Protein Structure
0.0(0)
Studied by 15 peopleCard Sorting
1/18
Earn XP
Description and Tags
Last updated 1:37 AM on 10/21/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
1
New cards
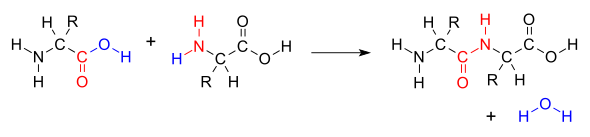
Peptide bond
amide bond that joins two amino acids
2
New cards
residue
amino acid units in a protein
3
New cards
Amino acid sequence
N-C (NH3+ to COO-)
order of AA
order of AA
4
New cards
Sanger protein sequencing
1. hydrolysis of polypeptide into small bits
2. separating bits
3. arranging bits to find the sequence
2. separating bits
3. arranging bits to find the sequence
5
New cards
Fragmentation
Trypsin - Lys, Arg (+) KR
Chymotrypsin -Phe, Tyr, Trp (aromatic) FYW
CNBr - Met (start) M
Chymotrypsin -Phe, Tyr, Trp (aromatic) FYW
CNBr - Met (start) M
6
New cards
Secondary structure
H-bonds between C=O & H=N
a-helix
-right handed spiral (1 turn is 3.6 residues)
b-sheet
a-helix
-right handed spiral (1 turn is 3.6 residues)
b-sheet
7
New cards
a-Helix
peptide is planner (cis or tans)
H bond between every 4th aa
disruptions:
-electrostatic repulsion
-bulky R groups
-small R groups (favour b)
-proline
H bond between every 4th aa
disruptions:
-electrostatic repulsion
-bulky R groups
-small R groups (favour b)
-proline
8
New cards
b-Conformation
peptide is planner (cis or trans)
H bond between adjacent strands
-antiparallel 7
-parallel 6.5
small R groups
H bond between adjacent strands
-antiparallel 7
-parallel 6.5
small R groups
9
New cards
Tertiary structure
globular form
-hydrophobic core (hydrophobic effect)
stablizing:
-disulfide
-electrostatic/ionic
-h bonds
-metal chelation
-hydrophobic core (hydrophobic effect)
stablizing:
-disulfide
-electrostatic/ionic
-h bonds
-metal chelation
10
New cards
Disulfide bonds
crosslink protein with covalent bonds
Cys
Cys
11
New cards
Metal chelation
salt bridge to stabilize charges
N(+) |||| Me++ |||| O-
N(+) |||| Me++ |||| O-
12
New cards
Quaternary structure
interaction of globular polypeptides
-same factors as 3
-same factors as 3
13
New cards
Protein folding
dependent on aa sequence
14
New cards
Protein denaturation
native protein converted to unfolded
1. heat
2, change in solvent
-few proteins can reverse spontaneously (need chaperons)
1. heat
2, change in solvent
-few proteins can reverse spontaneously (need chaperons)
15
New cards
Prostheic groups
attached non-amino acids
cofactor
-organic (coenzymes)
-inorganic
cofactor
-organic (coenzymes)
-inorganic
16
New cards
Ion exchange chromatography
protein purification
1. beads with sulfonic acid (anionic)
2. AA in buffer at 2
3. AA washed with higher pH
4. pH is pI of AA it will wash away
-salt can remove AA (compete for binding sites)
1. beads with sulfonic acid (anionic)
2. AA in buffer at 2
3. AA washed with higher pH
4. pH is pI of AA it will wash away
-salt can remove AA (compete for binding sites)
17
New cards
Size-Exclusion (Gel Sieving) chromatography
1. bead with pores
2. mixture of proteins
Big proteins -quick, can't go through pores
Small proteins -slow, go through beads
2. mixture of proteins
Big proteins -quick, can't go through pores
Small proteins -slow, go through beads
18
New cards
Affinity Chromatography
1. beads with ligand
2. mixture of proteins
3. hexokinase binds, others washed
4. ATP added (takes binging site) to unbind hexokinase
2. mixture of proteins
3. hexokinase binds, others washed
4. ATP added (takes binging site) to unbind hexokinase
19
New cards
SDS-PAGE
1. crosslinked polyacrylamide gel
2. detergent binds to proteins
3. electrical potential moves proteins
4. stained
2. detergent binds to proteins
3. electrical potential moves proteins
4. stained