Orgo 2 Exam 4
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
Effect of Neighboring Hydrogens in HNMR
N+1, but the more neighboring hydrogens, the more deshielded the Ha is, meaning that it is shifted slightly downfield.
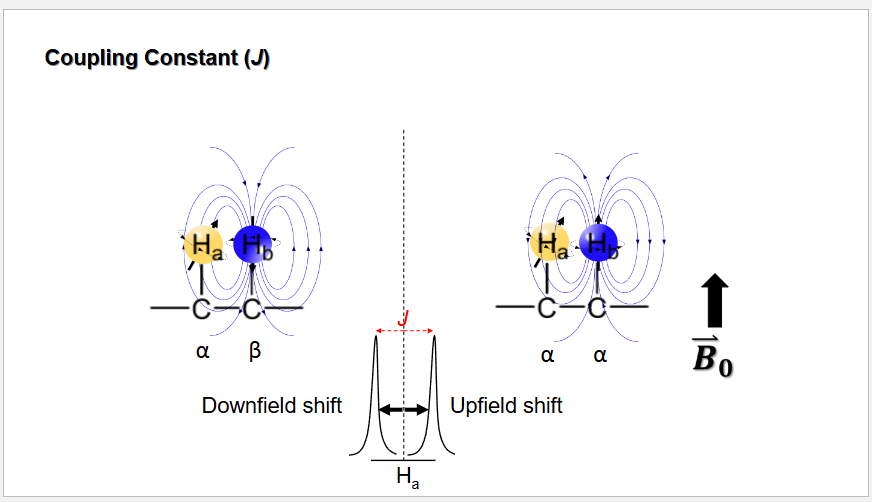
Coupling constant definition
The distance between Peaks in HNMR (Measured in Hz)
J Value for “Vicinal” Hydrogens
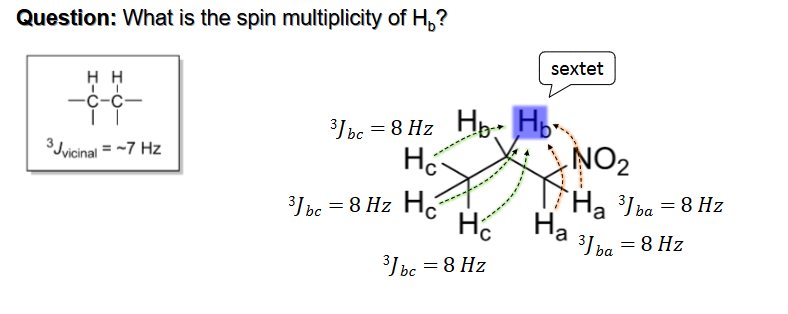
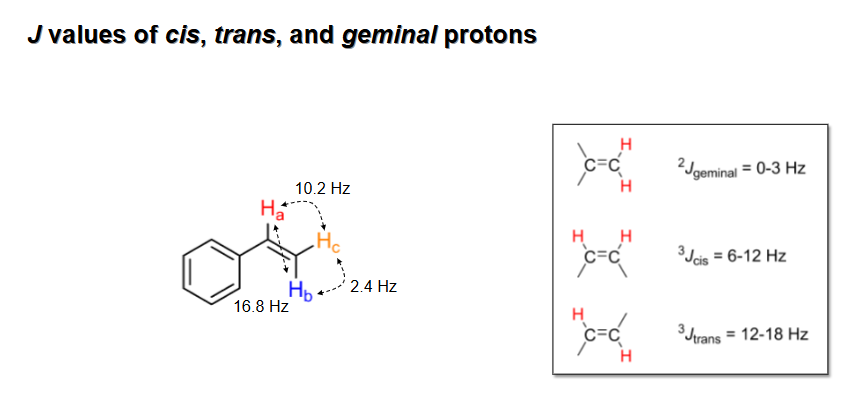
J Values of “cis,” “trans,” and “Geminal” protons
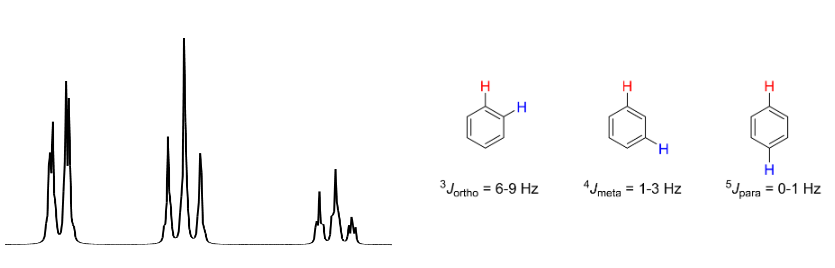
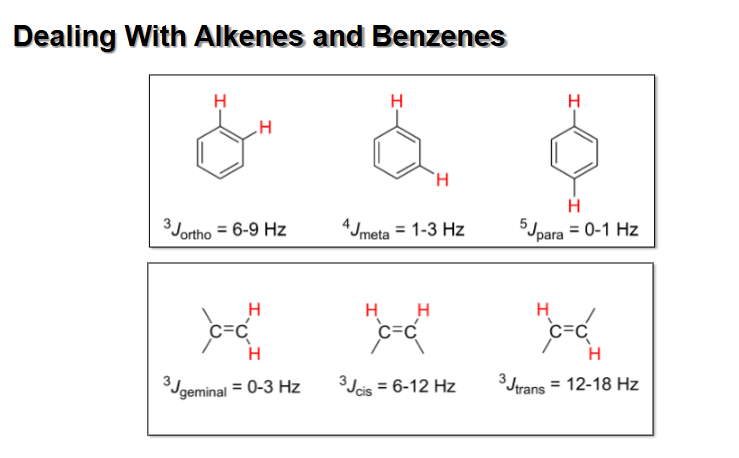
J values for O, p, and m in Benzene rings
Para protons don’t really couple
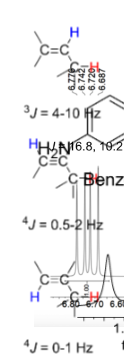
J for 3 carbon vinylic systems
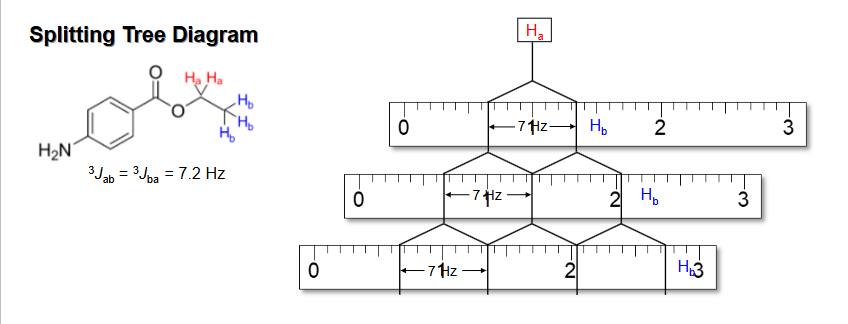
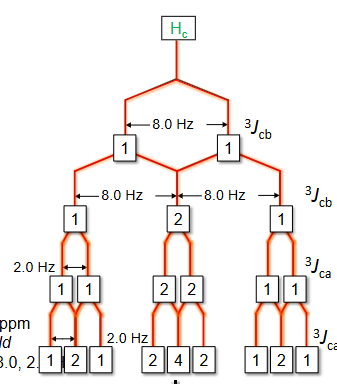
How to do Splitting tree for multiple Js
Shielding/Deshielding in Benzene ring for EDG
Ortho and para are more shielded (upfield) because resonance effect gives them lone pair while meta is deshielded
Splitting pattern USUALLY for ortho hydrogen
Doublet of doublet
Splitting pattern USUALLY for Meta hydrogen
triplet
Splitting pattern USUALLY for Para hydrogen
Triplet of triplet
J for alkenes and Benzenes
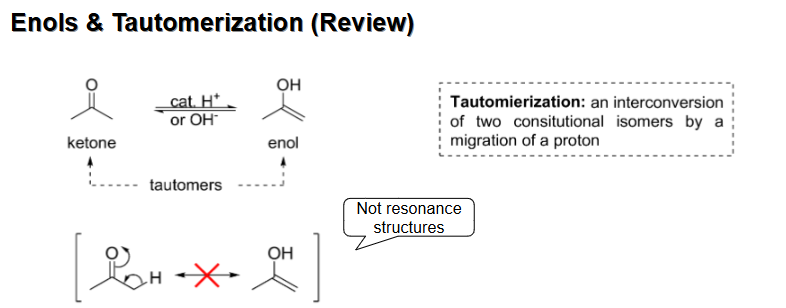
Enol formation from tautomerization
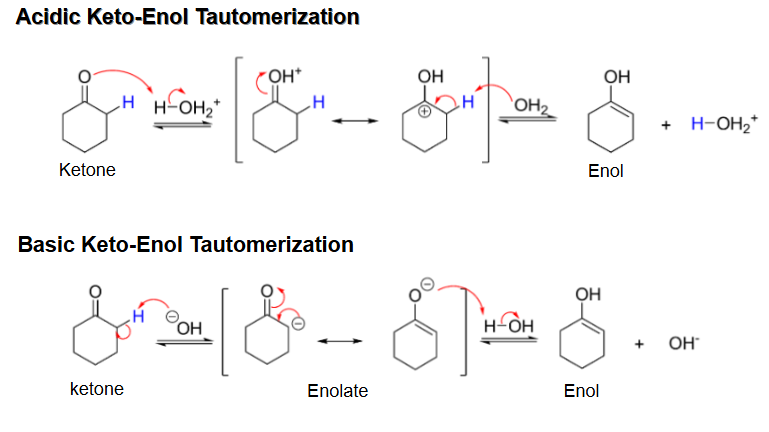
Acidic vs basic ketone tautomerization mechanism
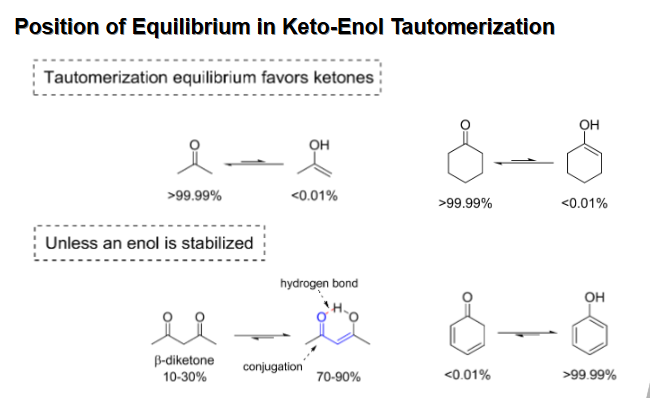
Tautomerization equilibrium position
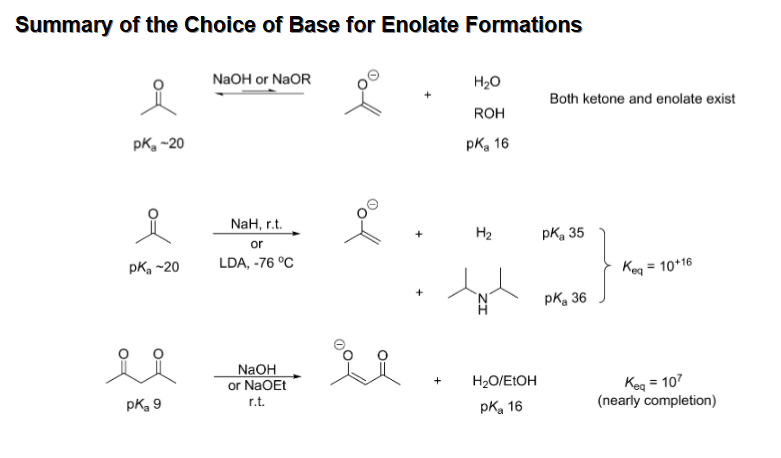
Bases used for Enolate Formation
NaOEt and t-BuOK are used for reversible enolate formation (t-BuOK is stronger) while NaH and LDA are used for irreversible but greater yield enolate formation
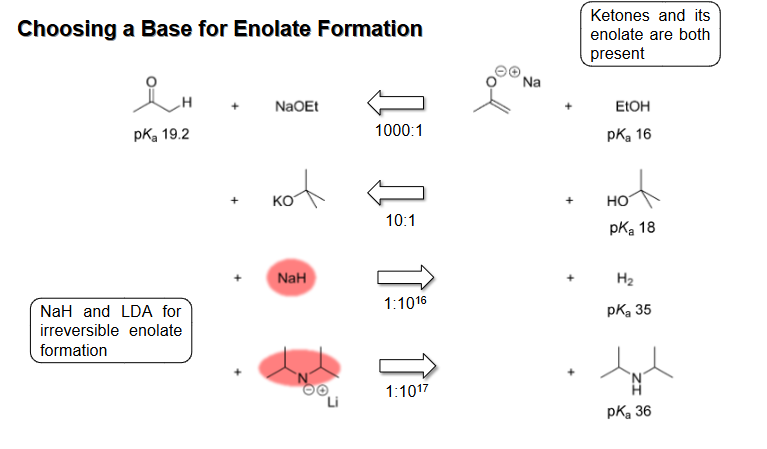
Choice of base when enolate is resonance stabilized
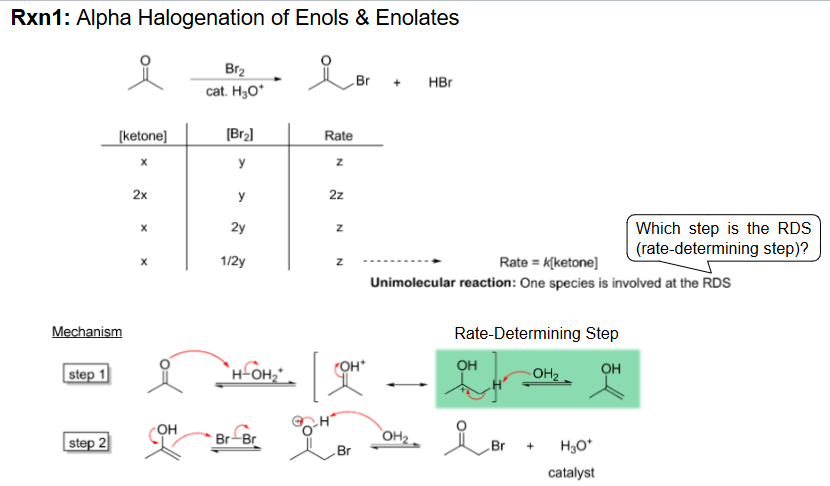
Alpha Halogenation of Enols and Enolates Reagents and Mechanism
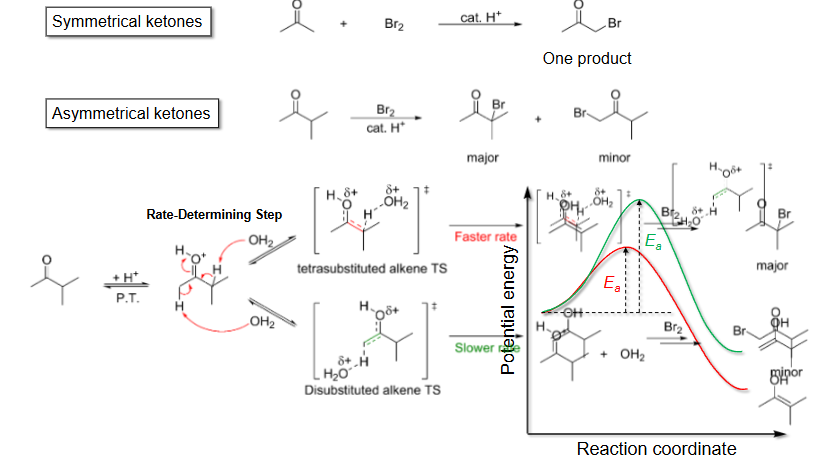
Regioselectivity in alpha halogenation
Alpha bromination of carboxyllic acids, esters, or amides
Doesn’t happen with the same reagents as ketones and aldehydes because the enols are unstable
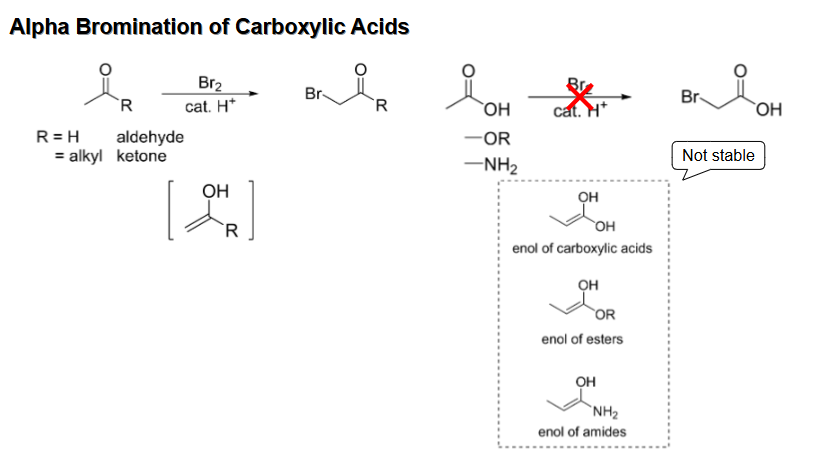
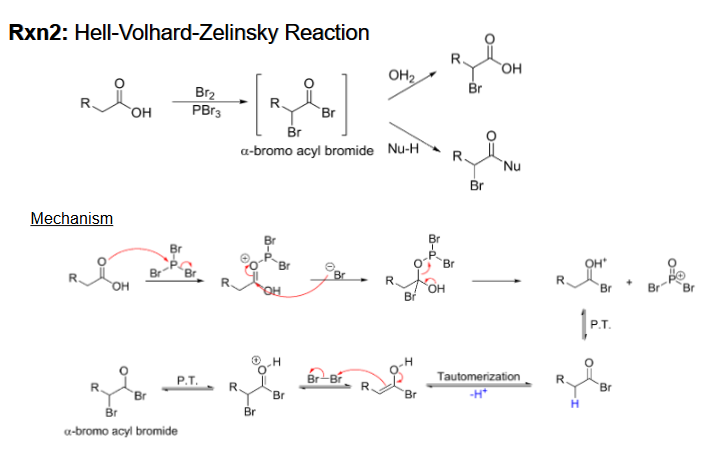
Hell-Volhard-Zelinsky Reaction (Alpha halogenation of COOH) Reagents and mechanism
Reagents are Br2 and PBr3 and second step is H2O (or nucleophile with H)