Chem 2 lab exam short answer
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
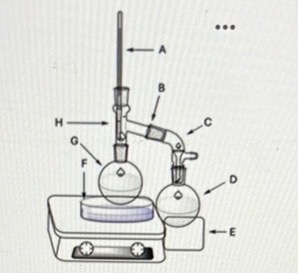
What is the name of the glassware at point C?
Vacuum adapter
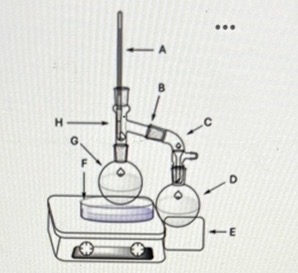
What is the difference between the positioning of the thermometer at point H compared to the other similar setups we conducted this semester?
It is measuring the temperature of the vapor, not the liquid
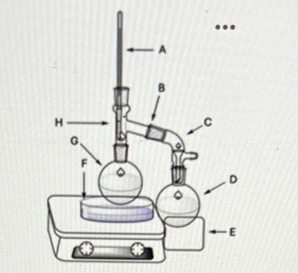
What is being referenced at point E? What is the purpose of this feature?
The ice bath. When vapor travels to the collection flask, the ice bath helps condense it into a liquid distillate.

Water and phenylamine are combined in a mixture, although they are immiscible with each other. The normal atmospheric pressure in the lab is 101.325 kPa. At 98 °C, the saturated vapor pressures of these two pure liquids are:
What would the total vapor pressure of a water-phenylamine mixture be at 98 °C?
101.37 degrees Celsius.

If atmospheric pressure is 101.325 kPa, what is happening to the mixture of water and phenylamine at 98 °C?
The mixture co distills at 98 degrees Celsius, which is lower than the boiling point of both the water and the phenylamine.

The boiling point of water is 100 °C, and phenylamine is 187 °C. How is this related to your answer to part (b)? What has happened to this system of immiscible liquids?
Because they are immiscibile, their vapor pressures combine and this leads to the mixture boiling at a lower boiling point which mimics azeotropic behavior.
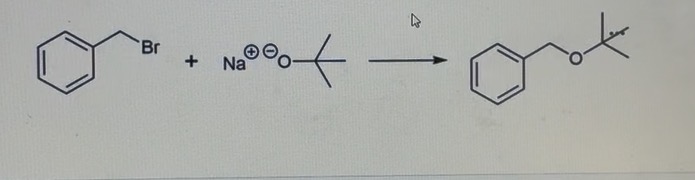
Label the nucleophile and electrophile in this Williamson Ether Synthesis reaction. What reaction mechanism would this undergo?
Nucleophile: sodium ethoxide
Electrophile: 1-bromobutane
It would undergo an SN2 reaction
Compare and contrast the short-path and steam distilation techniques with the first simple distillation we performed in Lab 2. Identify at least two similarities and two differences between each. (Hint: when is each used, and how do the setups compare
Simple distillation: When boiling points are sufficiently far apart, uses a reflux column, poor separation, etc.
Steam Distillation: When two substances are immiscible and/or one substance will degrade before its boiling pt, collected in still, etc
Short Path Distillation: When boiling points are very far apart, uses no reflux and/or only uses vacuum adapter, etc
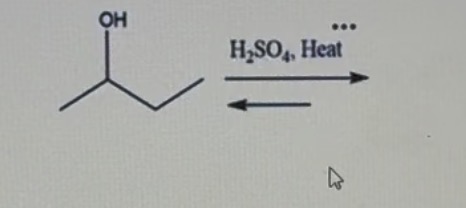
Draw the possible products) to complete the reaction.
1-butene, cis-2 butene, and trans-2 butene.
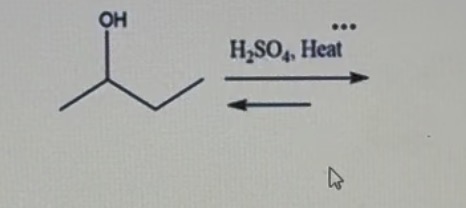
What is the name of the rule used to predict major and minor products in this case?
Zaitzev's rule
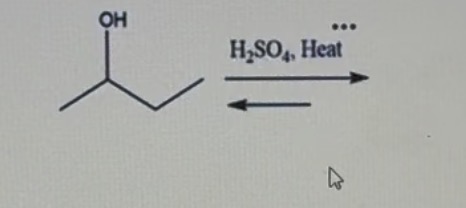
What type of instrumentation did we use to analyze products from this reaction?
Gas chromatography .
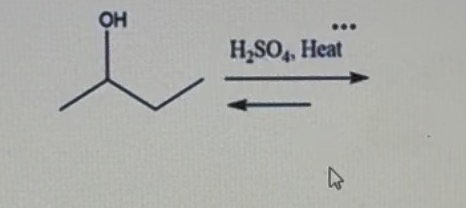
What did that instrumentation tell us about the product(s) generated?
It identifies the alkenes in the mixture by separating them based on boiling points. It uses retention times to distinguish between isomers and peak areas to determine the major and minor products, confirming which isomer was favored by the reaction.
What are the three steps of an acid-catalyzed hydration reaction? Draw a picture to explain if it helps.
first step: protonation by a H3O+ ion which leads to formation of carbocation
second step: a water molecule attacks a carbocation forming alkyloxonium ion
Third step: deprotonate the oxonium ion which leads to formation of an alcohol and acid catalyst
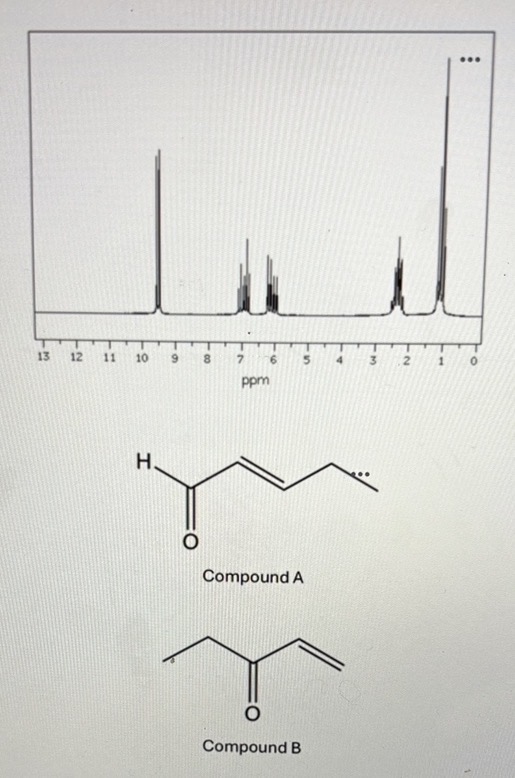
Consider the 1H NMR spectra below. Determine which of the possible products (A or B) has likely been produced in the lab based on the features of the NIR spectra we have discussed. For full credit, discuss the number of signals, shifts, splitting patterns, and integrations
Product A because there is an aldehyde signal at 9-10ppm which is only seen in product A.