Chapter 8
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Spontaneous process
a reaction or change that occurs naturally under specific conditions without continuous external energy input
Gibbs free energy, G
Allows us to predict direction and if a chemical/physical change is spontaneous in particular conditions
How is gibbs free energy calculated
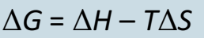
How is delta G and spontaneity related (when is it spontaneous and when is it not)

System
A particular part of the universe (one or more chemical species)
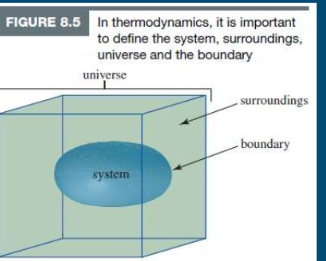
Surrounding
Everything else aside from the system
Open system
Can exchange both matter and energy with the environment
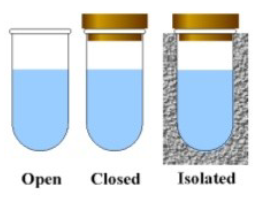
Closed system
Can exchange energy but not matter with the environment
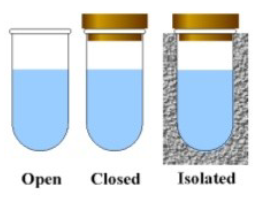
Isolated system
Can exchange neither energy nor matter with the environment
Heat
Energy used to cause the temperature of an object to rise (thermal energy)
Potential energy
Energy an object possesses due to its position or chemical composition
Kinetic energy
Energy an object possesses by its motion
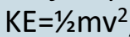
Radiant energy
Energy transmitted by light
Electrical energy
Energy involved in the movement of electrons, can be mechanically generated or chemically generated
Chemical energy
Energy associated with the strength of bonds between different elements
Heat vs temperature
Heat is the transfer of energy due to a temperature difference in K
Temperature is a measurement of how hot an object is
Temperature
A measure of the average kinetic energy of an objects atoms, molecules, or ions
State function
Properties independent of how a state is prepared, are the functions of variables that define the current state of a system (pressure, volume, temperature)
They are properties the state possesses

Path function
Properties related to the path/preparation of the state of the substance
The work done in preparing the state and energy transferred as heat are path functions
The system does not posses the heat or work
Internal energy (U)
The energy within a system even if the existence of the energy is not obvious, is the sum of kinetic and potential energies


Pressure-volume (PV) work
the energy transferred when a system (like a gas) expands or compresses against an external pressure

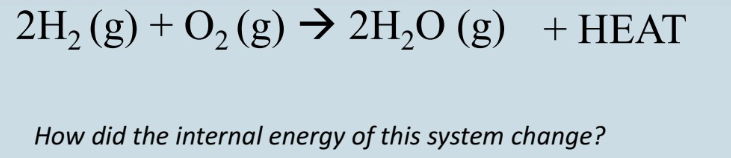
First law of thermodynamics
The total energy of the universe is constant

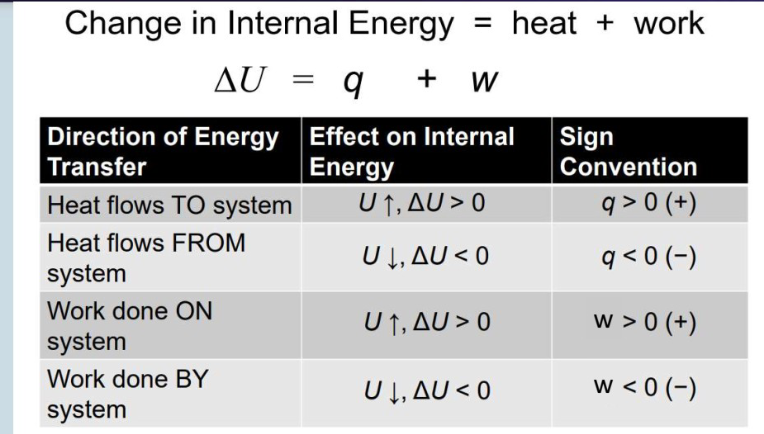
Keeping track of internal energy
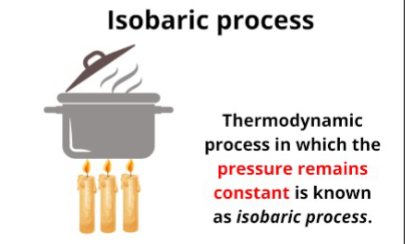
Isobaric process
Thermodynamic process in which pressure is constant
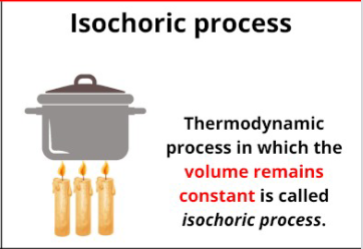
Isochoric process
Thermodynamic process in which volume is constant
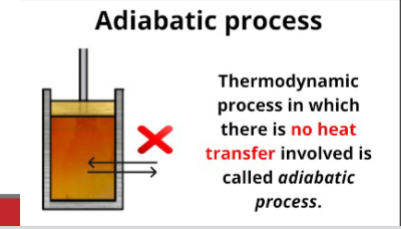
Adiabatic process
Thermodynamic process in which no heat is transferred
Isothermal process
The process in which the temperature remains constant
Internal energy (U), heat (Q), and work (w) equations

when is q>0
When the system absorbs heat from the surroundings (endothermic)
When is q<0
system releases heat to the surroundings (exothermic)
When is w>0
When work is done ON the system (energy flows in)
when is w<0
When work is done BY the system (energy flows out)
Heat capacity equation
q=mc(delta)T

Enthalpy
a thermodynamic state function representing the total heat content of a system, defined as the sum of its internal energy ( or ) and the product of pressure () and volume (): . It measures the energy required to create a system and make space for it at constant pressure.
Positive delta H
Endothermic reaction
Negative delta H
Exothermic
Types of enthalpies
