All of Transition metals:
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Definition of a transition metal, and four main properties:
A transition metal forms a stable complex ion with a partially filled d sub-shell.
There are four main properties of transition metals:
They form complex ions
Formation of coloured ions
Variable oxidation state
Catalytic activity
How can transition metal absorb light, and appear colourful?
For a transition metal to absorb visible light, there must be room for one electron to be excited from one orbital to another orbital, and the difference between the orbitals energy must correspond to the frequency of visible light. And the transition metal, must have partially filled d-orbitals, if it has full or empty d orbitals then the solution is not colourful.
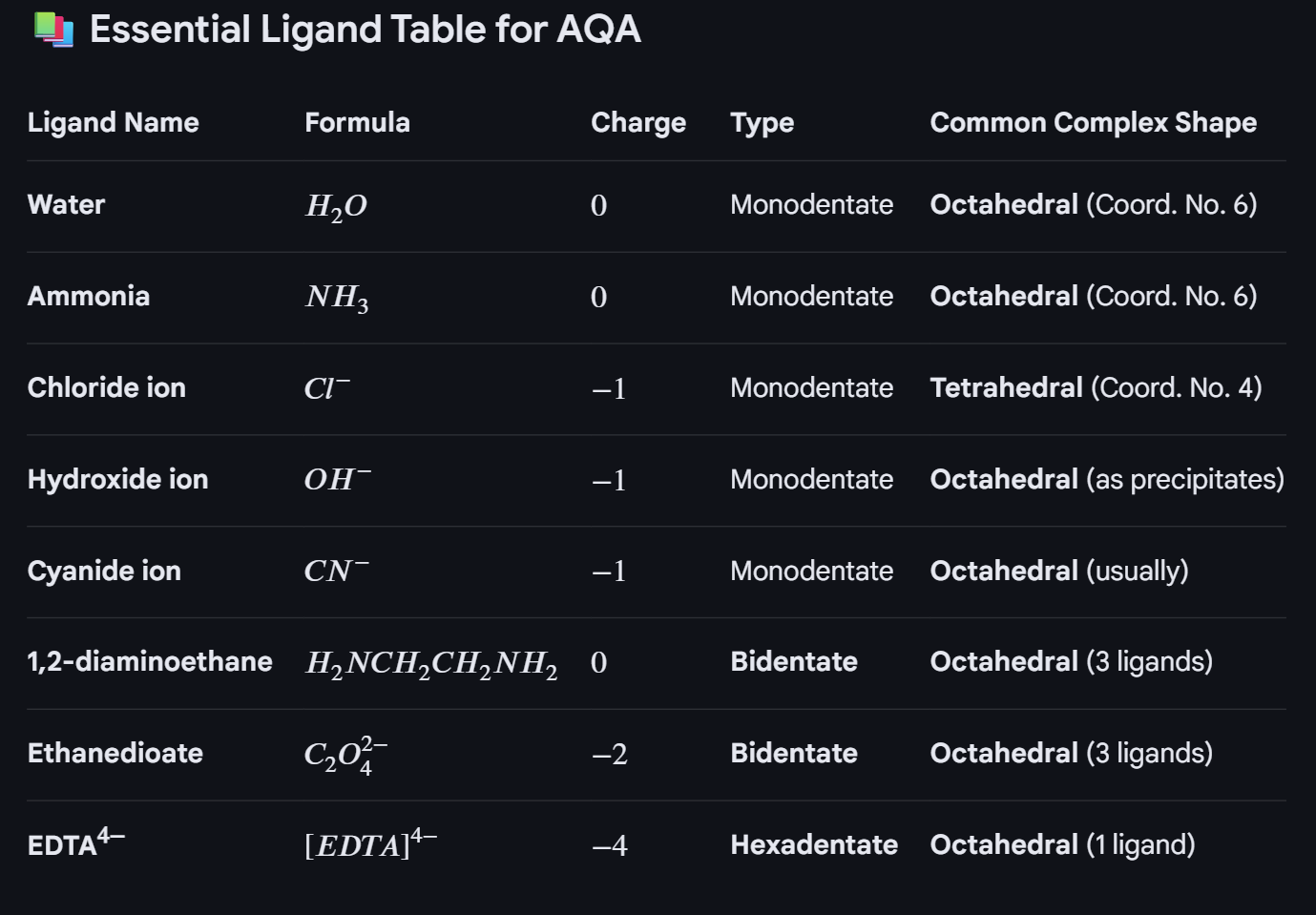
Define Ligand and coordination number and complex ion:
Ligand, is a molecule or ion with a lone pair, which forms a dative covalent bond (co-ordinate bond) with a transition metal.
Coordination number: the number of dative covalent bonds to each metal ion
A complex, is a central metal ion surrounded by ligands
Key ideas how to name transition metals:
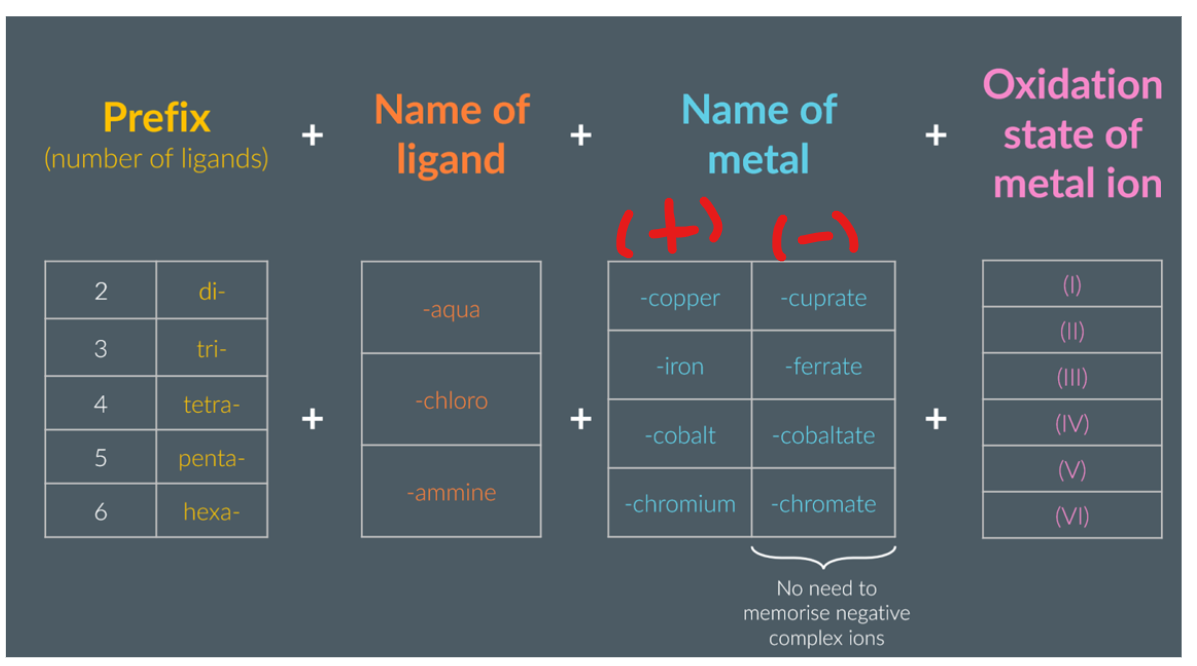
When there are more than one type of ligand, then you name the complex ion with the ligands in alphabetical order.
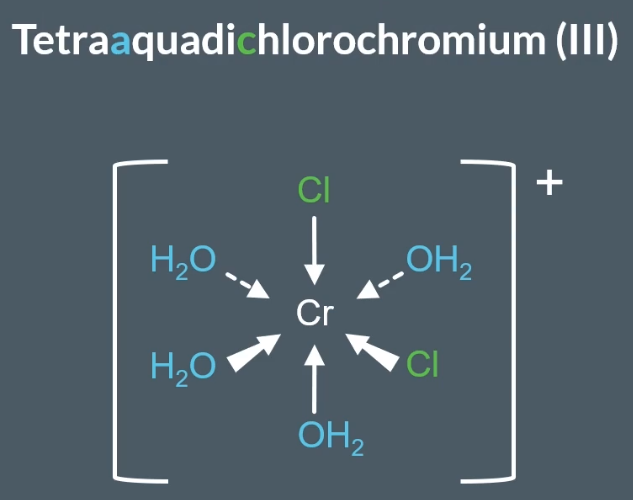
The key idea, is that the (III) OR (II) OR (I) represents the oxidation state of the metal ion
How to draw bonds on transition metals:
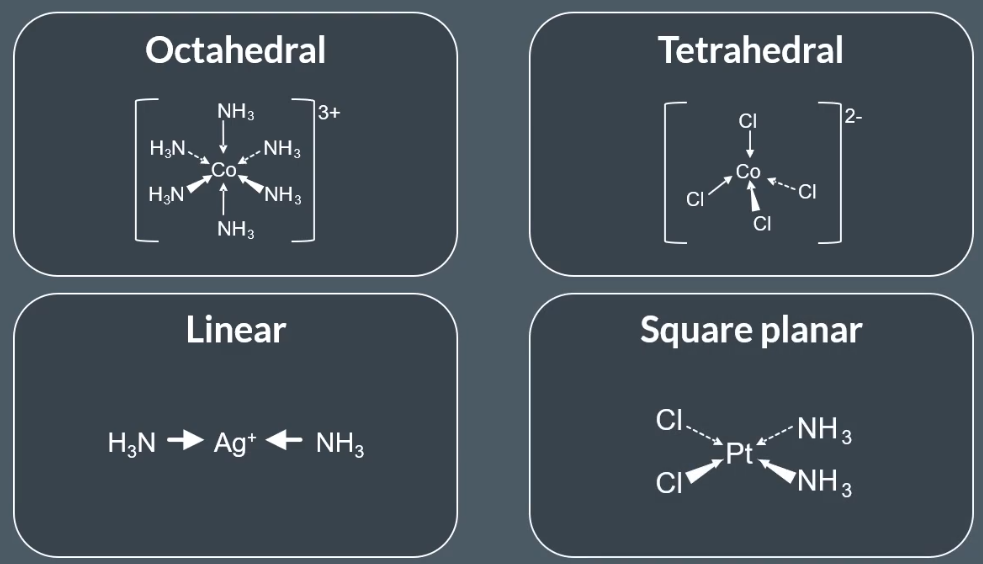
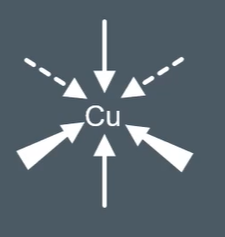
Solid line: The bonds are in the plane of the paper
Thick wedged line: The bonds are coming out of the paper towards you
Dotted line: The bonds are going into the paper away from you
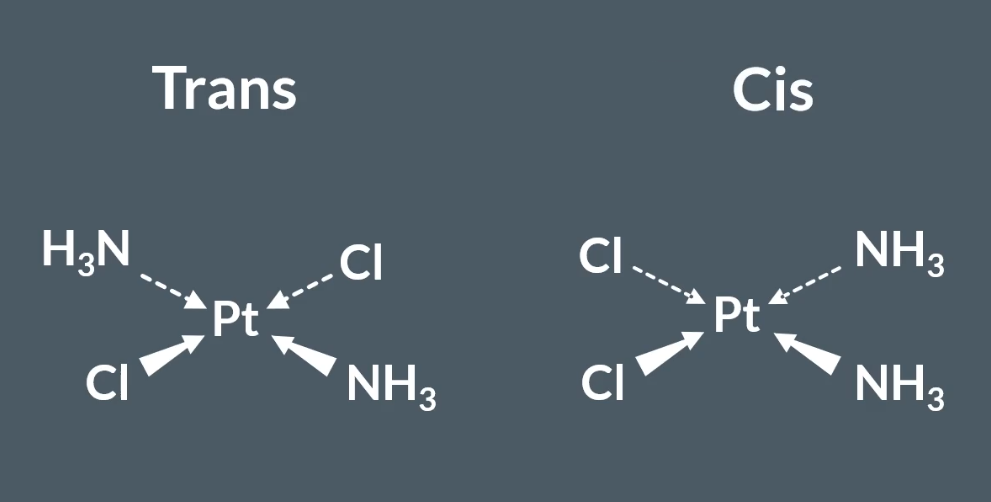
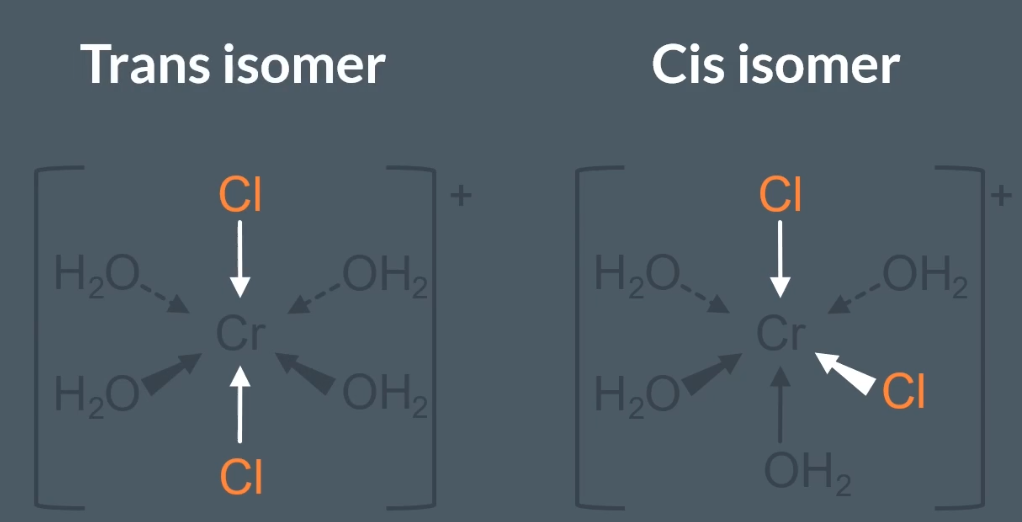
Sterioisomerism present in metal ion complexes:
The colour wheel, and its importance
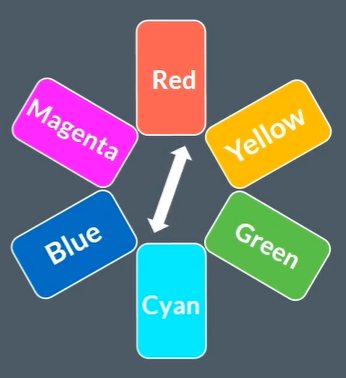
If one colour is absorbed, then the complementary colour is reflected, and thats what the colour of the solution is.
For example, if cyan colour is absorbed, then the rec colour is reflected, and that is the colour being seen.
The difference between ground state and excited state:
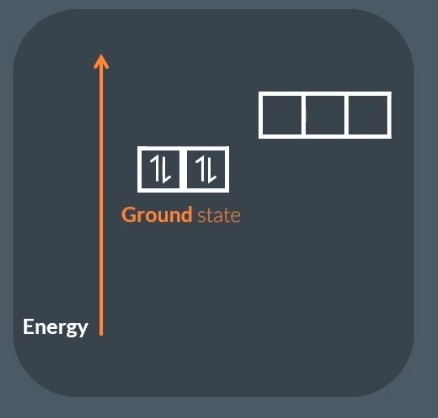
Ground state, where all the electrons in the atom occupy the lowest energy level.
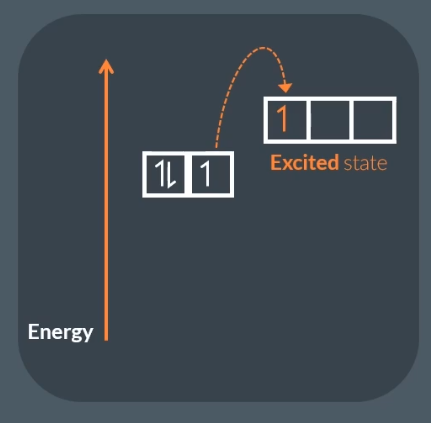
Excited state, where at least one of the electron occupies a sub shell of higher energy level.
The equation for calculating the energy difference between split d-orbitals, and how they appear colourful?
In a transition metal complex ion, the metal ligands interact with the metals d-orbitals, causing some of the orbitals to increase in energy level, and others to decrease in energy level. This creates a small gap in energy levels, corresponding to one of the wavelengths of visible light. So, light of that colour excites the electron to a higher energy d-orbital, and gets absorbed.
Key idea: They absorb some wavelengths of light while other wavelengths of light are absorbed or transmitted.
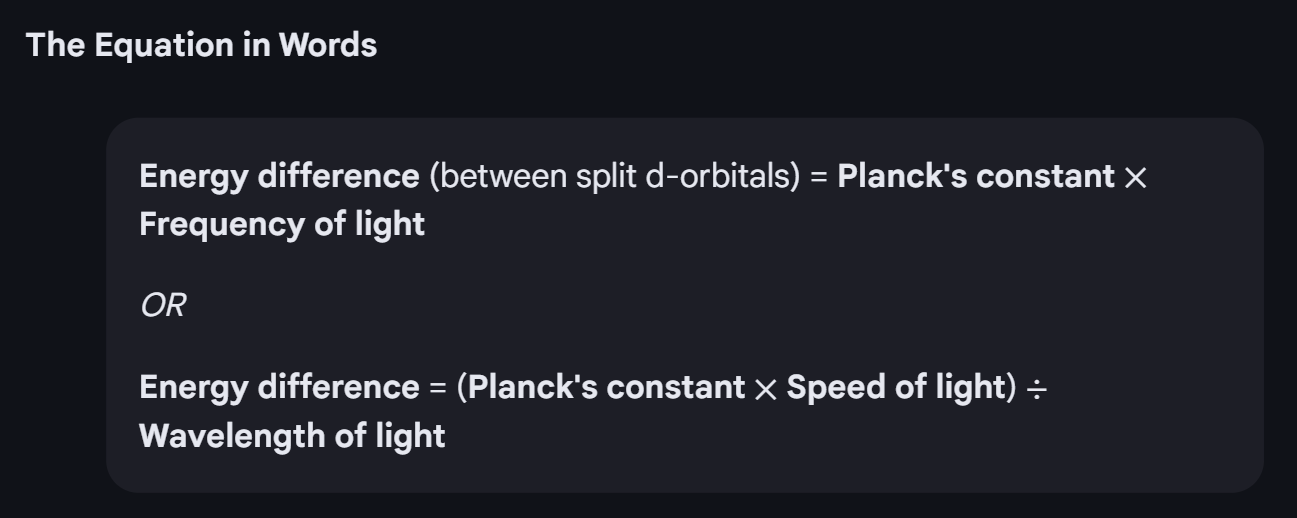
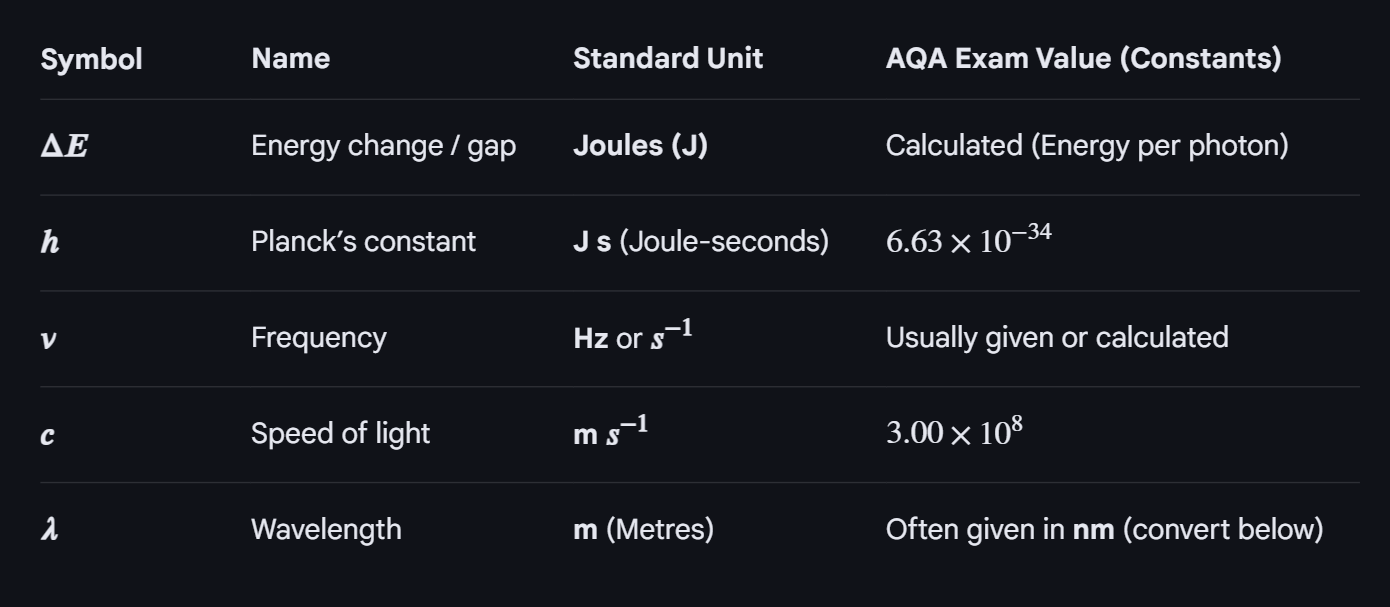

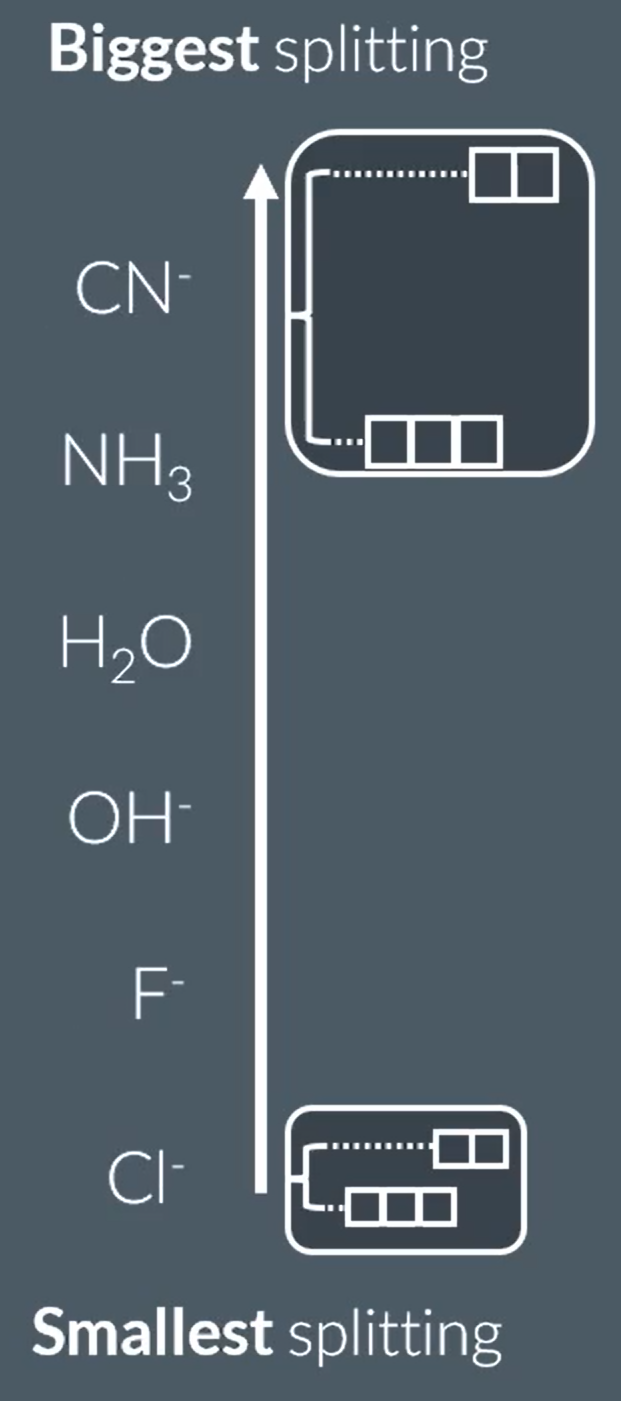
What are the factors that determine how much the splitting of d-obitals takes place?
The higher the oxidation state (of metal ion), the more the splitting. The lower the oxidation state, the less the splitting.
The higher the coordination number the greater the number of splitting, the lower the coordination number the less the number of splitting.
The splitting of d-orbitals depends on the ligand type
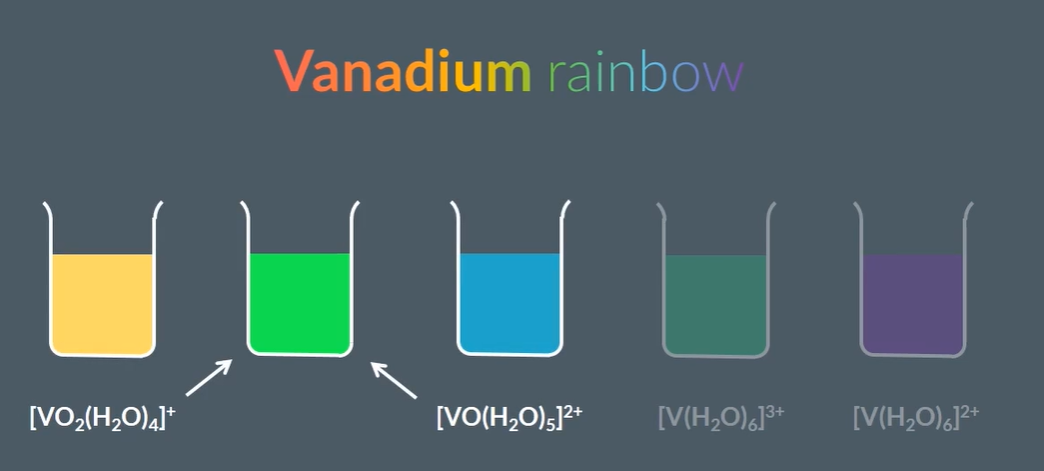
For variable oxidation states: The vanadium rainbow
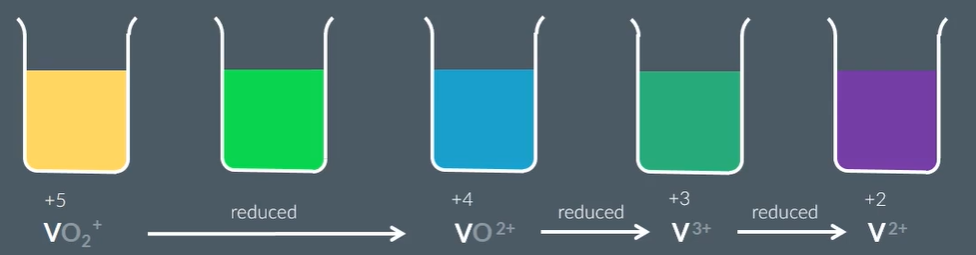
When you add Zinc, this series of vanadium rainbow takes place, because Zinc has a low electrode potential, VO2+ can be reduced three times. And the lower the E the stronger the reducing agent power, so can reduce the vandaium (+5) to Vandium (+2)
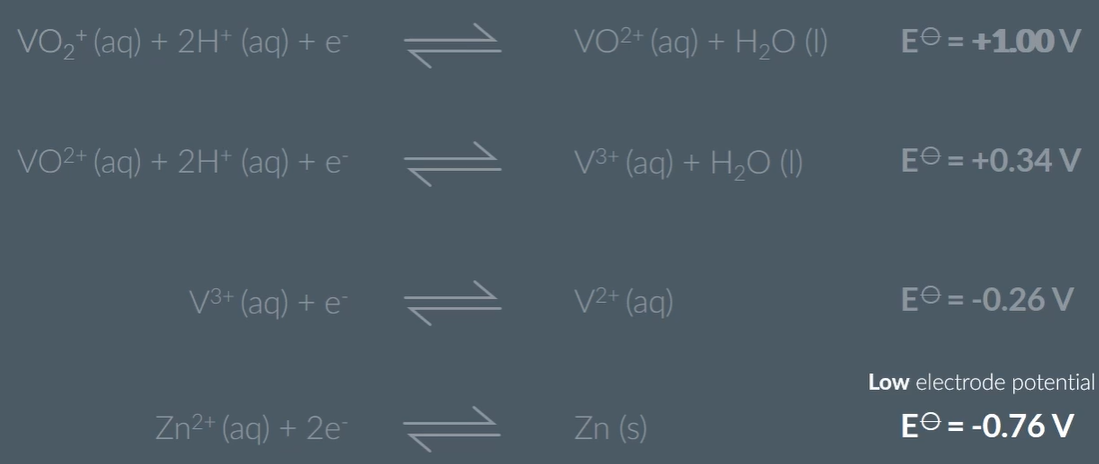
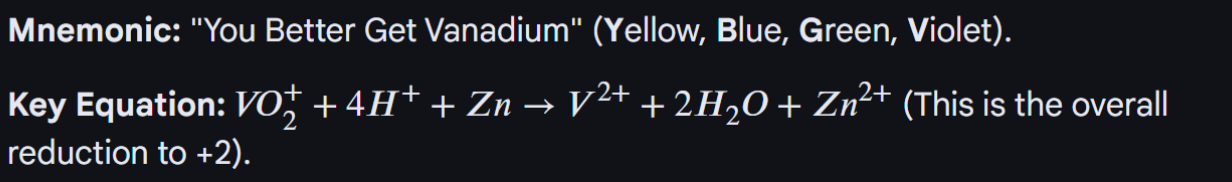
Variable oxidation states, Maganese compounds:
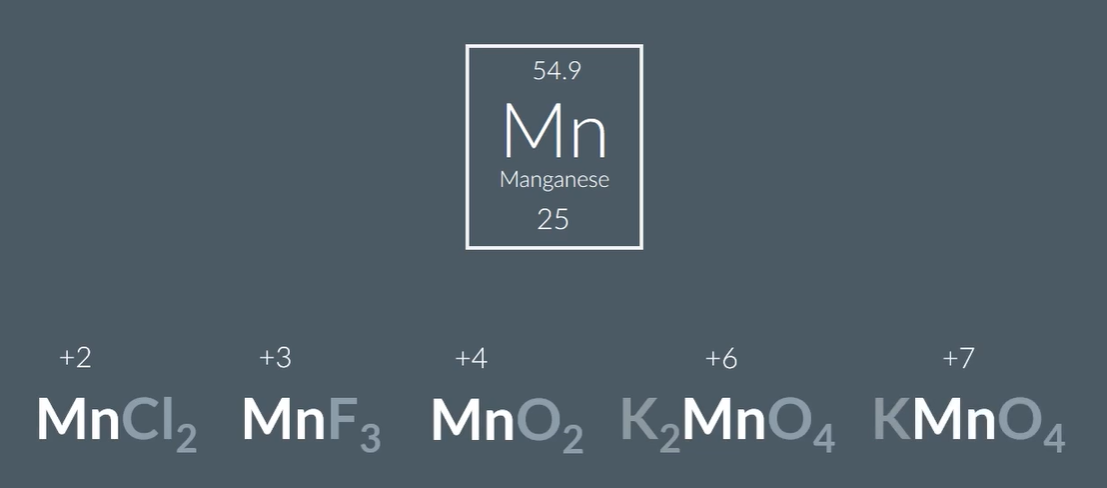
Oxidising agents in acidic conditions:
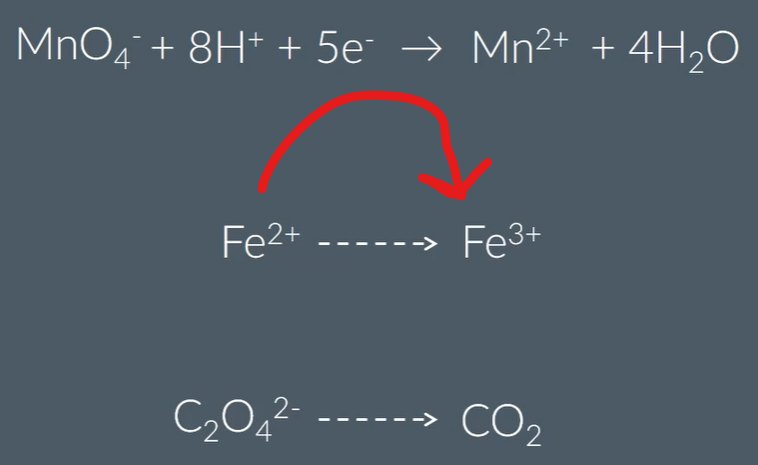
Maganate ions oxidise the following compounds
Maganate reacts with Iron in a 1:5 ratio
Magante reacts with ethane dioate in a 2:5 ratio

Testing if the Iron tablet contains 10-12 mg of Fe2+, we use a redox titration method, It is a self-indicating titration
First crush up the tablet, then make up a volume of 250 cm cubed using volumetric flask, then take out 25 cm cubed and add it to a conical flask.
Then in burette full of potassium maganate solution (of known concentration), slowly start adding to the conical flask
Initally, the solution turns colourless upon impact, however later turns back purple, because the Fe2+ ions are used up.
If you calculate how much potassium magnate used, then you can calculate how much Fe2+ in sample.
Define the terms Heterogeneous and Homogeneous catalysts:
Heterogeneous catalysts: A different phase from the reactants (is the only one that can provide an active site)
Homogeneous catalysts: The same phase as the reactants
If the question is 2 marks, say: speeds up the rate of reaction without being used up
The haber process:
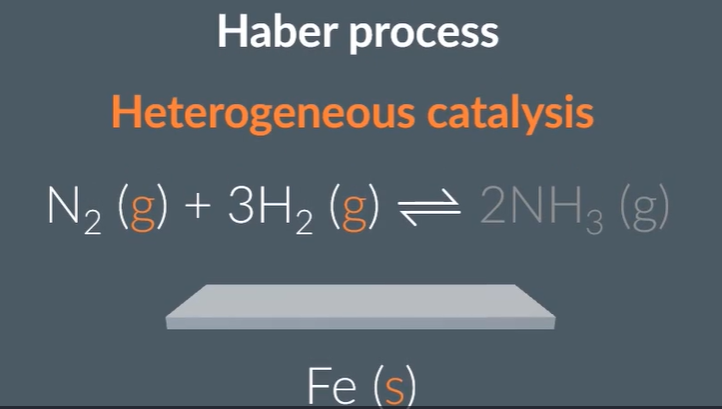
When nitrogen and hydrogen collide, they require a high amount of kinetic energy for a successful collision, however an alternative way is by using Iron solid. Where the nitrogen and hydrogen adsorb onto the Iron solid (called the active site), this causes the hydrogen and nitrogen bonds to break, where the Nitrogen atom bonds with 3 hydrogen atoms. Where the Iron, lowers the activation energy, which increases the rate of reaction.
The greater the surface area of the Iron, the more active sites that are present, can achieve this by turning the solid to a powered, or by making it out into a very thin shape
The very thin Iron coating can be stuck onto a ceramic which has a good porous properties, and the ceramic is called the support medium (so can increase the surface area, with reduced cost)
The contact process:
The V2O5 acts as a heterozygous catalyst

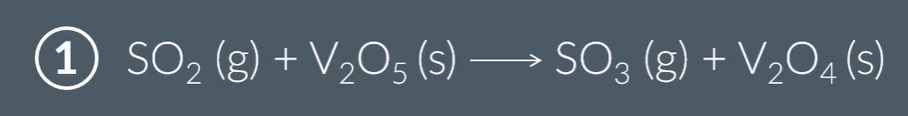
The sulfur dioxide adsorbs onto the V2O5, and then a redox reaction takes place to form the products.
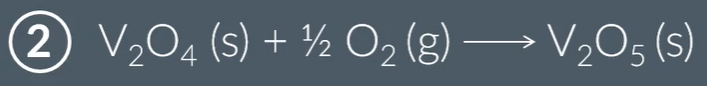
V2O5 is regenerated, which shows that catalysts are not used up in the reaction.
Overall, V2O5 uses the variable oxidation states of Vanadium to provide an alternative reaction route.
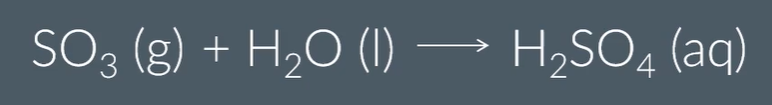

However, adding SO3 directly to water is dangerous (since hightly exothermic, and forms an acidic fog), so first add it to some H2SO4 to form H2S2O7, then add water. This allows the reaction safer and easier to control.
Catalyst poisoning:
Industrial used catalysts contain impurities, such as Aresenic trioxide, which also adsorbs to the active site on the catalyst, but doesn’t react blocking the active site for SO2. And replacing catalysts will become expensive over time.E
Example of a homogeneous catalyst:
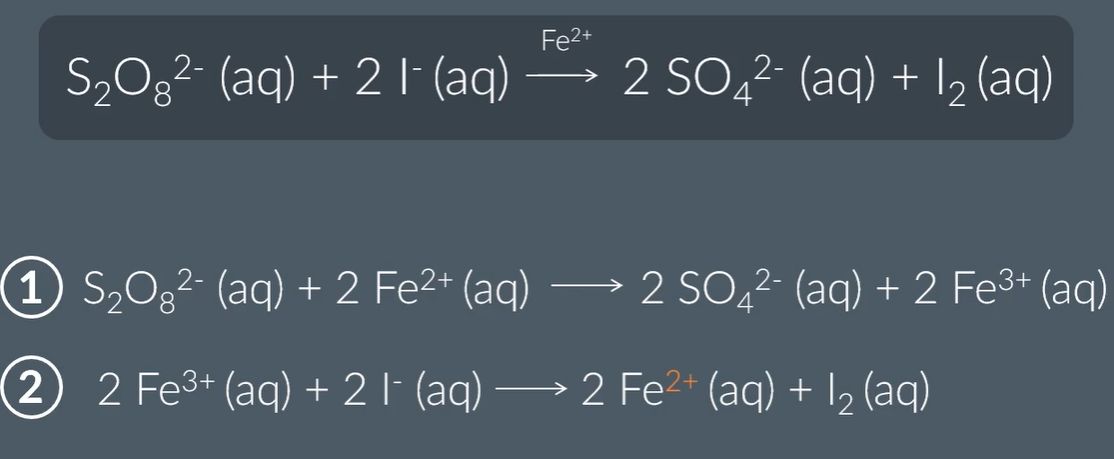
The two ways in which Iron helps: by attracting the opposite charged molecules, which lowers the activation energy, and also with Irons’ variable oxidation states, proving an alternative reaction route. Being a homogeneous catalyst, it cannot provide active sites.
The importance of catalysts having varibale oxidation states, enables them to readily accept and loose electrons, which provides for an alternative lower activation energy pathway.
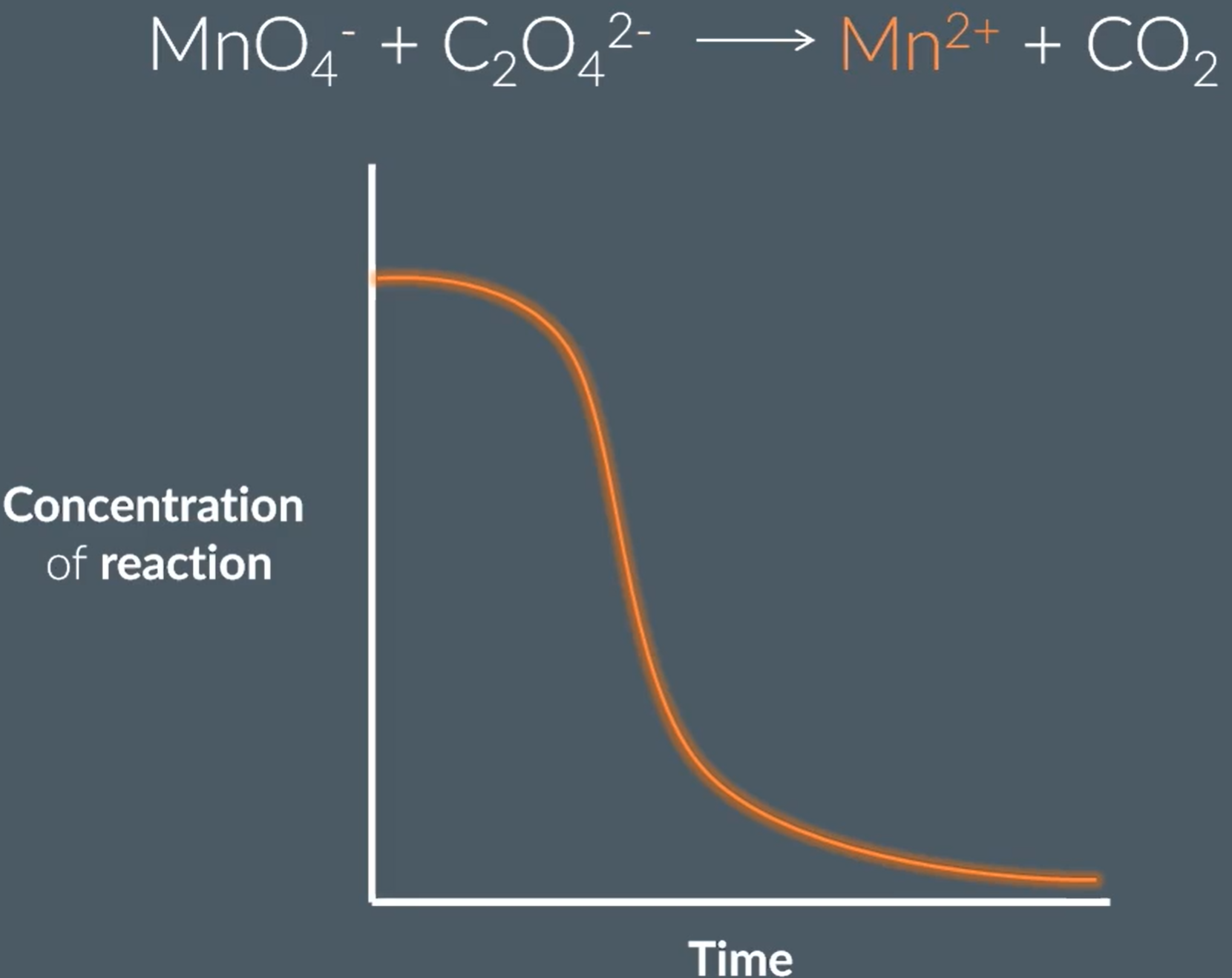
Define autocatalysis, and explain the shape of the graph and two equations for autocatalysis
The reason for this shape, is because initially the rate of reaction is slow as there is repulsion between the negative species, so lots of energy required for them to react successfully. However, the product forms Mn2+, which is positive so attracts the negatively charged species, which lowers the activation energy, so increases the rate of reaction. Finally, as most or all of the reactants have been used up, the rate of reaction decreases to zero.
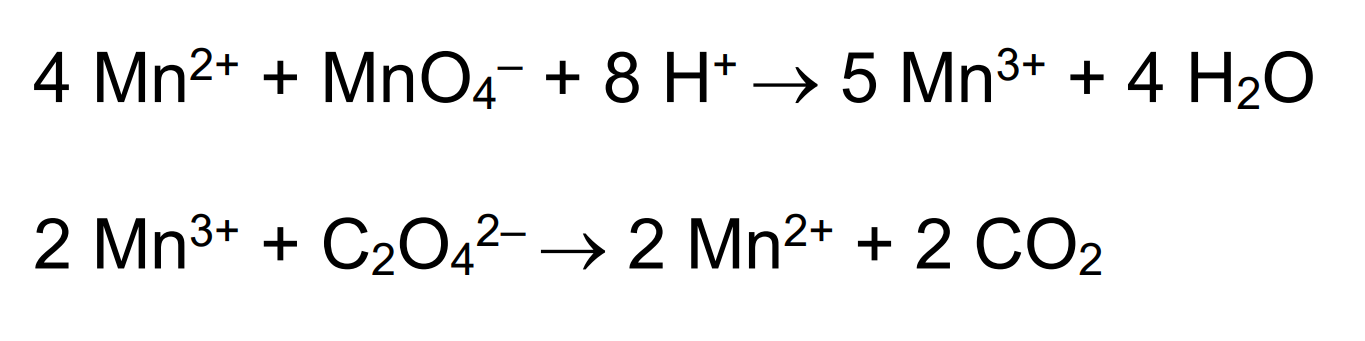
Essential key points about ligands, and complex ion formation:


What is a bidentate ligand, and what are the requirements:
Bidentate ions: where a molecule has two lone pairs, so can form 2 co-ordinate bonds withe the central ion.
They have to be long enough, in order to form a covalent bond to two different places on a molecule
They have to be flexible so can form the coordinate bond (not have lone pairs on opposite sides of the molecule)
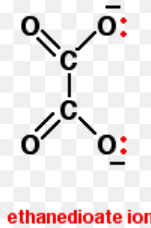
Draw the structure of ethandioate, ethane-1,2-diamine ligand:
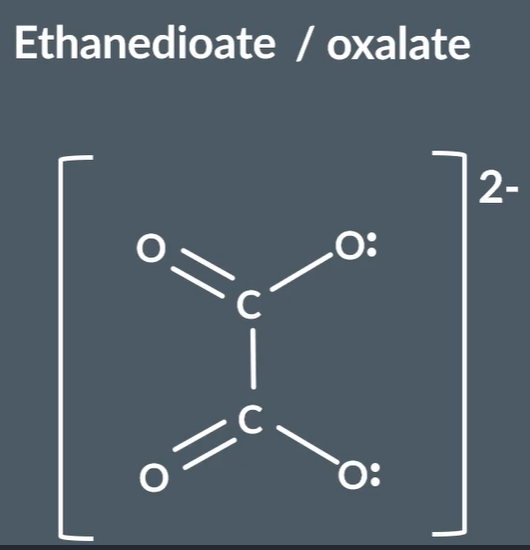
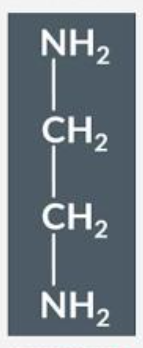
What is the chelate effect?
When a subsitution reaction takes place, in which the forward reaction contains more moles of molecules, then there is increased entropy.
When a monodentage ligand is substituted with bidentate ligands or polydentate ligands
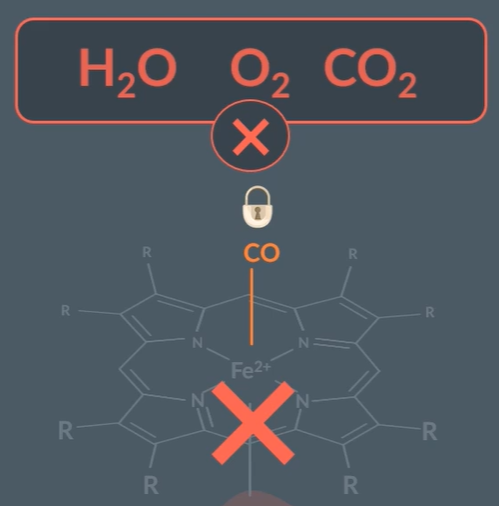
Everything about the Heam ligand:
Is a large ligand around the Fe2+, which forms 4 co-ordinate bonds, a quadridentate ligand.
CO forms a stronger coordinate bond than O2

Carbon monoxide posioning: When carbon monoxide forms a stable bond with the metal ion, that doesn’t undergo any further substitution reactions.
Key points about the information behind metal aqua complexes:
All metal aqua complexes are acids, because…When looking at the central metal ion, it has a greater positive charge, so attracts the electrons from the oxygen towards the Al3+, so this increases the partial positive charge on the hydrogen - so the other water molecules are attracted to the partially positive hydrogen, and the O-H bond on the complex bond breaks more easily.
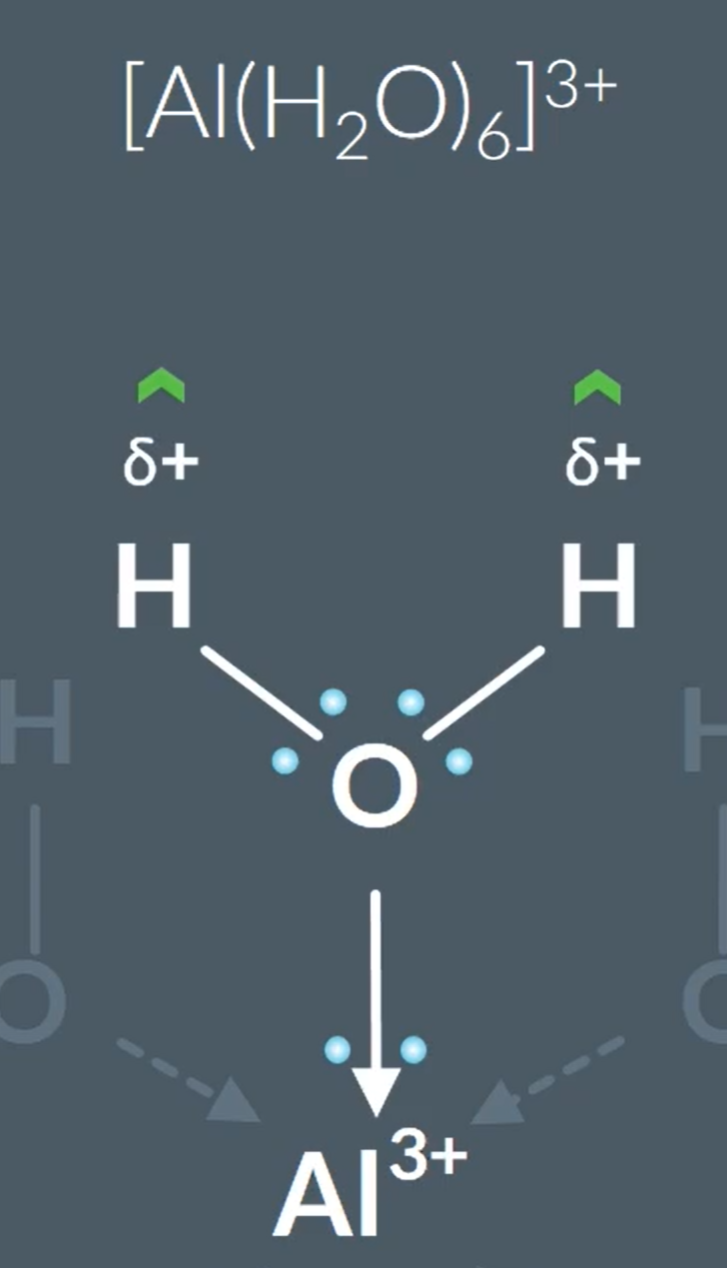
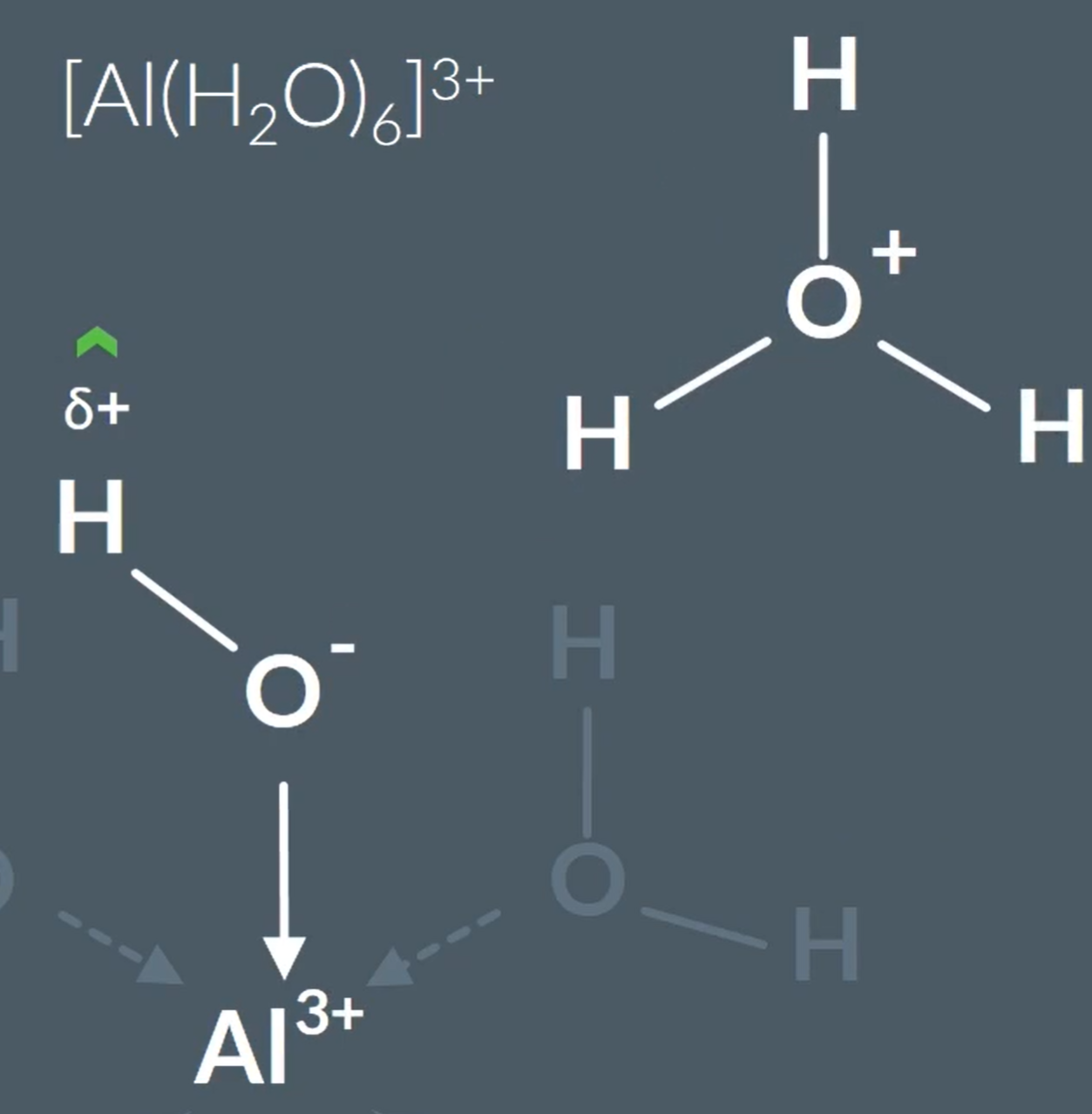
Comparing Metal-aqua ion acidity:
Metal ion charges with a 3+ > acidic than 2+.
This is because with higher charge more attraction between metal ion and electrons on outermost shell are more attracted to the nucleus, so has a smaller ionic radius. So, has a higher charge to size ratio. So, the electrons in the oxygen that is next to a metal ion will have the pair of electrons attracting the metal ion more closely, which causes the proton to leave from water ligand more readily.
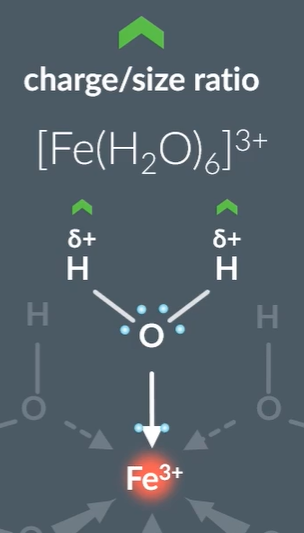
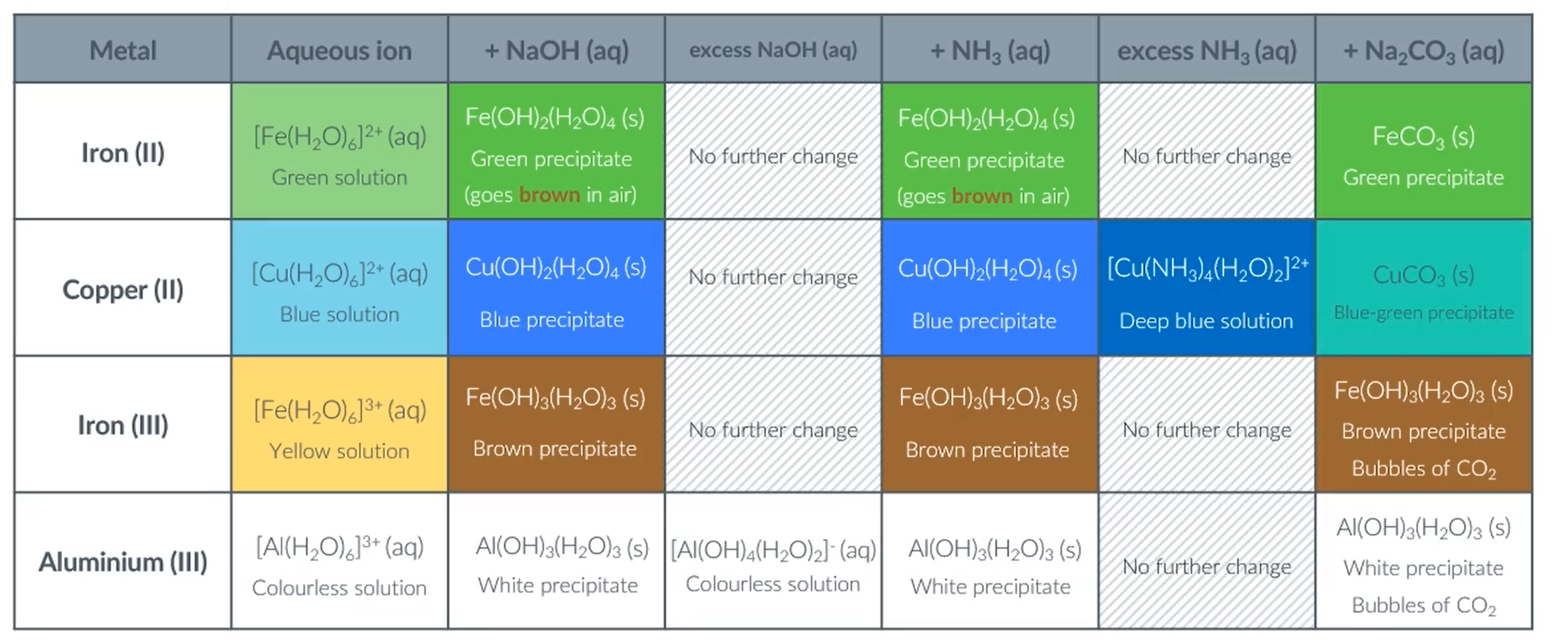
The addition of NaOH:
The overall equation:

However, in excess NaOH, the aluminium percipitate dissolves to form a colourless solution. (This is because it is a stronger acid)

When exposed to air the Fe2+ complex oxidised to Fe3+ complex:
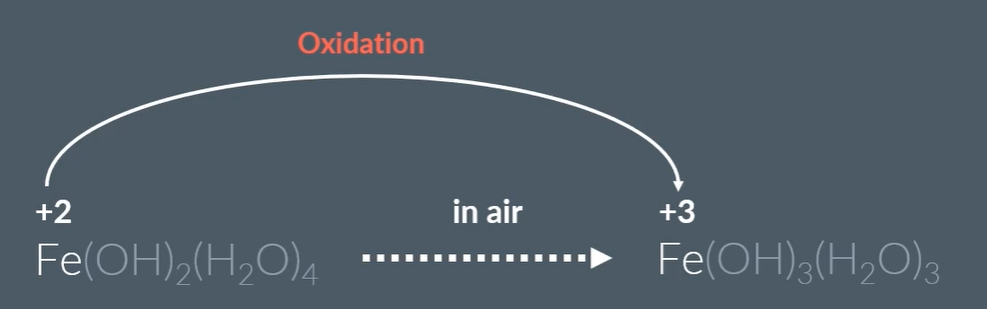
When some of the green percipitate is exposed to air it turns brown:
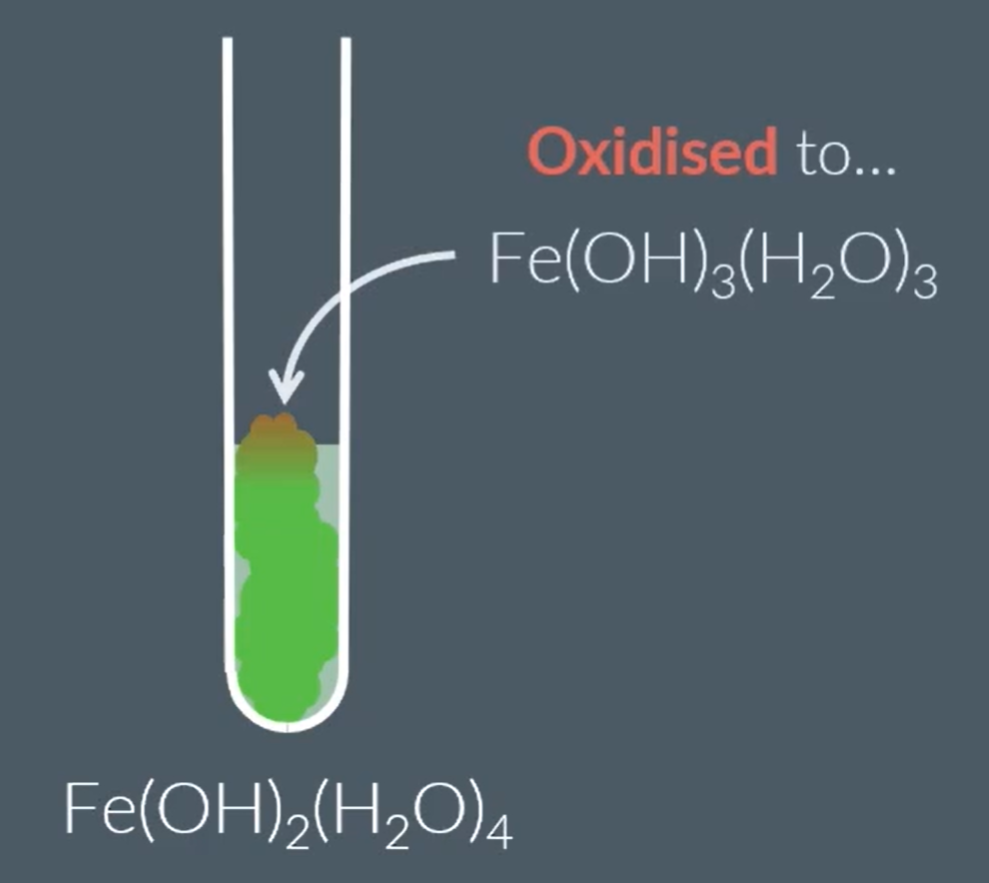
Amphoteric metal hydroxides:
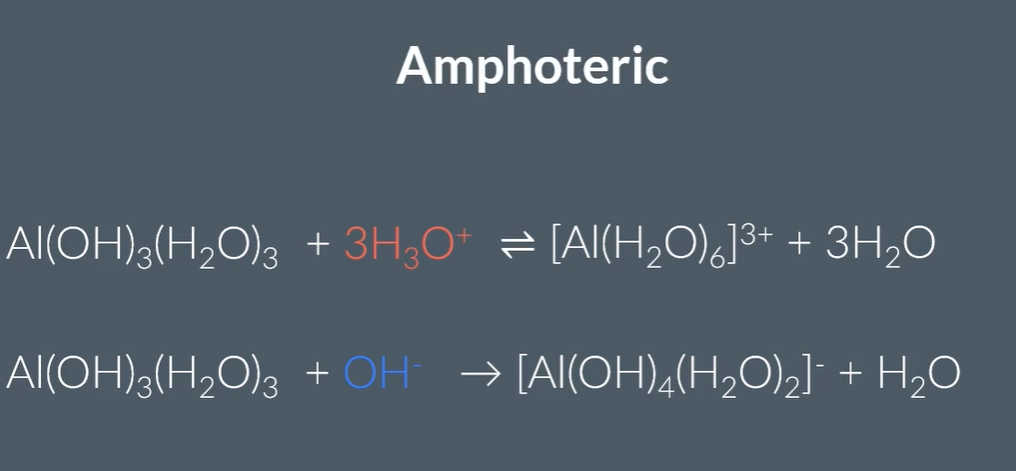
Why is it amphoteric?

So, suggests that hexaaquaaluminate is the strongest acid, so is strong enough to react with one more hydroxide.
Reactions of metal-aqua ions with ammonia:
Reactions of metal-aqua ions with ammonia:

With excess ammonia, hexaaquacopper turns from blue pericipitate to a dissolved royal blue solution: A ligand substitution reaction takes place.
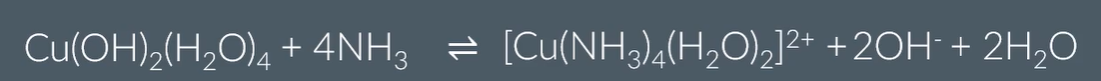
Reactions of metal aqua ions with Na2CO3
Reactions of metal aqua ions with Na2CO3 with 3+ complexes

Reactions of metal aqua ions with Na2CO3 with 2+ complexes:
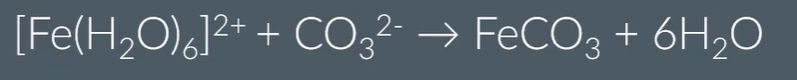
The reason for this difference, is because 2+ metal ions are weaker acids than 3+ metal ions, so are not strong enough to undergo the first reaction.
Summary of all the colour changes: