Psych/Nuero Final: Cav MS, ALS, MG
1/109
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
110 Terms
Multiple Sclerosis (MS): autoimmune, inflammatory disease of __
CNS

4 Different Types of MS:
1. ____-____ (most common!!, unpredictable attacks which may or may not leave permanent deficits followed by periods of remission)
relapsing-remitting

4 Different Types of MS:
2. ____ ____ (steady increase in disability without attacks)
primary progessive

4 Different Types of MS:
3. ___ ____ (initial relapsing-remitting that suddenly begins to have decline WITHOUT periods of remission)
secondary progressive

4 Different Types of MS:
4. ____-___ (steady decline since onset with superimposed attacks)
progressive-relapsing
MS Symptoms (5)
cognitive dysfunction, vision problems, balance issues, chronic fatigue, depression
What Happens In MS
In multiple sclerosis, the immune system attacks and destroys the ___ ___ around ____ in the CNS, leading to loss of nerve ____ _____ and formation of scarred demyelinated areas called ____.
myelin sheath, nerves, signal conduction, plaques
2 Steps Required to Induce Immune Response in MS
pro-inflammatory milieu in CNS, antigen-driven acquired immune response
Immune Reponse in MS
1. Antigens released from the CNS are taken up and presented by ____ ____, which then ______ → ___ and ___ cells in ____ ____ ____
dendritic cells, prime T, B, peripheral lymphoid tissues
Immune Reponse in MS
2. After the T and B cells are primed → they rapidly _____ (___ ___) in the _____ ____
multiply, clonal expansion, lymphoid tissue
Immune Reponse in MS
3. After clonal expansion in the lymphoid tissue → T and B cells _____ the ___
infiltrate, CNS
Immune Reponse in MS
4. After infiltrating CNS → B cells re-encounter their antigen, mature to plasma cells, and release large amounts of ___ antibodies, which bind to soluble or membrane-bound __ on expressed cells
IgG, antigen
Immune Reponse in MS
5. After infiltrating CNS → CD8+ T cells invade the ___ and encounter their specific peptide ligand which prompts direct __ to expressing cells
brain, damage
Immune Reponse in MS
6. After infiltrating CNS → CD4+ T cells encounter antigens presented by microglial cells, which lead to increased production of ____ ___ that then ___ other immune cells (like macrophages)
inflammatory cytokines, attract
As clinical efficacy of MS Treatments increases, so does ___ concerns
safety
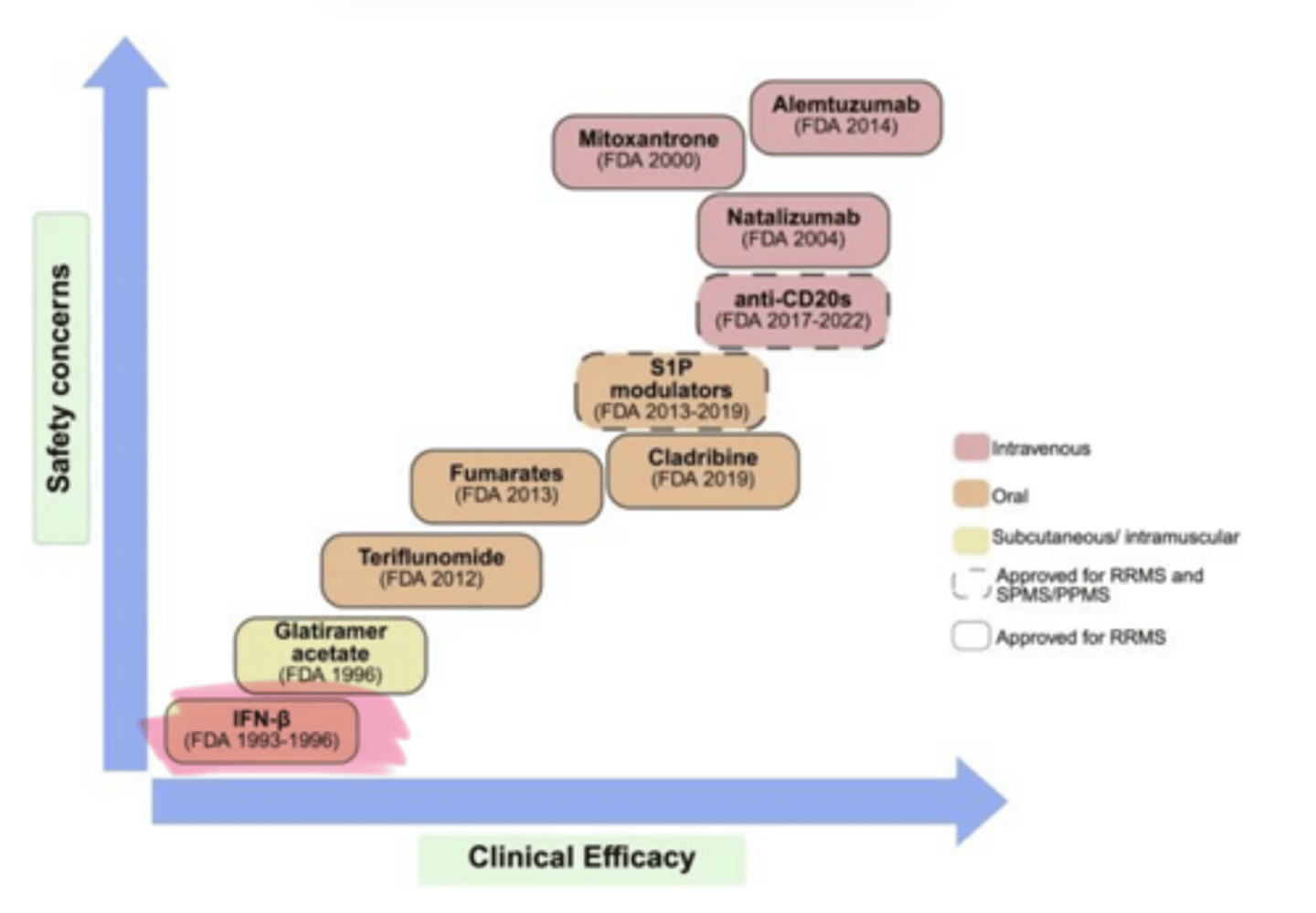
Interferon β-___ (Avonex, Rebif) and Interferon β-___ (Betaseron) are used in multiple sclerosis
1a, 1b
How do IFNβ drugs work in the periphery?
1. decreases T cell ____
2. decreases T cell ___ and ___ of the BBB
activation, adhesion, penetration
How do IFNβ drugs work in the CNS?
3. decreases antigen ___
4. promotes anti-inflammatory __ production
presentation, cytokine
How do IFNβ drugs work in the CNS?
5. decreases ___ cytokine release
6. decreases B-cell ___
Th1, proliferation
How do IFNβ drugs work in the CNS?
7. decreases bystander ___
damage
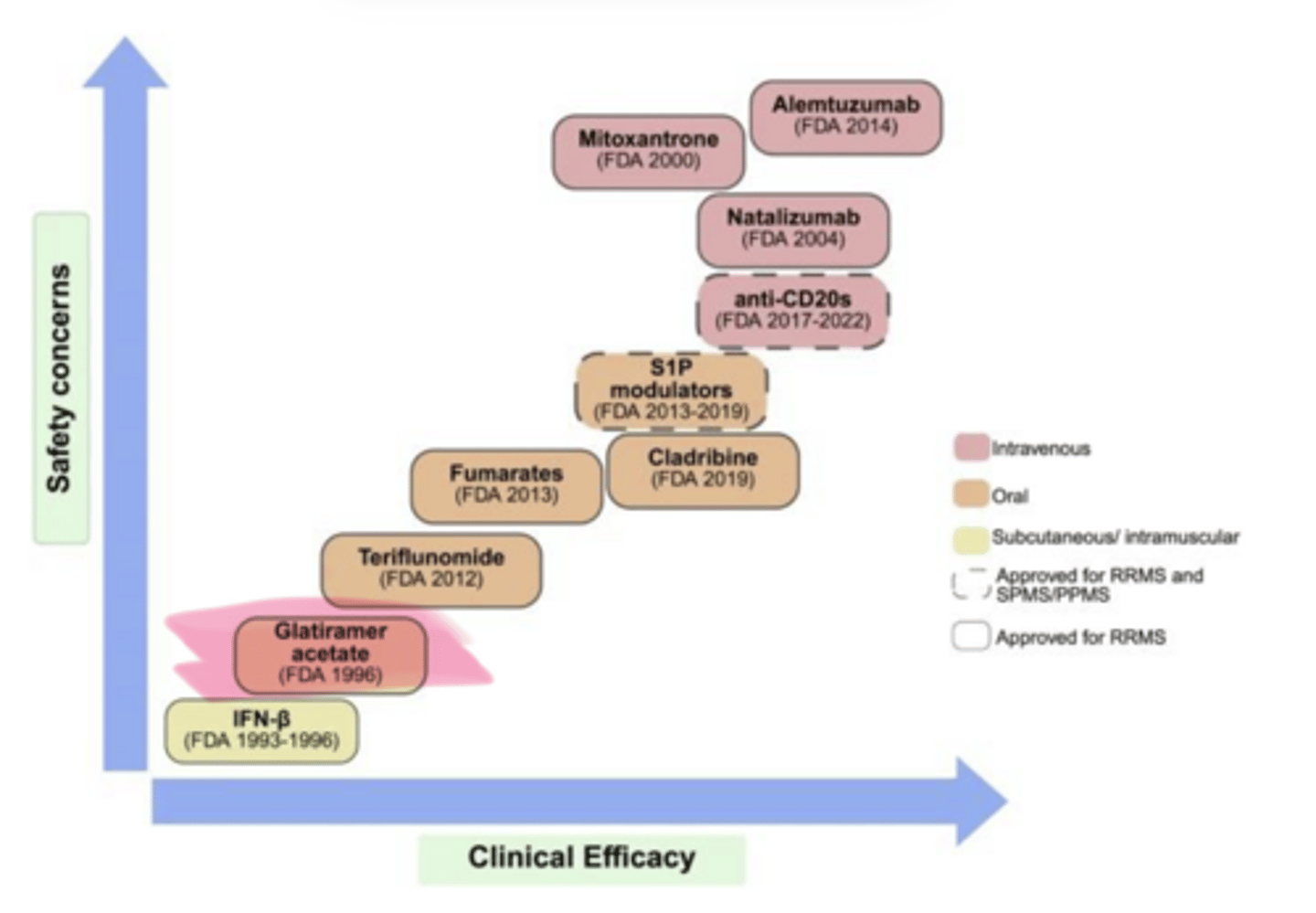
____ (Copaxone) is used in multiple sclerosis
Glatiramer
Glatiramer cannot penetrate the ___
BBB
Glatiramer acetate acts in the periphery as a "___" antigen (it resembles myelin basic protein, so T cells react to it instead of attacking the actual myelin in the CNS.)
decoy
Glatiramer shifts T-cell response from pro-inflammatory Th1 cells to anti-inflammatory ___ cells
Th2
Glatiramer
-the anti-inflammatory Th2 cells migrate into the CNS and release anti-inflammatory ____ and ___ factors (promote neuronal/glial survival)
cytokines, trophic
Glatiramer
-the result of peripheral Th2 cells crossing the BBB and releasing anti-inflammatory cytokines and trophic factors = "____ ____"
bystander suppression
"bystander suppression" refers to the reduction of ____ as a result of peripheral Th2 cells crossing the BBB and releasing anti-inflammatory cytokines and trophic factors
inflammation
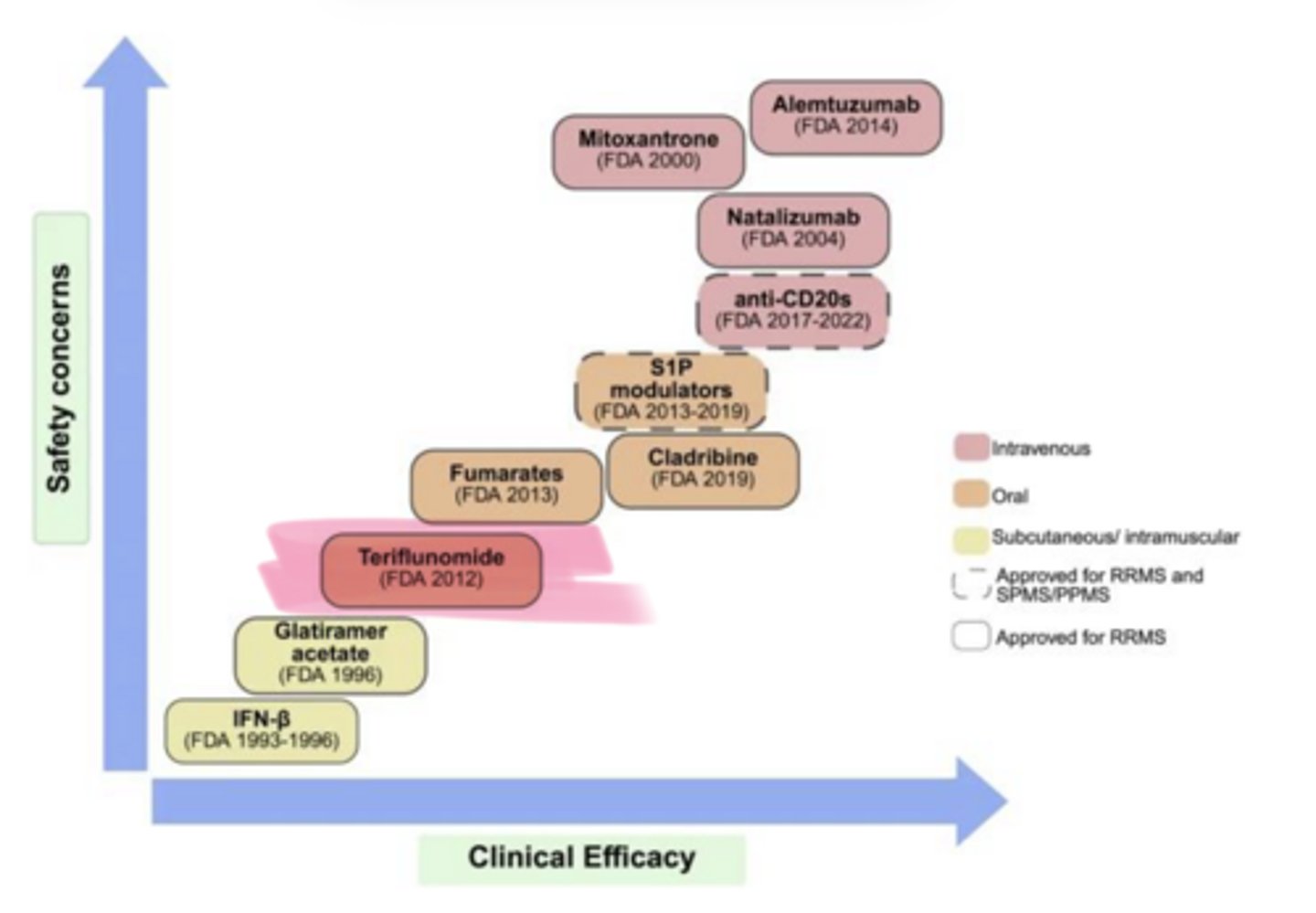
____ (Aubagio) is used in multiple sclerosis
Teriflunomide
Teriflunomide MOA
1. Inhibits dihydroorotate dehydrogenase (___) in mitochondria → resulting in decreased de novo ___ synthesis)
DHODH, pyrimidine
Teriflunomide inhibits DHODH to decrease pyrimidine synthesis → effect = less production of ___, ___, and ___, which ultimately impairs cell function and leads to cell death.
glycoproteins, phospholipids, nucleotides
Teriflunomide MOA
2. Inhibits ___-___ signaling pathway → resulting in decreased production of pro-inflammatory ___
JAK-STAT, cytokines
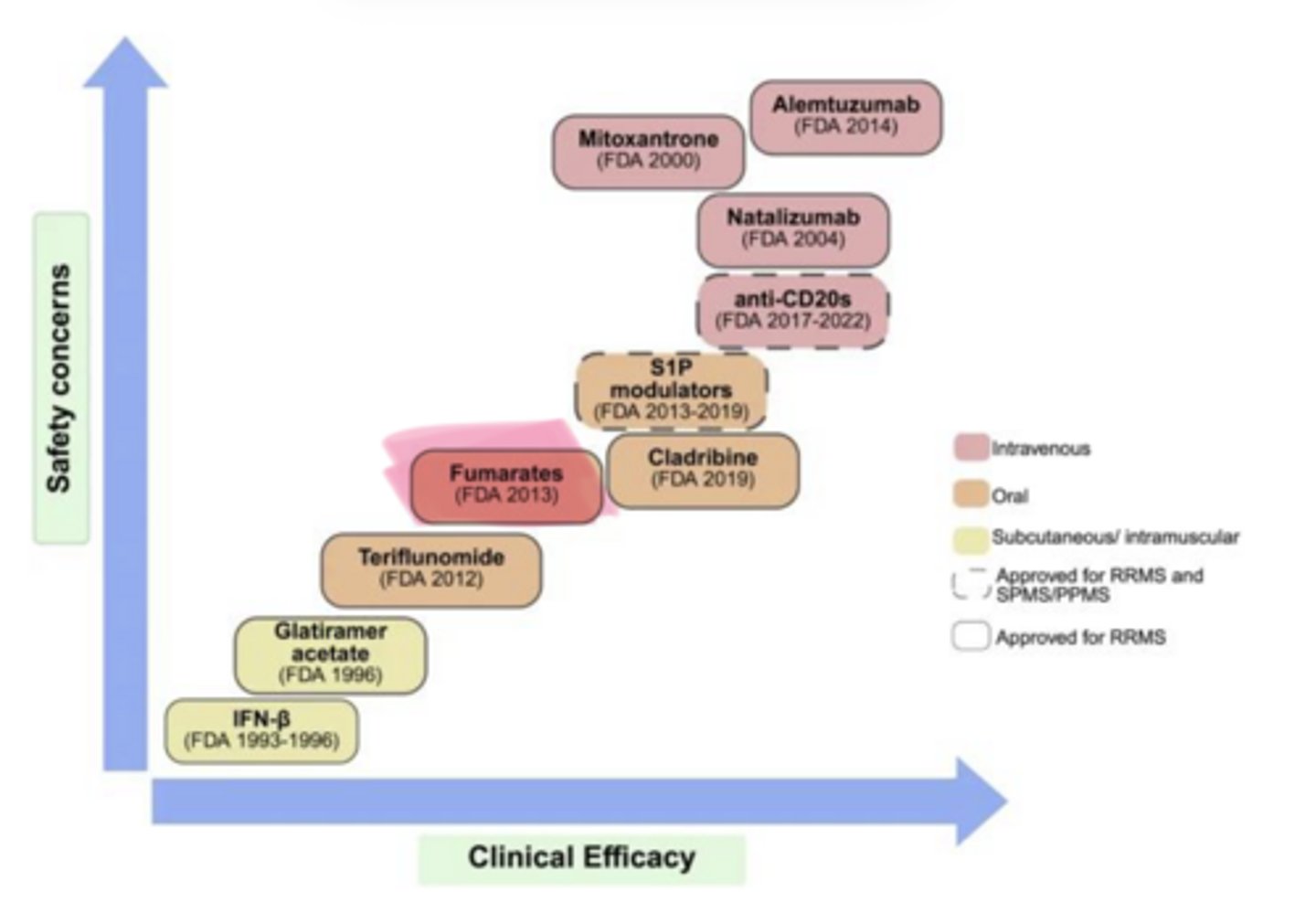
____ ___ (Tecfidera) is used in multiple sclerosis
Dimethyl Fumarate
Dimethyl Fumarate
-MOA is similar to glatiramer → Shifts immune balance in the periphery by increasing ___ anti-inflammatory cells and decreasing pro-inflammatory T and B cells to promote ___ ___ in the CNS
TH2, bystander suppression
Dimethyl Fumarate
-Remember glatiramer cannot cross BBB. The key difference with dimethyl fumarate is that it can enter the ___
CNS
Dimethyl Fumarate
-After entering the CNS, the drug activates ___ signaling pathway (ERK pathway) to support neuronal and glial cell survival
kinase
Dimethyl Fumarate
-By activating kinase pathways, it increases anti-apoptotic and antioxidant factors, which reduces ___ ___ and inflammation in the brain.
oxidative stress
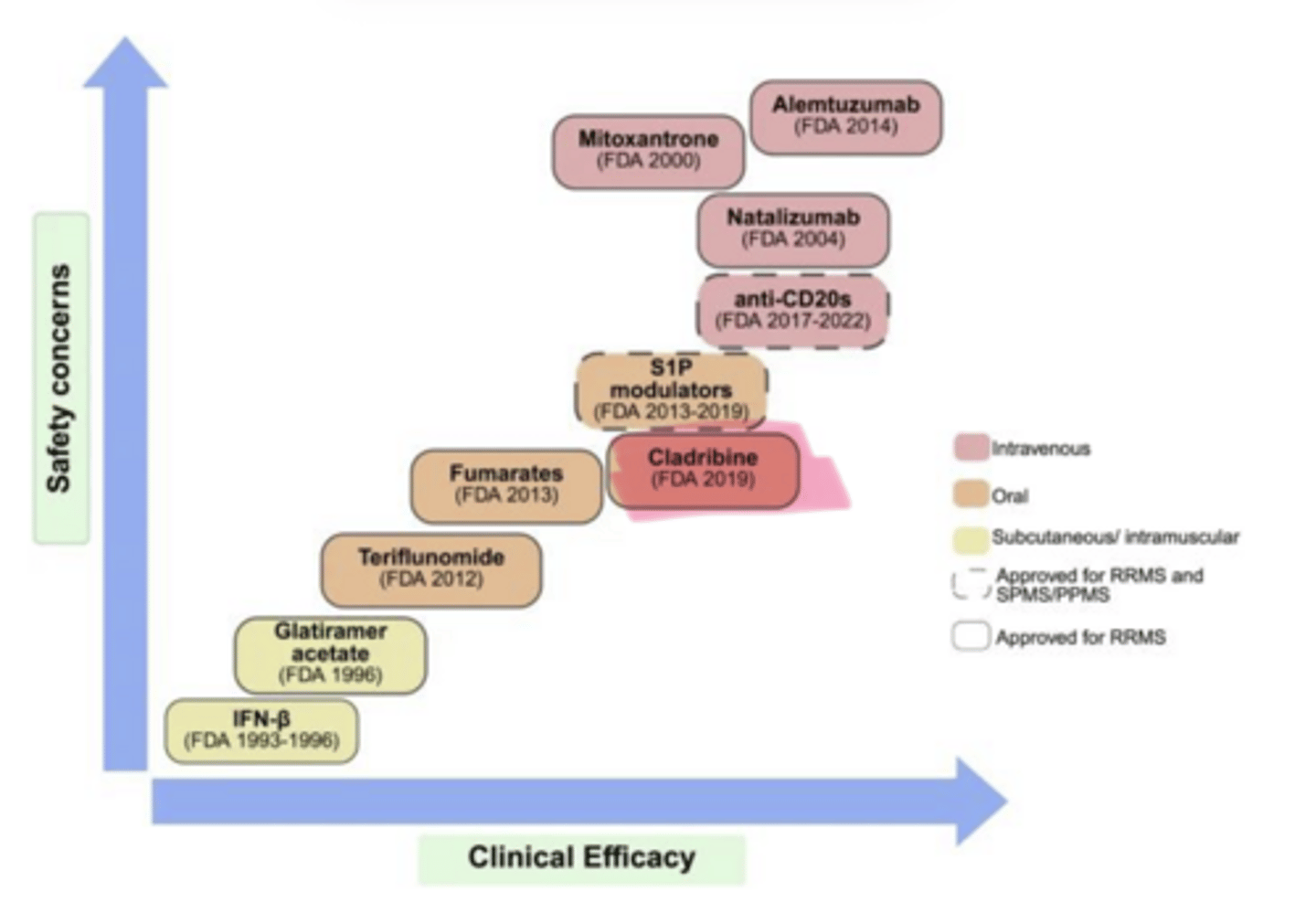
____ (Mavenclad) is used in multiple sclerosis
Cladribine
Cladribine is activated by ___
kinases
Cladribine is inactivated/degraded by ___
phosphotases
Cladribine
-in ___ (normal body) cells, there is low kinase and high phosphatase, meaning cladribine gets inactivated/degraded and has minimal efffect
somatic
Cladribine
-is preferentially activated in ___ due to higher kinase and lower phosphatase activity,
lymphocytes
Cladribine MOA
-selectively accumulates in lymphocytes, where it disrupts ___ synthesis/repair and causes cell death.
DNA
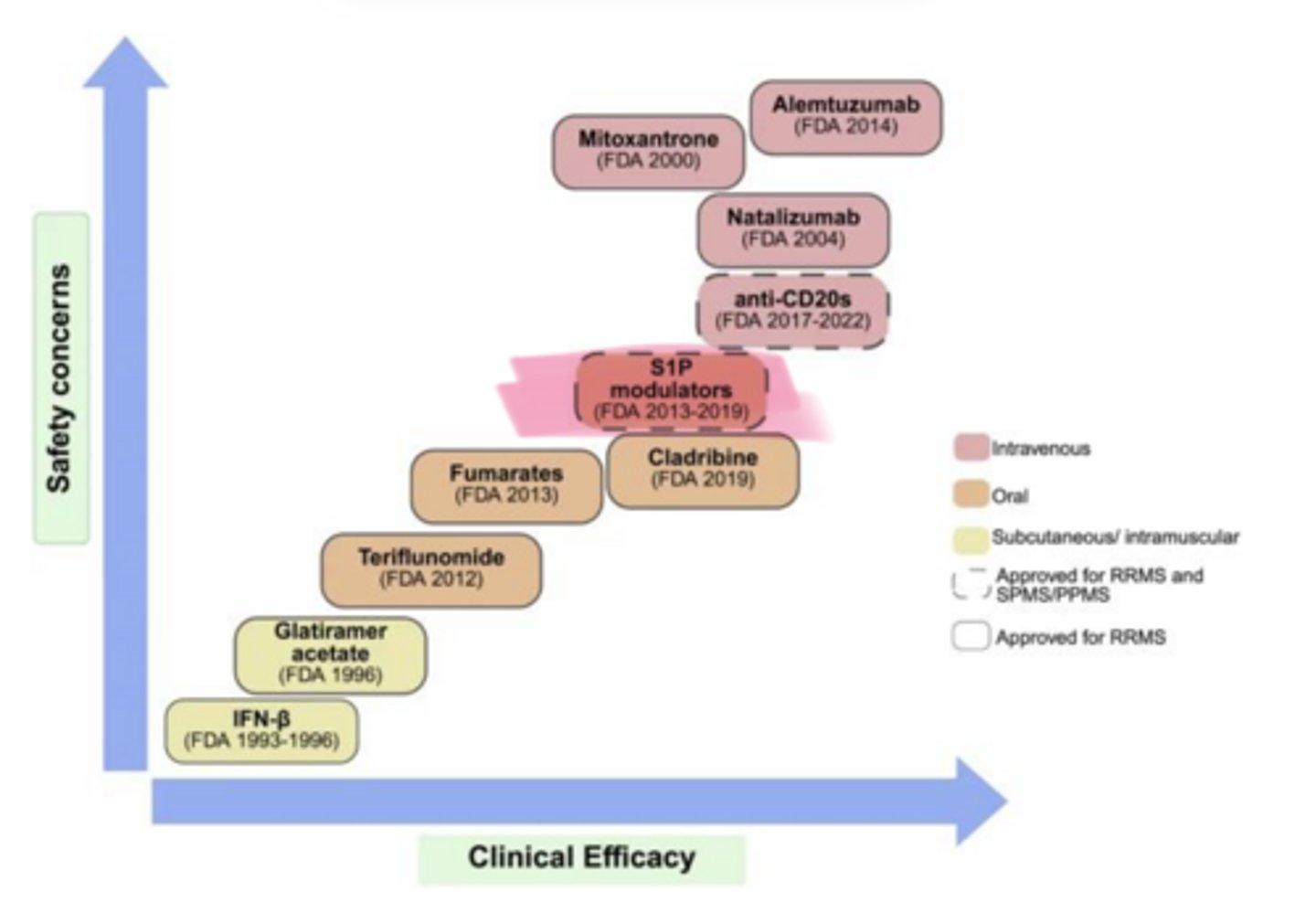
____ (Gilenya) and ___ (Mayzent) are used in multiple sclerosis
Fingolimod, Siponimod
Fingolimod, Siponimod
-Normally, T cells are activated in lymph nodes and are signaled to leave (egress) by ___ binding to its receptor
S1P
Fingolimod, Siponimod MOA
-prevent T cell egress from lymph nodes by antagonizing ___ receptors
S1P
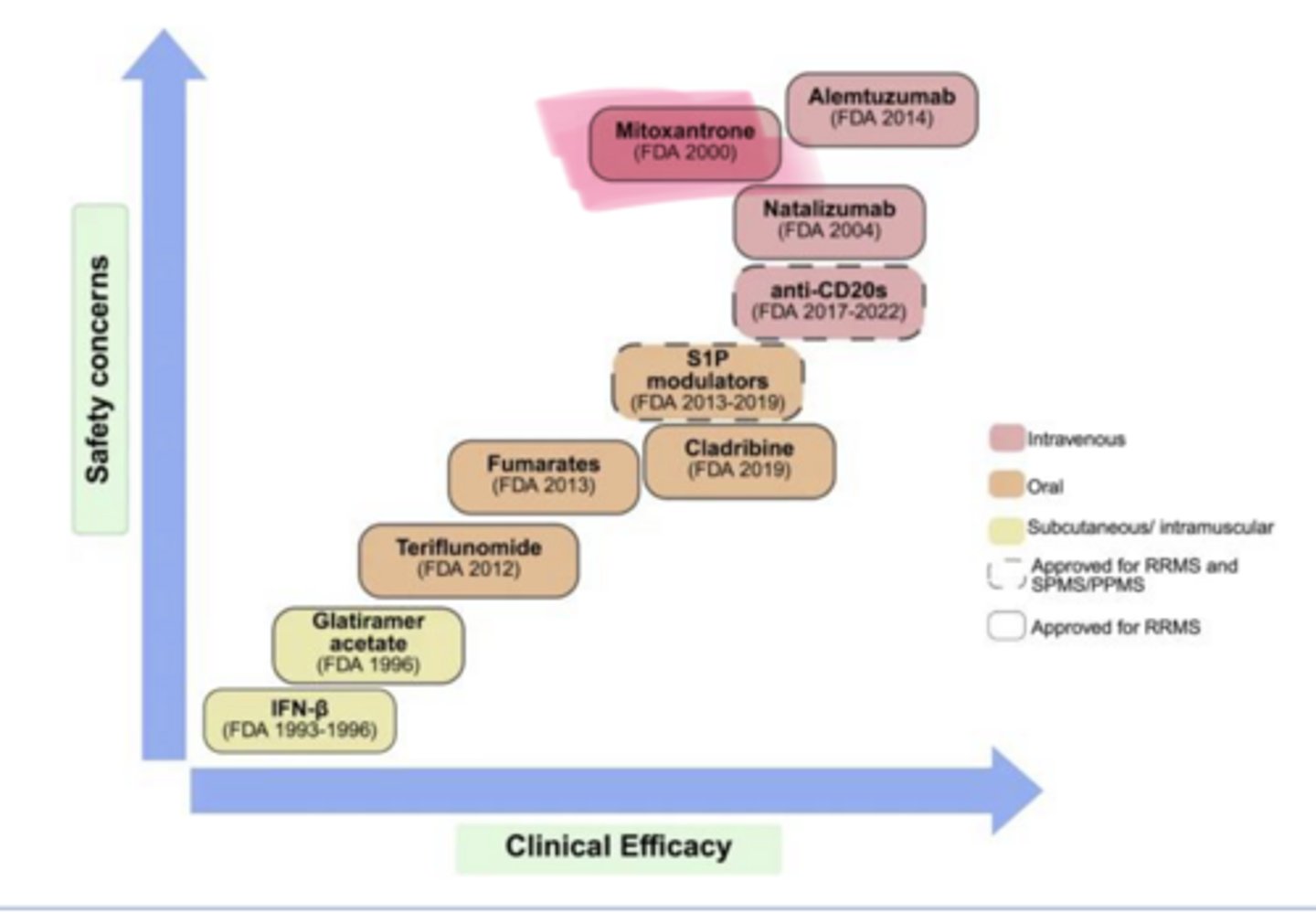
____ (Novantrone) is used in multiple sclerosis
Mitoxantrone
Mitoxantrone
-intercalates (ie inserts itself) into __
DNA
Mitoxantrone
-In DNA, has a __-___ ___ effect ("like dropping a bomb on immune response")
non-specific cytotoxic
Mitoxantrone Effects in DNA:
1. ↓ proliferation of __ cells, __cells, and ___
T, B, macrophages
Mitoxantrone Effects in DNA:
2. Impairs ___ presentation
antigen
Mitoxantrone Effects in DNA:
-↓ secretion of ___-___ __
pro-inflammatory cytokines
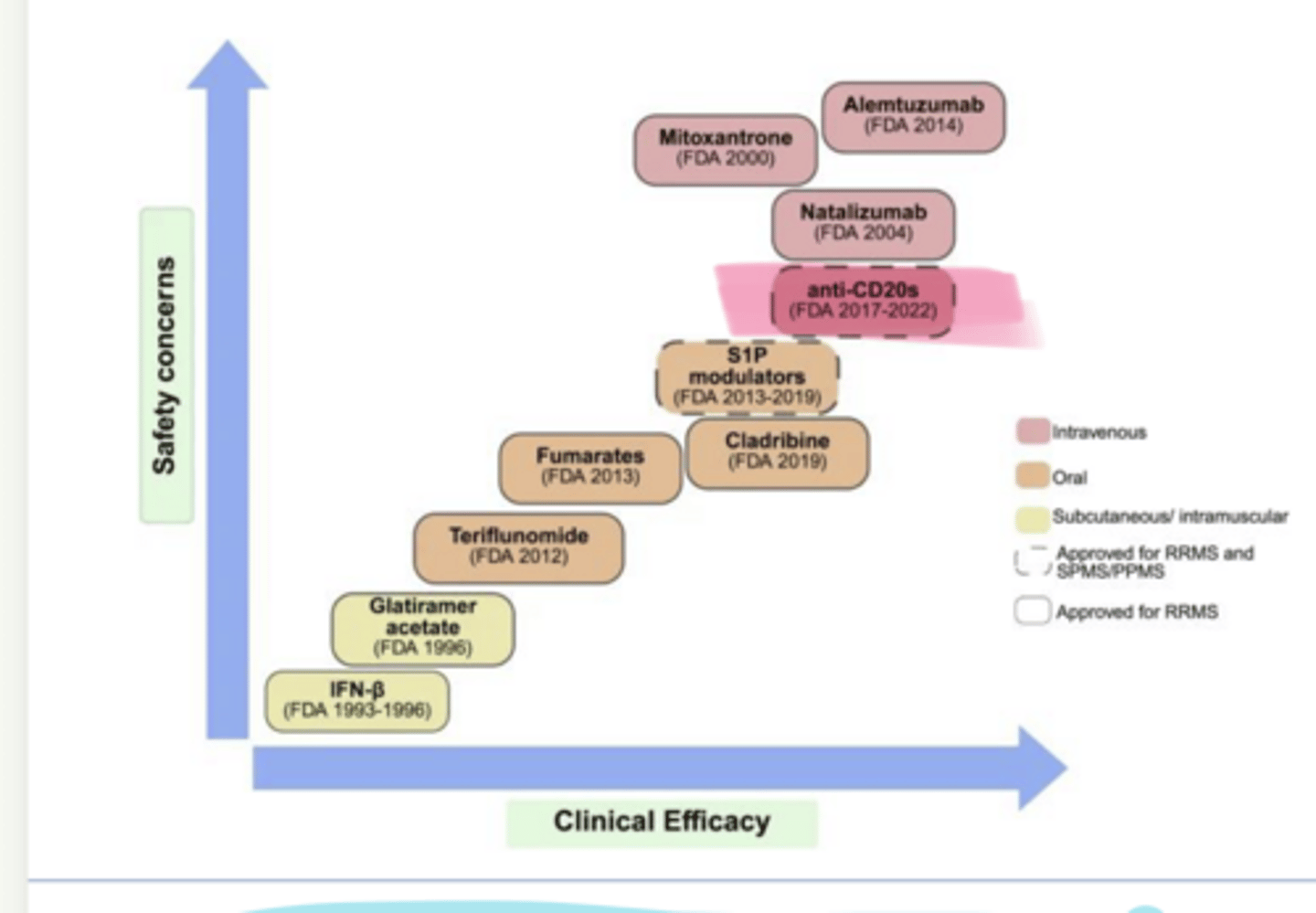
____ (Ocrevus), ____ (Kesimpta), and ____ (Briumvi) are used in multiple sclerosis
ocrelizumab, ofatumumab, ublituximab
Ocrelizumab, Ofatumumab, and Ublituximab bind to ___ on __ cells
CD20, B
Ocrelizumab, Ofatumumab, and Ublituximab bind to CD20 on B cells. This results in ___ of B cells
depletion
Ocrelizumab, Ofatumumab, and Ublituximab deplete B cells in 3 ways:
1. ___-dependent cell-mediated cytotoxicity
2. ___-mediated cytotoxicity
3. ___ induction
antibody, complement, apoptosis
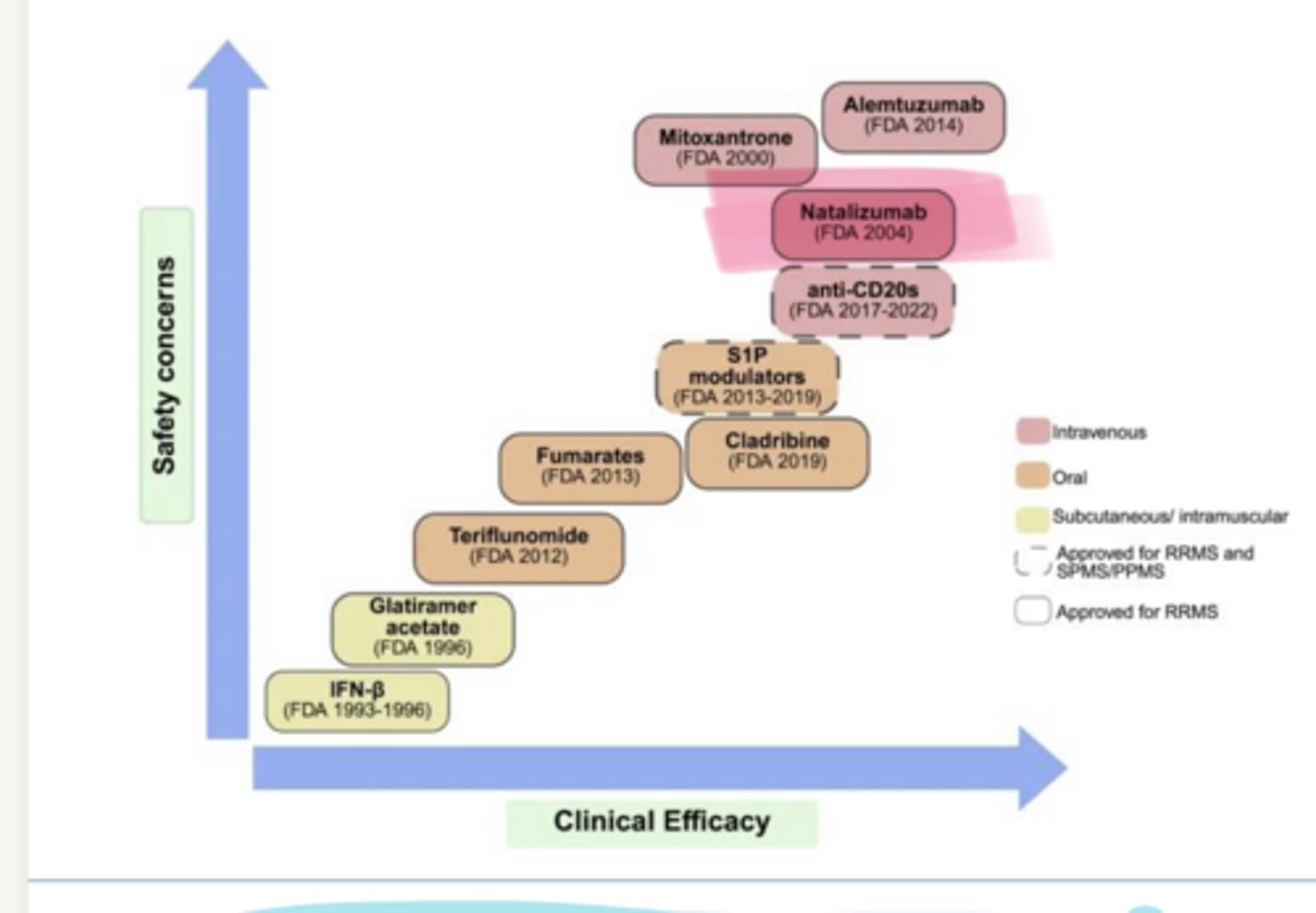
____ (Tysabri) is used in multiple sclerosis
Natalizumab
Natalizumab
-Monoclonal antibody against α4β1 integrin on __ __ (not CD20 like the B-cell drugs)
T cells
Natalizumab
-Blocks T-cell adhesion to ___ on endothelial cells
VCAM-1
Natalizumab
-Final effect = decreased T-cell entry into the ___ (therefore decreased inflammation and myelin damage)
CNS
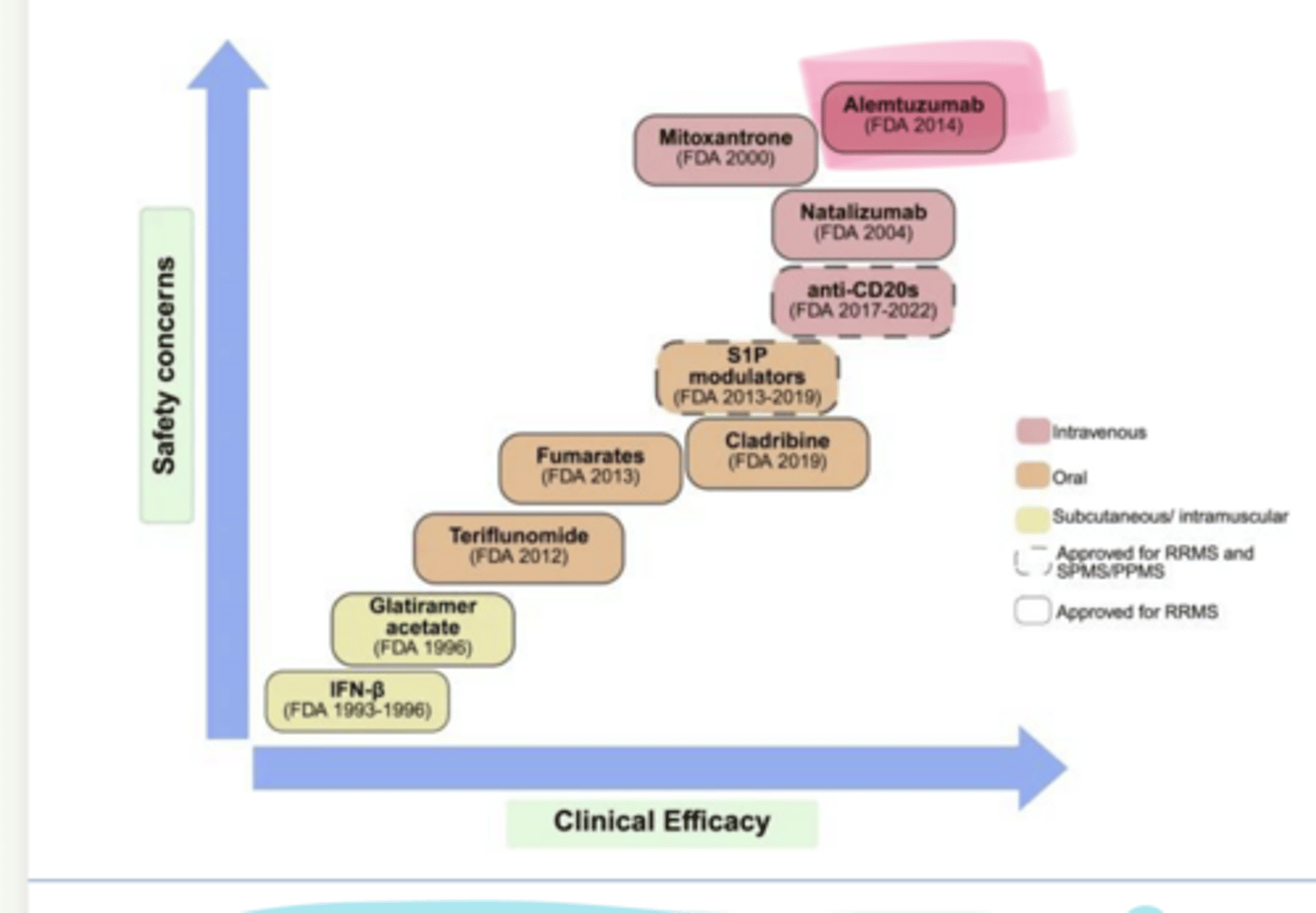
_____ is used in multiple sclerosis
Alemtuzumab
Alemtuzumab
-Monoclonal antibody against ___ on lymphocytes (B and T cells)
CD52
Alemtuzumab
-Causes lymphocyte depletion via ___-mediated cytotoxicity and
___-dependent cellular cytotoxicity
complement, antibody
Amyotrophic Lateral Sclerosis = __
ALS
ALS
-rapid, progressive muscle ___
-muscle ___
-spasticity
-compromised ___
weakness, atrophy, respiration
ALS-the following are spared
-___
-___
-___
sensory, cognitive, autonomic
ALS is ___ and usually ___
progressive, fatal
ALS
-a disease of __ and ___ motor neurons (patients have varying degrees of both)
upper, lower
ALS
-upper motor neuron disease → causes ___ called ___
stiffness, spasticity
ALS
-lower motor neuron disease → causes ___, loss of ____ (atrophy), and muscle ___ (fasciculations)
weakness, muscle, twitching
Bulbar ALS → primarily affects muscles involved in __, ___, and __ movements
speech, swallowing, tongue
ALS can be __ or __
sporadic, genetic
Sporadic ALS Etiology
-viral ___
-autoimmunity
-excitotoxicity (____ uptake)
-___ ___ toxicity
infection, glutamate, free radical
Genetic ALS Etiology
1. mutation in ___ (superoxidase dismutase 1, which protects from damage from the toxic free radical superoxide)
SOD1
Genetic ALS Etiology
-the mutated SOD1 ____ ("forms clumps in brain")
aggregates
Genetic ALS Etiology
2. mutation in ___, ___, ___ (control RNA transcription an splicing)
TARDBP, FUS, ANG
Genetic ALS Etiology
3. mutation in ____ (optineurin gene) that leads to apoptosis
OPTN
ALS Disease Progression-Early Stage
Astrocytes normally remove excess ___, but in ALS this system fails → leading to increased glutamate and Ca²⁺ influx → neuronal toxicity
glutamate
ALS Disease Progression-Early Stage
___ = glutamate transporter located on astrocytes
EAAT2
ALS Disease Progression-Early Stage
-Protein ___ (e.g., SOD1 mutations) leads to mitochondrial dysfunction and impaired axonal transport
aggregation
ALS Disease Progression-Symptomatic Stage
-___ factors are released from activated microglia/astrocytes
toxic
ALS Disease Progression-End Stage
-involves caspase-mediated apoptosis of motor neurons with microglial phagocytosis, leading to severe __ atrophy and respiratory failure.
muscle
____ (Rilutek, Exservan, Tiglutik) is a glutamate antagonist used in ALS
Riluzole
Riluzole
1. Main mechanism = Decreases ___ release by blocking voltage-gated ___ channels → reduces neuronal firing in glutamate pathways
glutamate, sodium
Riluzole
2. Increases ___ of glutamate
reuptake
Riluzole
3. Weakly blocks glutamate __
receptor
Riluzole is an oral drug that has limited BBB penetration due to P-gp efflux, whereas Edaravone is given ___, and therefore better reaches the CNS
IV
Edaravone
-MOA = ___ ___ ___ (antioxidant that neutralizes oxidative stress to neuron damage)
free radical scavenger
Tofersen is approved for ALS who have been identified to have mutant __
SOD1
Tofersen
-an antisense ___ (ASO) that targets the __ produced by mutated SOD1 genes
oligonucleotide, RNA
Tofersen
-blocks transcription of RNA to stop toxic SOD1 protein ___
production
Tofersen
-$150,000-200,000 per year, but covered under __ for qualified patients
medicare
Myasthenia Gravis is an autoimmune disease where antibodies target nicotinic acetylcholine receptors at the ___ junction
neuromuscular
Myasthenia Gravis (MG)
-ptosis (drooping of eyelids), diplopia (double vision), difficulty speaking/swallowing, extremity weakness
-severe disease that affects all muscles including ___
respiratory
Myasthenia Gravis involves antibodies against nicotinic __ receptors and/or ___
acetylcholine, MuSK
MuSK
-a receptor tyrosine kinase crucial for forming and maintaining ___ ___ (the connections between nerves and muscles)
neuromuscular junctions
MuSK
-activated by ___, a nerve derived protein, and plays a key role in clustering ___ receptors and other proteins in the synapse
agrin, acetylcholine
Myasthenia Gravis Treatment
-___ (Mestinon; acetylcholinesterase inhibitor)
-___ (Prednisone, Azathioprine)
pyridostigmine, immunosuppressants
Myasthenia Gravis Treatment
-surgery (___-which is removal of thymus)
thymectomy
Myasthenia Gravis Treatment
-____, (recycling of blood), IVIG, SCIG
plasmapheresis
3 Newer Drugs for MG = ___ (Vyvgart), ____ (Rystiggo), and ___ (Zilbrysq)
Efgartigimod, Rozanolixizumab-noli, Zilucoplan