1)
n(CH3COO–) = (n NaOH) = 0.05 (mol) (1)
[H+]= (Kax[CH3COOH])/[CH3COO-] (1)
pH = 3.80 (1)
A student plans to titrate butanoic acid solution with a solution of ethylamine. Explain why this titration could not be done using an indicator.(2)
This is a weak acid and weak base/alkali titration (1)
pH change is too gradual/not sharp (at the equivalence point so colour change of indicator is difficult to judge) (1)
Ammonium chloride, when dissolved in water, can act as a weak acid as shown by the following equation.
NH4+(aq)+NH3(aq) + H+(aq)
The following figure shows a graph of data obtained by a student when a solution of sodium hydroxide was added to a solution of ammonium chloride. The pH of the reaction mixture was measured initially and after each addition of the sodium hydroxide solution.
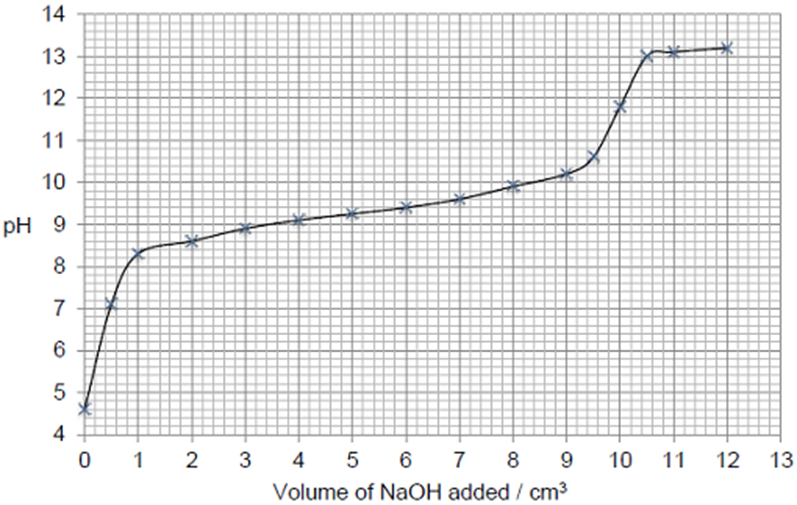
Use information from the curve in the figure above to explain why the end point of this reaction would be difficult to judge accurately using an indicator. (2)
The change in pH is gradual / not rapid at the end point (1)
An indicator would change colour over a range of volumes of sodium hydroxide (1)
The expression for the acid dissociation constant for aqueous ammonium ions is
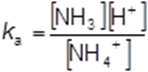
The initial concentration of the ammonium chloride solution was 2.00 mol dm−3.
pH= 4.6
Use the pH of this solution, before any sodium hydroxide had been added, to calculate a value for Ka (3)
At this point, [NH3] = [H+]
Therefore Ka =[H+]2 /[NH4+] (1)
[H+] = 10–4.6 = 2.51 × 10–5 (1)
This question is about acidic solutions.
The acid dissociation constant, Ka, for ethanoic acid is given by the expression
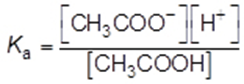
The value of Ka for ethanoic acid is 1.74 × 10−5 mol dm−3 at 25 °C
A buffer solution with a pH of 3.87 was prepared using ethanoic acid and sodium ethanoate. In the buffer solution, the concentration of ethanoate ions was 0.136 mol dm−3
Calculate the concentration of the ethanoic acid in the buffer solution.
Give your answer to three significant figures. (3)
[H+] = (10−3.87 =) 1.3489 × 10−4 (1)
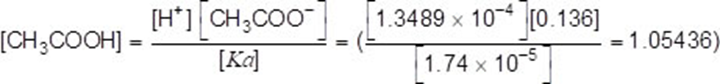
(1)
1.05-1.06(mol dm-3)
In a different buffer solution, the concentration of ethanoic acid was 0.260 mol dm−3 and the concentration of ethanoate ions was 0.121 mol dm−3
A 7.00 × 10−3 mol sample of sodium hydroxide was added to 500 cm3 of this buffer solution.
Calculate the pH of the buffer solution after the sodium hydroxide was added.
Ka = 1.74×10-5
Give your answer to two decimal places. (6)
moles of ethanoic acid = 0.130 (1)
moles sodium ethanoate = 0.0605 (1)
mol CH3COOH after addition = (0.130-0.007) =0.123 (1)
mol CH3COO− after addition = (0.0605+0.007) = 0.0675 (1)
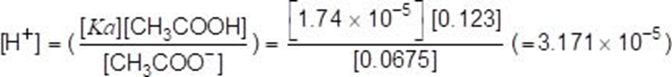
pH=4.50 (must be 2dp)
This question is about pH.
Pure water dissociates slightly.
H2O(I) ⇌ H+(aq) + OH–(aq) ΔH = +57 kJ mol–1
The equilibrium constant, Kc =
The ionic product of water, Kw = [H+][OH−]
Explain why [H2O] is not shown in the Kw expression. (1)
H2O is almost constant (1)
Table 1 shows how Kw varies with temperature.
Temperature / °C | Kw / mol2 dm–6 |
10 | 2.93 × 10−15 |
20 | 6.81 × 10−15 |
25 | 1.00 × 10−14 |
30 | 1.47 × 10−14 |
50 | 5.48 × 10−14 |
Explain why the value of Kw increases as the temperature increases (2)
Equilibrium is endothermic (in forward direction) (1)
Equilibrium shifts to the RHS to minimise/oppose temperature increase (1)
Suggest why the pH probe is washed with distilled water between each of the calibration measurements. (1)
Different solutions must not contaminate each other (1)
The calibrated pH meter is used to monitor the pH during a titration of hydrochloric acid with sodium hydroxide.
Explain why the volume of sodium hydroxide solution added between each pH measurement is smaller as the end point of the titration is approached. (1)
to avoid missing the end point (1)
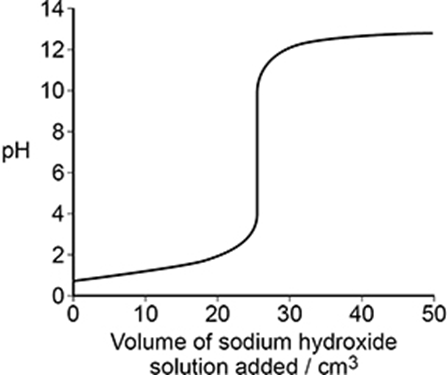
Table 2 shows data about some indicators.
Table 2
Indicator | pH range | Colour at low pH | Colour at high pH |
Bromocresol green | 3.8 – 5.4 | yellow | blue |
Phenol red | 6.8 – 8.4 | yellow | red |
Thymolphthalein | 9.3 – 10.5 | colourless | blue |
The student plans to do the titration again using one of the indicators in Table 2 to determine the end point.
State why all three of the indicators in Table 2 are suitable for this titration. (1)
All have a colour change within the vertical part of the titration (1)
A buffer solution with a pH of 4.50 is made by dissolving x g of sodium
propanoate (C2H5COONa) in a solution of propanoic acid. The final volume
of buffer solution is 500 cm3 and the final concentration of the propanoic
acid is 0.250 mol dm–3
Calculate x in g
For propanoic acid, Ka = 1.35 × 10–5 mol dm–3
(6)
[H+ ] = 10-4.5 = 3.16 x 10-5 (mol dm-3 ) (1)
[C2H5COO-]= (0.250×1.35×10-5) / 3.16×10-5 (1)
[C2H5COO-]=0.1068 (moldm-3 ) (1)
Mr sodium propanoate = 96 (1)
mass (sodium propanoate,x) =0.0534×96=5.13 (g)
Which statement about pH is correct?
A The pH of a weak base is independent of
temperature.
B At temperatures above 298 K, the pH of pure
water is less than 7.
C The pH of 2.0 mol dm–3 nitric acid is approximately 0.30
D The pH of 0.10 mol dm–3 sulfuric acid is greater than that of 0.10 mol dm–3 hydrochloric acid. (1)
B (1)
When temperature increases:
Kw increases
More H⁺ and OH⁻ are formed in pure water
A 0.10 mol dm–3 aqueous solution of an acid is added slowly to 25 cm3 of a 0.10
mol dm–3 aqueous solution of a base.
Which acid–base pair has the highest pH at the equivalence point?
A CH3COOH and NaOH
B CH3COOH and NH3
C HCl and NaOH
D HCl and NH3 (1)
A
For pure water at 40 °C, pH = 6.67
A student thought that the water was acidic.
Explain why the student was incorrect. (1)
[H+ ] = [OH– ] (1)
A student prepared a buffer solution by adding 0.0136 mol of a salt KX to
100 cm3 of a 0.500 mol dm–3 solution of a weak acid HX and mixing
thoroughly.
The student then added 3.00 × 10–4 mol of potassium hydroxide to the
buffer solution.
Calculate the pH of the buffer solution after adding the potassium
hydroxide.
For the weak acid HX at 25 °C the value of the acid dissociation constant,
Ka = 1.41 × 10–5 mol dm–3
Give your answer to two decimal places. (6)
Ka = [H+]]X]/[HX] (1)
amount of HX = 0.0500 mol (1)
amount of HX after addn of KOH = 0.05 − 3 × 10–4 = 0.0497 mol (1)
amount of KX after addn of KOH = 0.0136 + 3 × 10–4 = 0.0139 mol (1)
[H+]= (1.41×10-5 ×0.0497) /0.0139= 5.04(15)x10-5 (1)
pH=-log105.04(15)x10-5 =4.30 (1)
A buffer solution has a constant pH even when diluted.
Use a mathematical expression to explain this (1)
ratio [HX]/[X-] (1)
A student is required to make 250 cm3 of an aqueous solution that contains an accurately measured mass of sodium hydrogensulfate (NaHSO4). Describe the method that the student should use to make this solution. (4)
weigh solid and transfer using a method that Allows exact mass to be known (there should be two weighings, one of which could be zeroing, and method could be by difference or with washings or directly weighed into container) (1)
dissolve in water in suitable container (NOT in 250 cm3 of water) (1)
transfer with washings into 250 cm3 volumetric/graduated flask (1)
make up to mark /250 cm3 AND THEN shake / invert / mix (1)
A solution that contains 605 mg of NaHSO4 in 100 cm3 of solution has a pH of 1.72
Some sodium sulfate is dissolved in a sample of the solution from part (d). Explain why this increases the pH of the solution.(2)
(HSO4 – ⇌ SO42– + H+ ) equilibrium moves/shifts left (to counteract / remove increased [SO42-] (1)
so [H+ ] decreases (1)
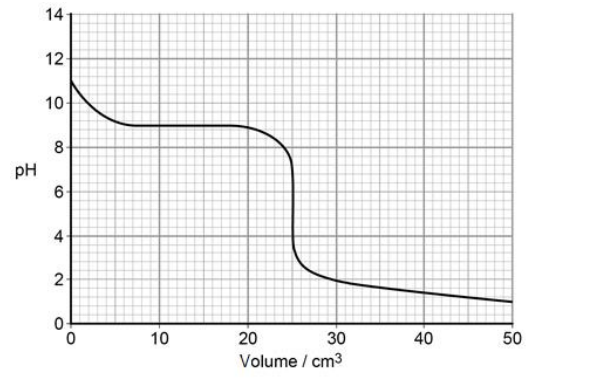
The graph was obtained from an experiment in which an acid was reacted with an alkali
Suggest briefly a practical procedure that a student could use to obtain data from which the curve in the graph could be plotted. (3)
Place a fixed volume of alkali in a flask or beaker (1)
Add acid in small portions from a burette (1)
Stir and use a pH meter to record the pH after each addition of acid (1)