BIO (Book 1): Cell Membrane and Transport Across Membrane
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Structure of membrane to its Function
Fluid Mosaic Model
a ‘mosaic’ of proteins randomly distributed in or loosely attached to a fluid phospholipid bilayer
a fluid phospholipid bilayer whereby phospholipids are free to move about laterally
Characteristics of the fluid mosaic model:
The fluid layer is asymmetrical.
The two lipid layers (bilayer) may differ in composition and arrangement of proteins and lipids.
The three major types of membrane lipids are phospholipids, cholesterol and glycolipids.
The phospholipid bilayer is fluid or mobile, i.e. lateral movement of phospholipids is possible.
The unit membrane is a dynamic structure, where the embedded proteins can float, some moving freely while others are fixed in positions by microfilaments on the cytoplasmic face.
Membranes are amphipathic.
The hydrophilic phosphate head of the phospholipids face outwards into the aqueous environment both inside and outside of the cell;
The hydrophobic hydrocarbon tails face inwards and create a hydrophobic core.
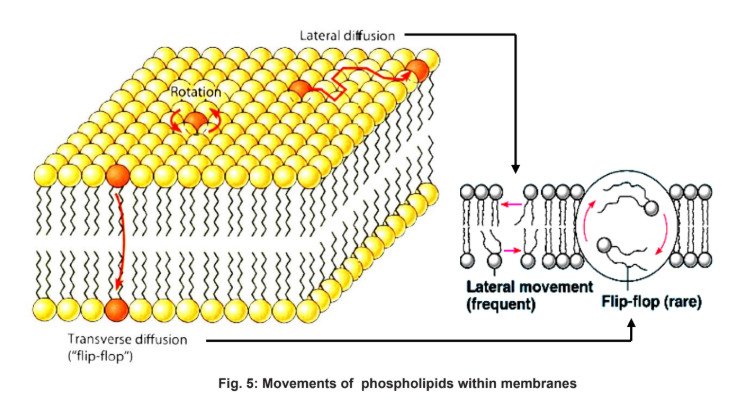
Membrane Fluidity
Bonds in the membrane:
The membrane comprises of phospholipid molecules which are held together primarily by hydrophobic interactions between the hydrophobic fatty acid tails.
As these are weak interactions, phospholipid molecules are free to move about laterally (slide left and right)
It is quite rare for the molecules to flip transversely across the membrane, switching from one phosphoplipid layer to the other. This is because the hydrophilic part of the molecule must cross the hydrophobic core of the membrane in order to do so
The ability of the phospholipid molecules to move within the membrane accounts for the fluidity of the cell membrane.
Factors affecting Membrane Fluidity:
Temperature
Length of fatty acid chains
Degree of saturation of fatty acid chains
Amount of cholesterol
Factors affecting Membrane Fluidity → TEMPERATURE
Overall trend:
As temperature decreases, membrane fluidity decreases, and vice versa.
Membranes remain fluid as temperature decreases, until finally the phospholipids settle into a closely-packed arrangement and the membrane solidifies.
This change of state (to semisolid) is known as the phase transition and the temperature at which it occurs is known as the phase transition temperature.
At LOW temperature:
Kinetic energy of the hydrocarbon chains decreases
Hydrocarbon chains are tightly packed
This results in an increased hydrophobic interactions between phospholipid molecules and thus, their motion is restricted.
The bilayer exists in a semisolid state, i.e. membrane is less fluid
At HIGH temperature:
The kinetic energy and thus motion of the hydrocarbon chains increases
This increase allows for increased lateral movements of individual molecules, flexing of the chains and transverse flipping,
Thus overcoming hydrophobic interactions between phospholipids, resulting in increased space between adjacent phospholipid molecules.
The bilayer exists in a fluid state, i.e. membrane is more fluid
Factors affecting Membrane Fluidity → LENGTH of fatty acid chains
Overall trend:
As length of fatty acid chains increases, membrane fluidity decreases.
In general, the longer the hydrocarbon chains, the higher the melting point (phase transition temperature) due to increased hydrophobic interactions between hydrocarbon chains.
Factors affecting Membrane Fluidity → Degree of saturation of fatty acid chains
General trend:
As degree of saturation of fatty acid chains increases, membrane fluidity decreases and vice versa.
Saturated lipids: (C—C single bond)
Have long, straight hydrocarbon chains
This allows for close packing and thus enhances membrane solidification
Unsaturated lipids: (C—C double bond)
Have kinks
Kinks prevent the hydrocarbon chains from packing closely together thus enhances membrane fluidity
Mixtures of phospholipids broaden the temperature range of the transition phase considerably.
Factors affecting Membrane Fluidity → Amount of cholesterol
General trend:
Cholesterol increases the stability and regulates the fluidity of membranes in animal cells.
Components of Cell Membrane
1. Phospholipids
2. Cholesterol
3. Proteins
4. Carbohydrates
Phospholipids
Each phospholipid molecule consists of:
a glycerol backbone (a three-carbon molecule) with three hydroxyl (-OH) groups
two fatty acid chains – contributing the hydrophobic hydrocarbon “tails”
a negatively charged phosphate group (PO43-)
additional small, charged molecules which may be linked to the phosphate group
Phospholipid molecule is amphipathic:
It has a hydrophobic “tail” and a hydrophilic “head”
This property results in the formation of phospholipid bilayers in an aqueous environment (i.e. hydrophilic heads are exposed to water, while hydrophobic tails are sheltered from water)
Long hydrocarbon chains of fatty acids:
Form an effective hydrophobic barrier against polar and charged solutes.
As the fatty acid chains are much longer than the glycerol head, their length dictates the thickness of the membrane, which is about 8 nm.
Cholesterol
Cholesterols are steroids
Commonly found wedged between phospholipid molecules in the cell membranes of animal cells.
Functions:
Membrane stability:
Cholesterol molecules are usually found in both layers of the cell membrane, intercalated into the lipid monolayers
Its rigid steroid ring interferes with the motions of the hydrocarbon chains of phospholipids, thus enhancing the mechanical stability of the membrane.
Membrane fluidity:
At HIGH temperature:
→ Cholesterol restrains the movements of phospholipids by interfering with the motions of the hydrocarbon chains,
→ Resulting in decreased membrane fluidity.
At LOW temperature:
→ Cholesterol prevents the hydrocarbon chains from packing closely together
→ Thus decreasing the tendency of the membrane to freeze upon
→ Resulting in increased membrane fluidity.
In short, cholesterol has dual effects on the fluidity of the membrane; resisting changes in membrane fluidity that can be caused by changes in temperature, acting as a “temperature buffer” for the membrane.
Membrane permeability:
Presence of cholesterol molecules decreases the permeability of a lipid bilayer to ions and small polar molecules.
It does so by filling in spaces between hydrocarbon chains of phospholipids, thereby plugging transient gaps through which ions and small molecules might otherwise pass.
Proteins
Types of membrane proteins:
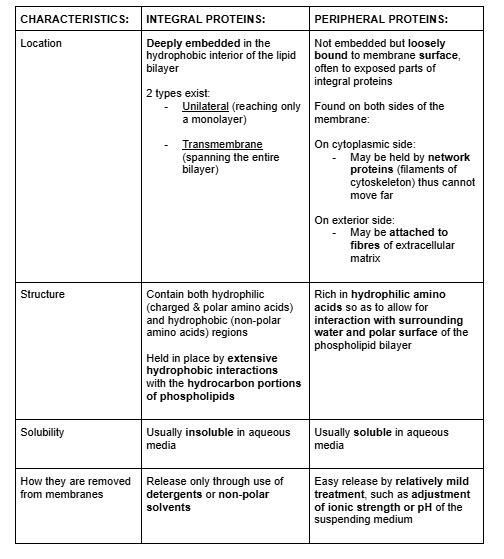
Integral (intrinsic) proteins
Peripheral (extrinsic) proteins
6 major types of function:
Anchorage
Transport
Enzymatic activity
Signal transduction
Cell-to-cell recognition
Intercellular joining
Functions of proteins → Anchorage
Anchoring proteins
Attach the cell membrane to other substances
Stabilise the position of the cell membrane
Help maintain cell shape.
Anchoring proteins attached to the extracellular matrix can coordinate extracellular and intracellular changes
On the cytoplasmic side: They are bound to microfilaments of the cytoskeleton.
On the exterior side: They may attach the cell to fibres of the extracellular matrix.
Functions of proteins → Transport
Carrier proteins:
Carrier proteins bind solutes and transport them across the membrane
This transport process involves a conformational change of the protein when solute binding occurs, and a return to its original form when the solute is released.
Energy in the form of ATP may or may not be required:
→ Facilitated diffusion: if solute moves down a concentration gradient (no energy external to the system is required)
→ Active transport: if solute moves against a concentration gradient (ATP is required)
Channel proteins:
Some integral proteins contain a water-filled central pore, or hydrophilic channel
This forms a passageway to permit the movement (down the concentration gradient) of water, ions and small hydrophilic solutes across the cell membrane.
It participates in FACILITATED DIFFUSION (no ATP required)
There are 2 major kinds of channels:
Leak channels
→ Permit movement of water at all times, e.g. aquaporins
→ Permit movement of ions at all times (though the rate may vary), e.g. Na+ or K+ leak channels
Gated channels
→ which can open or close to regulate ion passage
→ e.g. voltage-gated Na+ or K+ channels
Functions of proteins → Enzymatic activity
These enzymes catalyse reactions in the extracellular fluid or within the cytosol, depending on the location of the active site
In some instances, several enzymes can be grouped together to carry out sequential steps in a metabolic pathway.
Functions of proteins → Signal transduction
These proteins have very specific 3D conformations, making them ideal as receptor molecules for chemical signalling between cells.
Chemical signalling works by the binding of a ligand to the receptor protein which triggers changes in the cell
Ligand → an ion, molecule, or functional group that binds to a receptor protein
Cell membranes differ in the type and number of receptor proteins they contain. It is these differences that account for the differing sensitivities to hormones and neurotransmitters etc.
Functions of proteins → Cell-to-cell recognition
Recognition proteins are usually glycoproteins (carbohydrate attached to proteins)
There is a wide array of possible shapes to the carbohydrate side chains; hence each cell type has its own specific markers.
This enables cells to recognise other cells, and provides a means for foreign markers to be recognised and attacked by the immune system.
Functions of proteins → Intercellular joining
Membrane proteins of adjacent cells may adhere together in various kinds of intercellular junctions, such as gap junctions and tight junctions
Carbohydrates
Membrane carbohydrates are usually short, branched chains of fewer than 15 sugar units
Some of these are covalently bonded to polar ends of phospholipids molecules in the outer lipid layer, forming glycolipids
Some are covalently bonded to membrane proteins, forming glycoproteins
Functions:
The carbohydrate groups aid in maintaining the orientation of the glycoproteins and glycolipids within the membrane.
As carbohydrates are highly hydrophilic, the glycolipids and glycoproteins are kept in contact with the external aqueous environment and are unlikely to rotate towards the interior to diffuse transversely.
Due to the hydrophilic properties of carbohydrates, glycolipids and glycoproteins are important recognition components, such as those involved in:
Sorting of cells into tissues and organs in animal embryos
Binding extracellular signal molecules in antibody-antigen reactions
Intercellular adhesion to form tissues
Cell-to-cell recognition, that is the ability of a cell to distinguish one cell from another, e.g. the immune system identifies and acts upon foreign cells
Transport Across Membrane
Reasons for transport across membrane:
To maintain a suitable pH and ionic concentration within the cell for enzyme activity;
To obtain food supplies for energy and raw materials;
To excrete toxic substances
To secrete useful substances;
To generate the ionic gradients essential for nervous and muscular activity.
3 general types of transport:
Passive transport
Active transport
Bulk transport
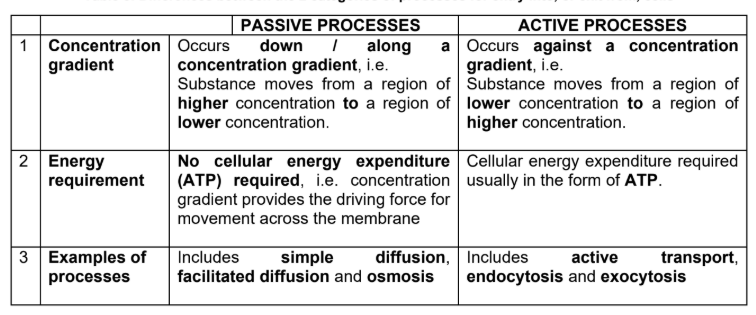
Passive transport (diffusion)
Types of diffusion:
Simple diffusion
Facilitated diffusion
Diffusion is the net movement of a substance (solute molecules) from a region of higher concentration to a region of lower concentration, that is, down a concentration gradient.
Though diffusion is regarded as a form of passive transport, it is incorrect to assume that no energy is involved in the process.
Instead, molecules have intrinsic kinetic (or thermal motion) energy, which is the tendency for them to spread out evenly into the available space. (but don’t require ATP)
Diffusion continues until dynamic equilibrium is reached.
Passive transport → Simple Diffusion
Simple diffusion occurs for molecules that are able to cross the phospholipid bilayer directly without any need for the aid of channel or carrier protein
Molecules that have a small molecular weight and / or are readily soluble in the lipid bilayer (hydrophobic molecules)
Diffusion can occur in either direction (i.e. from within the cell to outside the cell, or vice versa) depending on the concentration gradient.
Dynamic equilibrium is reached when concentrations of the diffusing substances are equal on both sides of the membrane. No net movement of substances then occurs
Energy from ATP is not needed.
Passive transport → Facilitated Diffusion
The principle of facilitated diffusion is similar to that of simple diffusion in that transport of substances occurs down a concentration gradient without the use of ATP until equilibrium is reached.
It is "facilitated" because a transport protein in the membrane is used to enhance / increase the rate of transport of the substance across the membrane.
Facilitated diffusion is used for larger, hydrophilic substances, such as glucose, amino acids and ions.
The involvement of the transport protein makes facilitated diffusion a type of channel-mediated or carrier-mediated transport, although it is passive.
The transport protein is specific to the substance being transported, i.e. it only transports that substance.