Pharm: CNS (parkinson's, alzheimer's, multiple sclerosis)
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
46 Terms
CNS drugs
drugs that act in the brain and spinal cord
how many nuerotransmitters are in the CNS
at least 21, and we do nit fully understand the function of all of them
what can pass the Blood brain barrier
only lipid-soluble drugs
protein bound or ionized drugs cannot
prolonged drug exposure In the CNS
prolonged drug exposure can lead to different effects
many psych drugs must be taken for several weeks before therapeutic effects develop
side effects may decrease but the therapeutic effects will not
Tolerance
decreased response occurring during the course of prolonged use
physical dependance
state in which abrupt discontinuation of drug use will precipitate a withdrawal syndrome
process of developing new psychotherapeutic drugs
structural analogs synthesized (modifying existing drugs)
biochemical and physical screening tests
serious toxicity ruled out then tested in humans
small advances vs major breakthroughs
what is parkinsons
neurodegenerative disorder of the Extrapyramidal system (controls involuntary movement, posture and tone)
gets worse over time, there is no cure
motor symptoms of Parkinsons
tremors at rest
rigidity
postrural instability
bradykinesda
non-motor symptoms of parkinson’s
autonomic disturbances
depression
psychosis and dementia
what causes parkinson’s
the degradation of neurons that supply dopamine to the striatum
balance of ACh and dopamine disrupted.
the disrupted balance balance causes overactivity of gamma-aminobutyric acid
causing motor symptoms
what 2 drug categories are used to treat Parkinsons
dopaminergic drugs - more commonly used
anticholinergic drugs
goal of treatment for PD
no cure exists to prevent further degradation of dopamine nuerons
goal is to improve ability to carry out ADLs
dopamine replacement drug
levodopa - precursor to dopamine
crosses BBB (which dopamine cannot do) and converts into dopamine in the brain
first line for severe symptoms
drawback to levodopa
beneficial effects diminish over time
may cause dyskinesias
how are levodopa induced dyskinesias managed
reduce the dose
give amantadine - NMDA agonist
last resort - surgery and electrical stimulation
carbidopa
decarboxylase inhibitor of levodopa in intestine and peripheral tissues
makes more levodopa available to the CNS
cannot cross BBB
has no therapeutic effect on its own, only with levodopa
Dopamine agonists
directly activate dopamine receptors in the striatum
first choice for mild-moderate symptoms
2 groups: ergot derivative and nonergot derivative
drawbacks of dopamine agonists
side effects: hallucinations, daytime sleepiness, orthostatic hypotension
usually only given to younger patients as they tolerate side effects better
DA: non ergot derivative
apomorphine
highly selective for dopamine receptors
preferred over ergot derivatives
apomorphine
non ergot DA
approved for hypo mobility during “off“ episodes of patients with advanced Parkinsons
DA: ergot derivative
bromocriptine
less selective
causes adverse effects bc it block serotonin and alpha receptors
bromocriptine
direct acting dopamine agonist
ergot derivative
when combined with levodopa, it can prolong therapeutic response and reduce motor fluctuations
MAO inhibitor
seligine
selective irreversible inhibition of MOA-B (the enzyme that breakd down dopamine in the striatum)
considered first line, though improvement of motor functions are modest
reduces the wear off effect when combined with levodopa
NMDA receptor agonist
amantadine
helps manage levodopa induced dyskinesia
promotes release of dopamine, but we are unsure of the mechanism
only NMDA antagonist approved for parkinson’s
alzhiemers disease
progressive memory loss
impaired thinking
neuropsychiatric symptoms
inability to perform routine ADLs
physiology of AZ
degrading nuerons
reduced cholinergic transmission (ACh 90% below normal)
beta-amyloid and neurotic plaques for outside of neurons
nueorfibrillary tangles and tau from inside neurons
how do neurons degenerate in AZ
the degenerate early in the hippocampus affecting memory
later in the cerebral cortex affecting speech, perception, reasoning and higher functioning
Donazepil
indicated for mild, moderate and severe stages of AZ
reversible Cholinestrase inhibitor
causes enhanced transmission of cholinergic nuerons that have not yet been destroyed
does stop stop progression of AZ
Memantine
indicated for moderate and severe stages of AZ
NDMA receptor antagonist
firs drug in new class
what Is multiple sclerosis
chronic inflammatory autoimmune disorder that damages myelin sheaths in the CNS
causes variety of sensory and motor deficits
unknown cause, but may be linked to Epstein-barr virus
demyelination causes
slowed or blocked axonal conduction
inflammation mechanism in MS
lymphocytes (T cells ) and macropahges, stick to CNS blood vessels
they then migrate across the BBB and initiate the inflammatory process
destroys myelin, axonal membrane and ogliodendrocytes
after an acute inflammatotion effect
some degree of recovery occurs
partial remyelination
functional axonal compensation
brain developed alternate circuits that bypass damaged region
the more episodes = the less recovery , irreversible injury
what happens to the myelin sheaths in the PNS
they are unaffected by MS bc they are made by Schwann cells not ogliodendrocytes
S/S of multiple sclerosis
parestesia
mucscle/ motor issues
visual impairment
bladder and bowel issues
sexual dysfunction
fatigue
cognitive impairment / depression
slurred speech
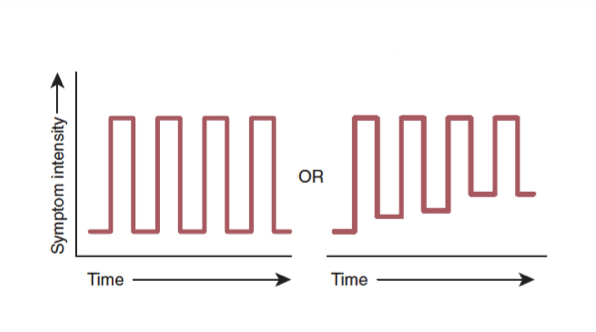
this graph represents?
relapsing - remitting MS
clearly defined episodes
periods of partial or full recovery
85%-90% have this form initially
affects 2x as many women as men
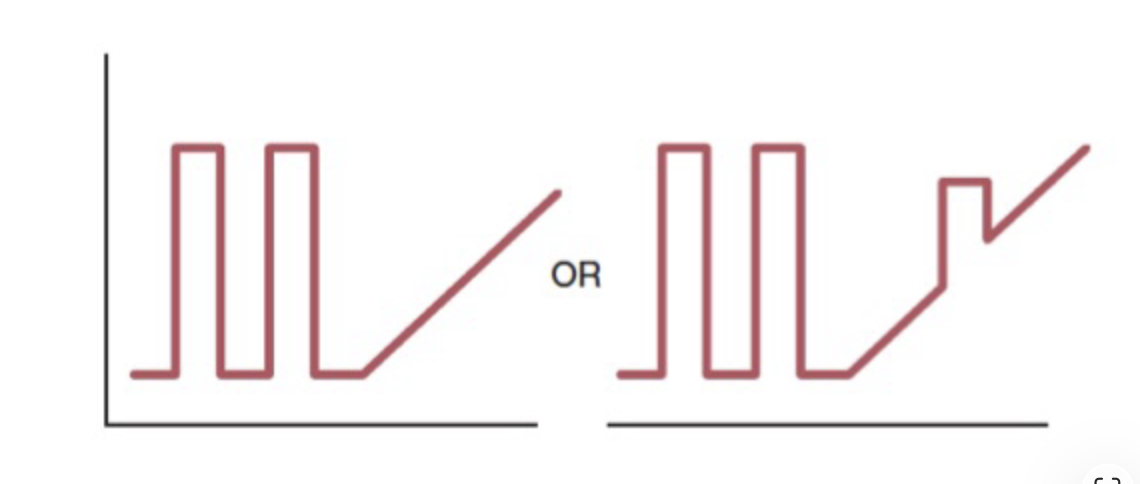
this graph represents?
secondary progressive MS
pt with relapsing -remitting MS develops steadily worsening dysfunction without occasional plateaus, acute exacerbations, or minor remissions
within 10-20 years 50% of people with relapsing -remitting MS develop secondary progressive MS
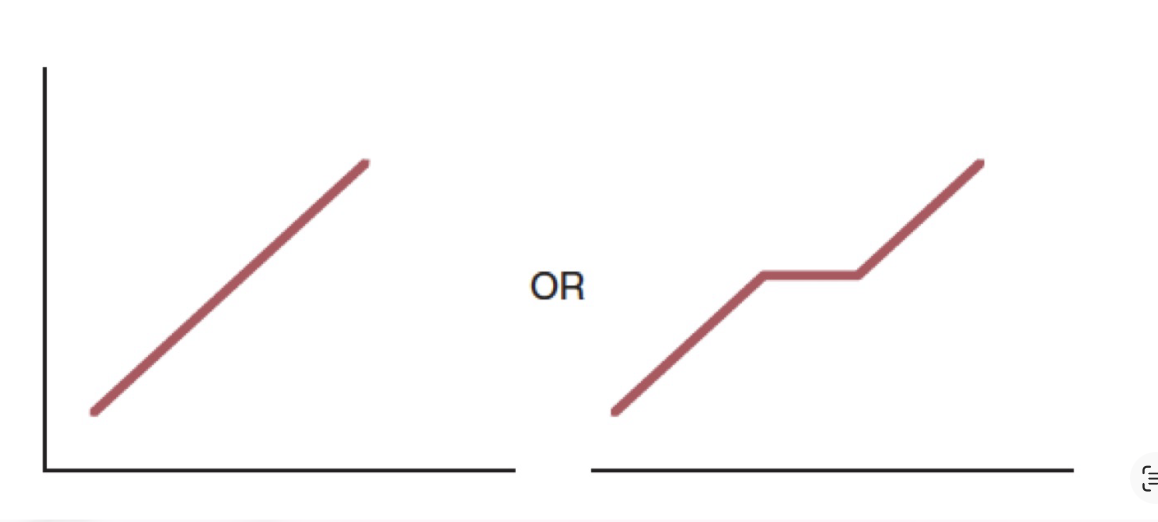
this graph represents?
primary progressive MS
symptoms grow progressively more intense from the onset
10% of MS
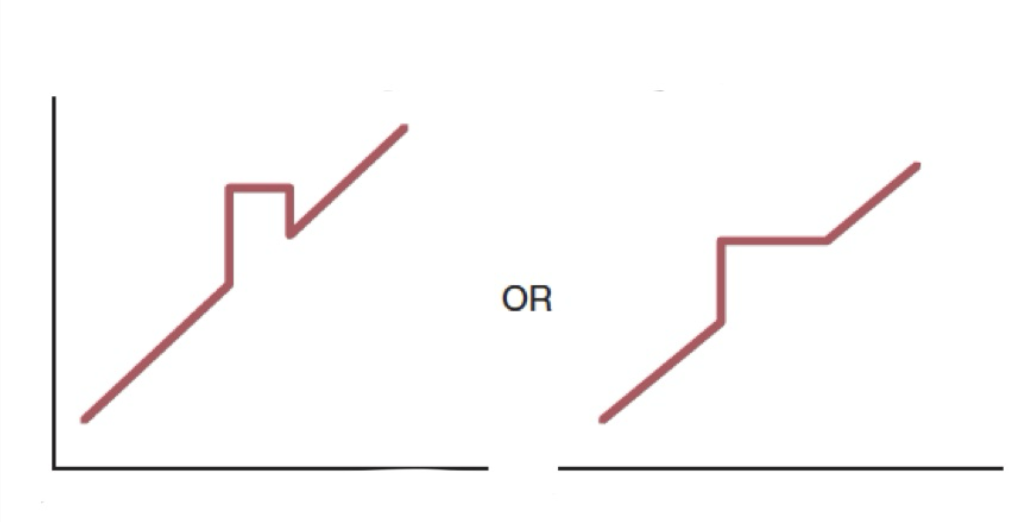
this graph represents?
progressing-relapsing MS
what 2 drug categories are used to treat MS
immunomodulators
immunosuppressants - more toxic
immunomodulators
tysabril - blocks adherence of leukocytes to CNS blood vessels, so they can’t cross BBB
interferons
interferons
naturally occurring glycoprotein with antiviral, anti proliferative and immunomodulatory actions
inhibits migration of inflammatory leukocytes across BBB
suppresses T-helper cell activity
Avonex (interferon beta 1a)
IM injection
contains 166 amino acids and glycoprotein
identical to human interferon data
Betaseron (interferon beta 1b)
SubQ injection
contains 156 amino acids and has no glycoproteins
risks of immunomodulators
formation of neutralizing antibodies (body sees as foreign substance)
hypersensitivity/ allergic reactions
infections
hematologic changes (low WBCs)
liver injury
vaccines are less effective