Week 4
0.0(0)
Studied by 0 peopleCard Sorting
1/3
There's no tags or description
Looks like no tags are added yet.
Last updated 3:46 AM on 4/21/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
1
New cards
Dalton’s law
in a mixture of gases, the total pressure is equal to the sum of the partial pressures of each gas
2
New cards
Formulas to find the total pressure
Answer
IF ALL CONTAINERS HAVE IDENTICAL CONDITIONS
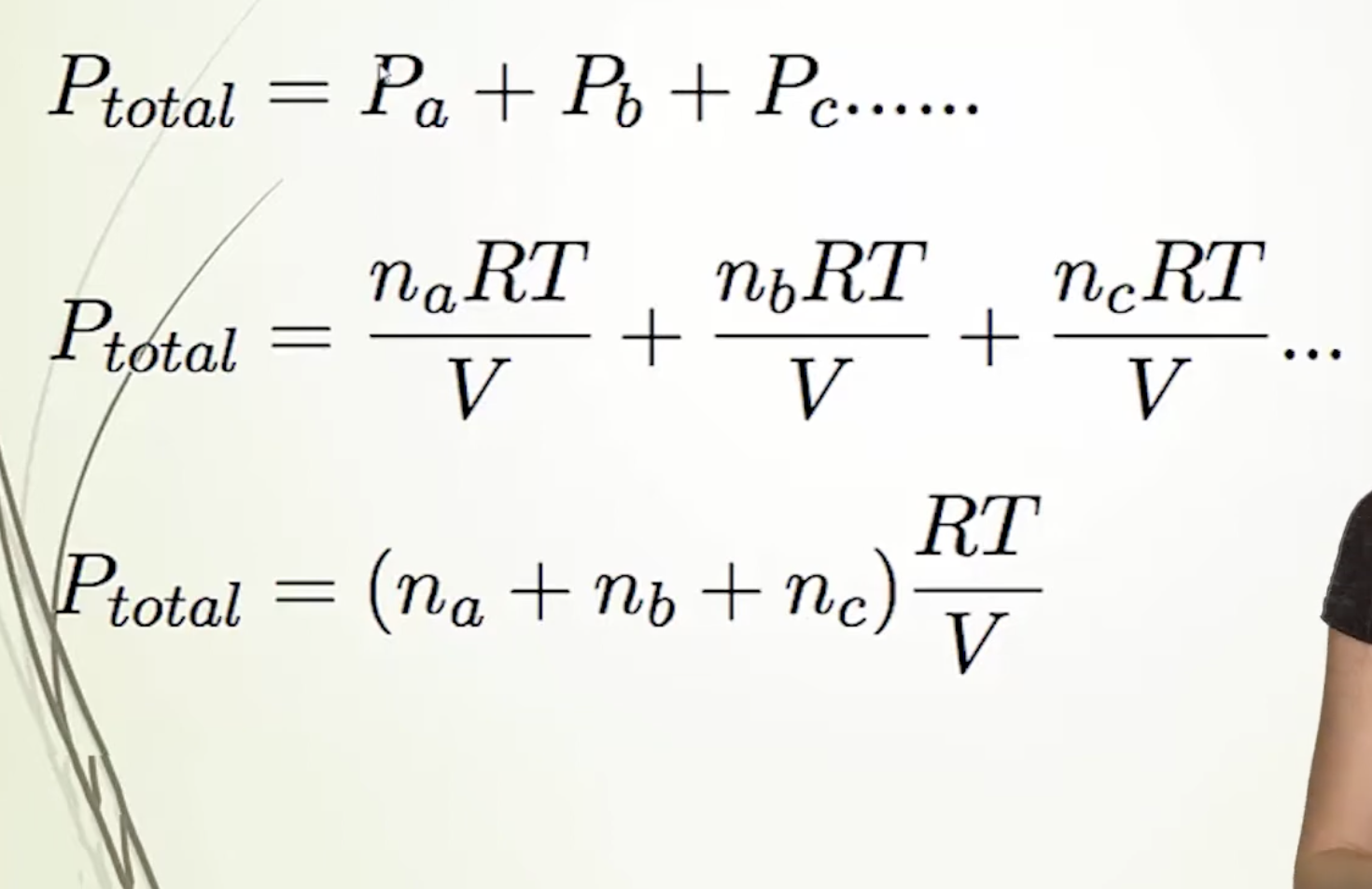
3
New cards
Partial pressure formula
Pa = Xa times Ptotal
(mol fraction of gas A times total pressure)
4
New cards
Mol fraction formula
Xa = na/ntotal
(number of mols of gas a/total number of mols)