Organic formulae and homologous series
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What is the electronic configuration of carbon? How many electrons does it need to become stable?
1s22s22p2 - In group 4 so has 4 electrons in its outer shell and needs 4 more electrons to become stable.
What does carbon form to become stable?
Four covalent bonds
What are the three different ways that carbon forms the four covalent bonds?
4 single covalent bonds
2 single bonds and 1 double bond
1 single bond and 1 triple bond
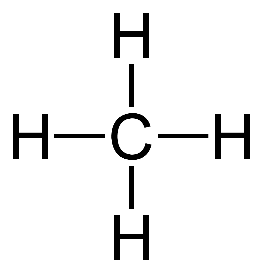
Example of carbon forming 4 covalent bonds
Methene
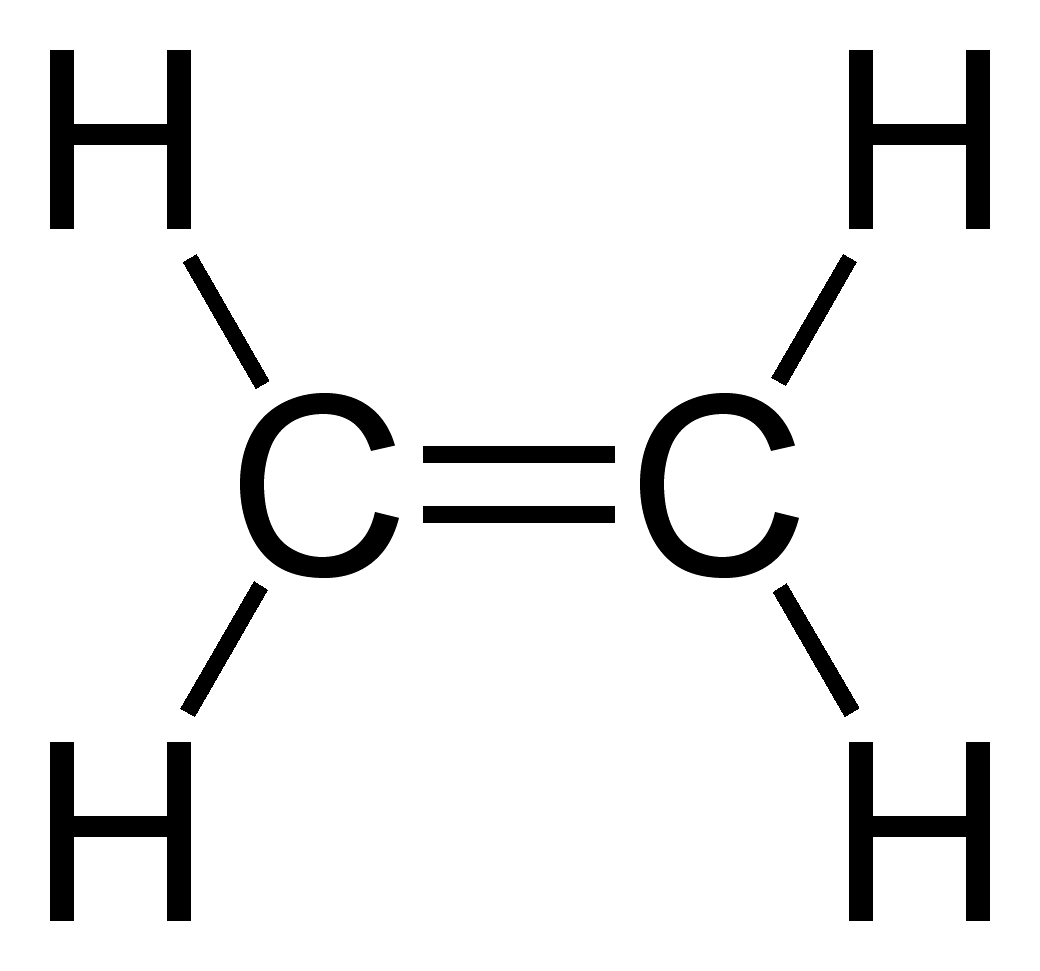
Example of carbon forming 2 single bonds and 1 double bond
Ethene
Example of carbon forming 1 single bond and 1 triple bond
Ethyne
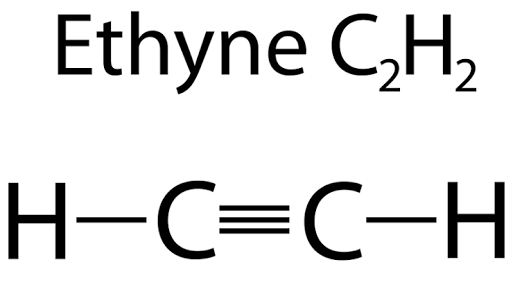
Types of formula summary:
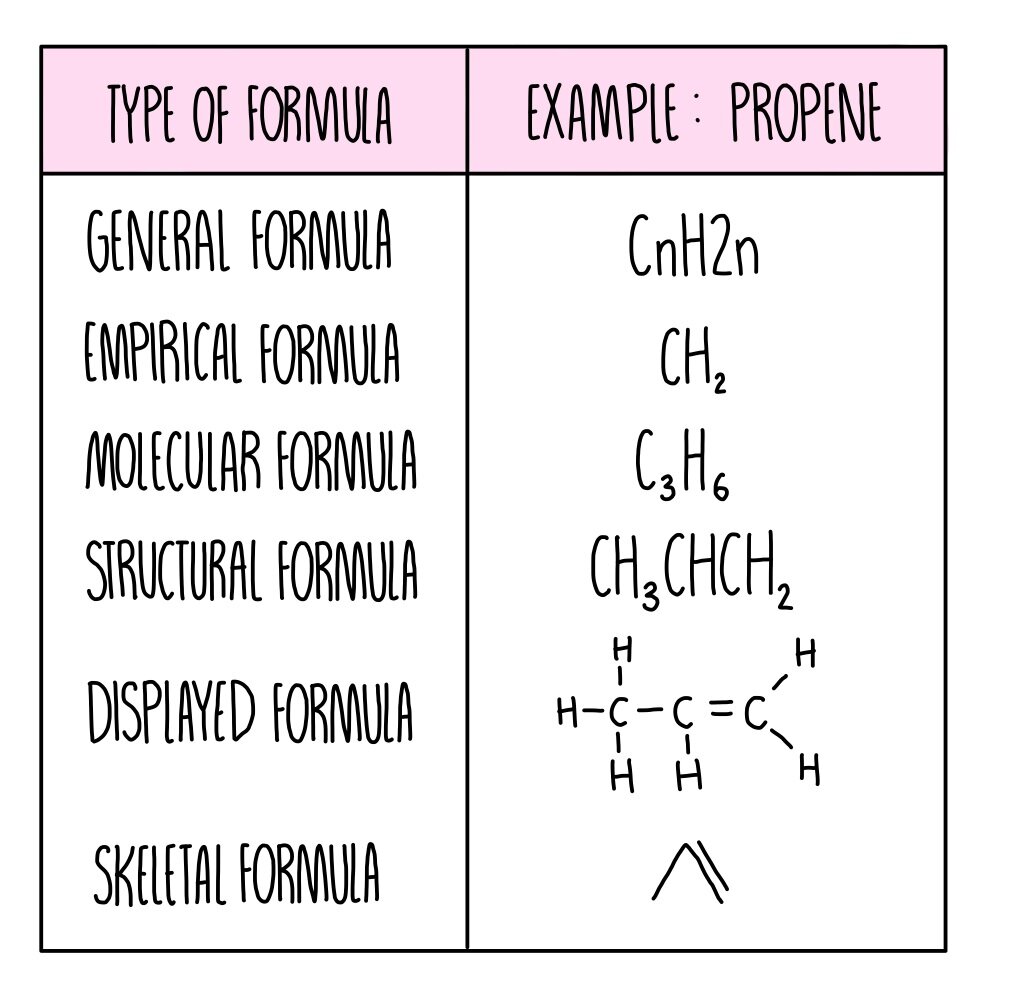
What is a molecular formula?
Gives the actual numbers of atoms of each element in a compound
What is an empirical formula?
Gives the simplest whole number ratio of atoms ofeach element in a compound
What is a displayed formula?
Shows all atoms and all bonds in a molecule
What is a structural formula?
Shows the arrangement of atoms in a formula
What is skeletal formula?
Carbon atoms are not drawn and C-H bonds aren’t drawn. The vertices represent the carbon atoms.
How would you show double bonds or elements beside carbon and hydrogen in skeletal formulae?
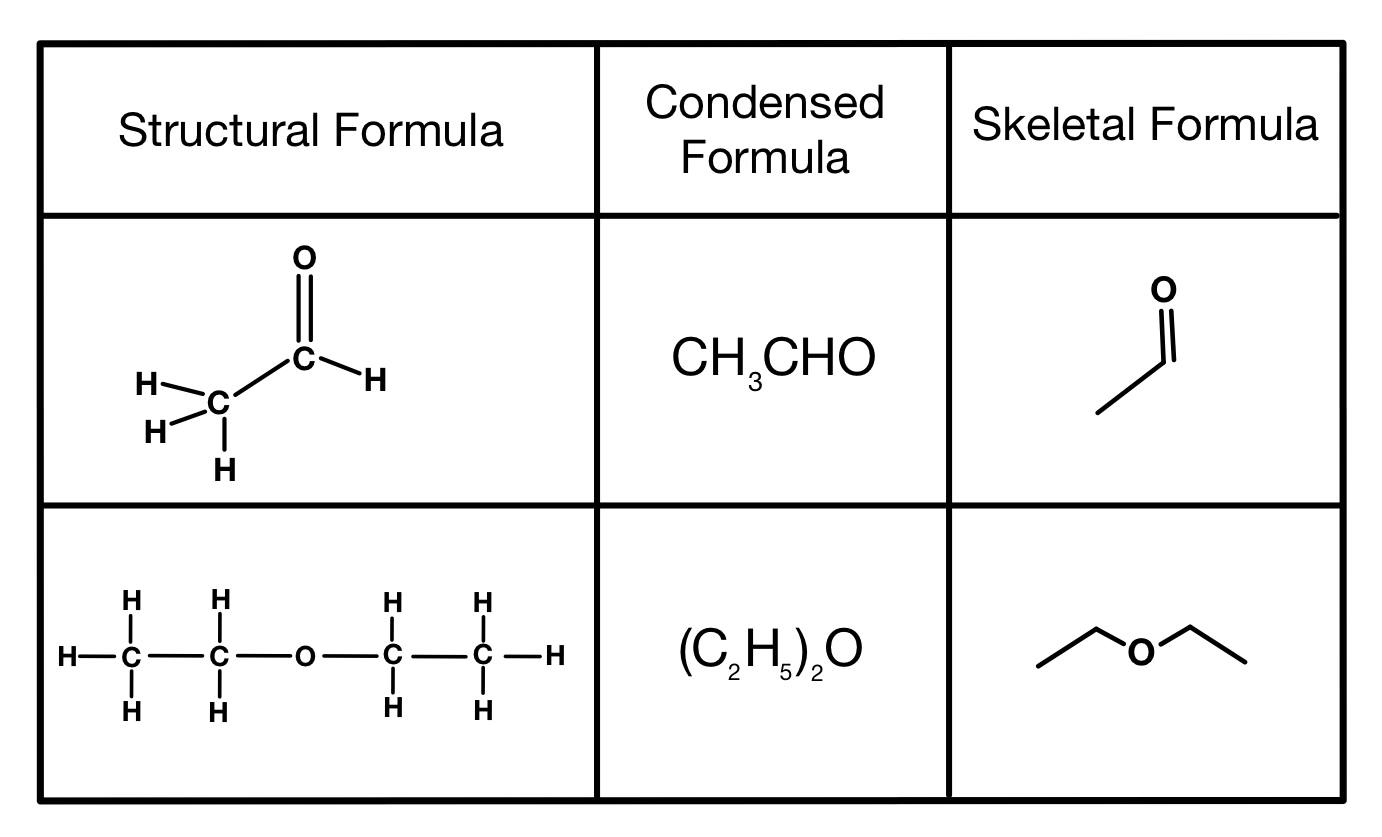
How would you add functional groups to skeletal formulae?
Draw a line down and add the functional group to the end in its structural formula
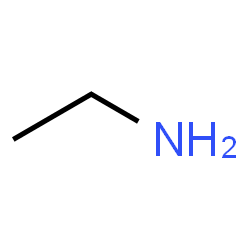
Skeletal formula for ethylamine - CH3CH2NH2
What is homologous series?
A family of compounds with the same functional group but different chain lengths
What are functional groups?
An atom of group of atoms responsible for the characteristics/reactivity of molecules
General formula for alkanes
CnH2n+2
What is the functional group for alkanes?
No functional group
General formula for alkenes
CnH2n
What is the funtional group for alkenes?
C = C bond
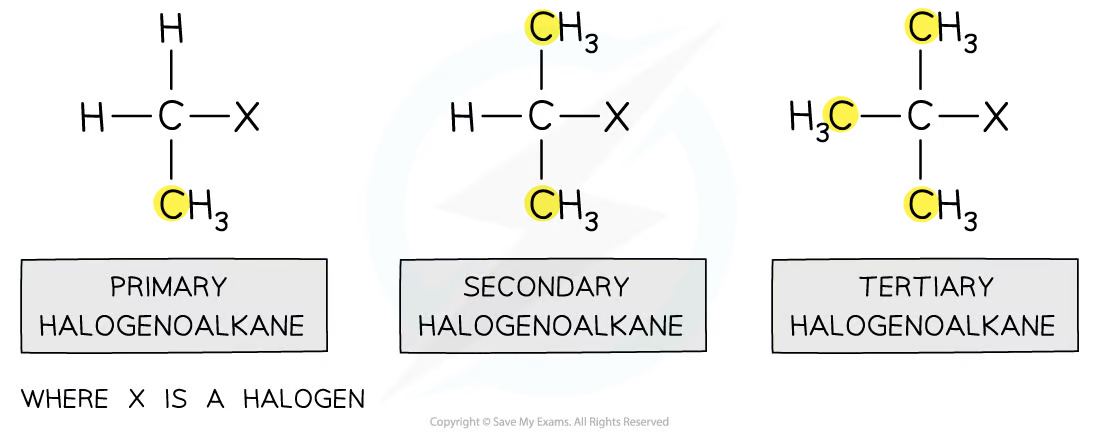
What are halogenoalkanes?
Alkanes with a halogen attatched to them
General formula for halogenoalkanes
CnH2n+1X
What is the functional group for halogenoalkanes?
C - X
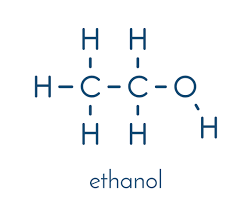
General formula for alcohols
CnH2n+2O
What is the functional group for alcohols?
R - OH
R represents the carbon chain
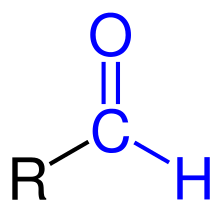
General formula for aldehydes
CnH2nO
What is the functional group for aldehydes?
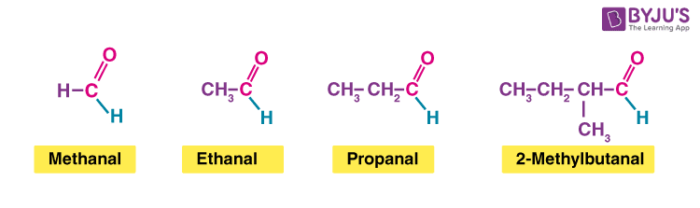
Homologous series of aldehydes
What is the general formula of ketones?
Same as aldehydes - CnH2nO
What is the functional group for ketones?
The carbonile group (C=O) is in the middle of the chain

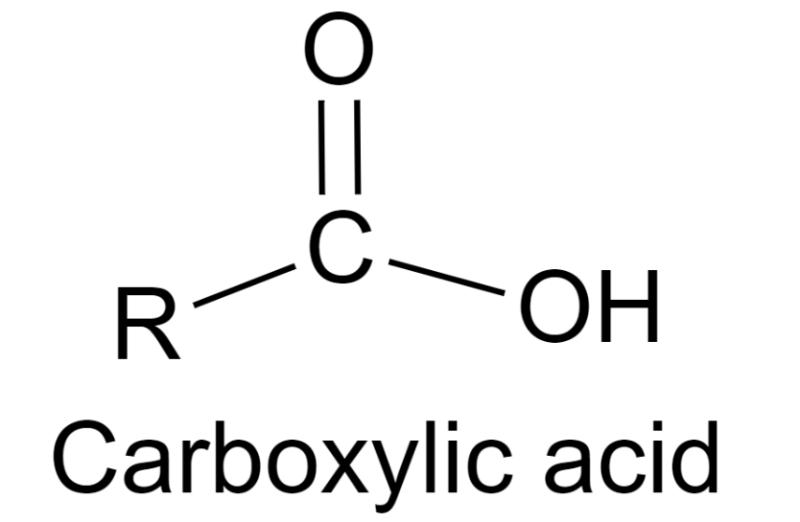
General formula for carboxylic acids
CnH2nO2
What is the functional group for carboxylic acids?
General formula for amines
CnH2n+2N
What is the functional group for amines?
R - NH2
Methylamine
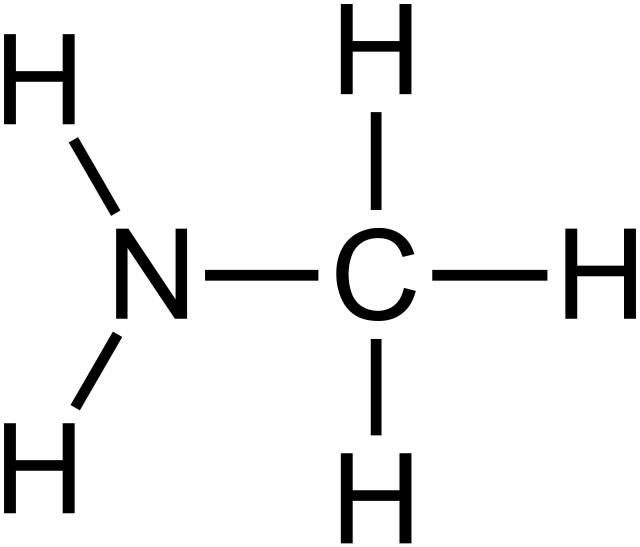
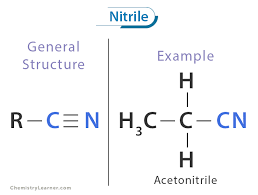
General formula for nitriles
CnH2n-1N
What is the functional group for nitriles?