Failures of the Body’s Defenses Genetic Immunodeficiencies
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
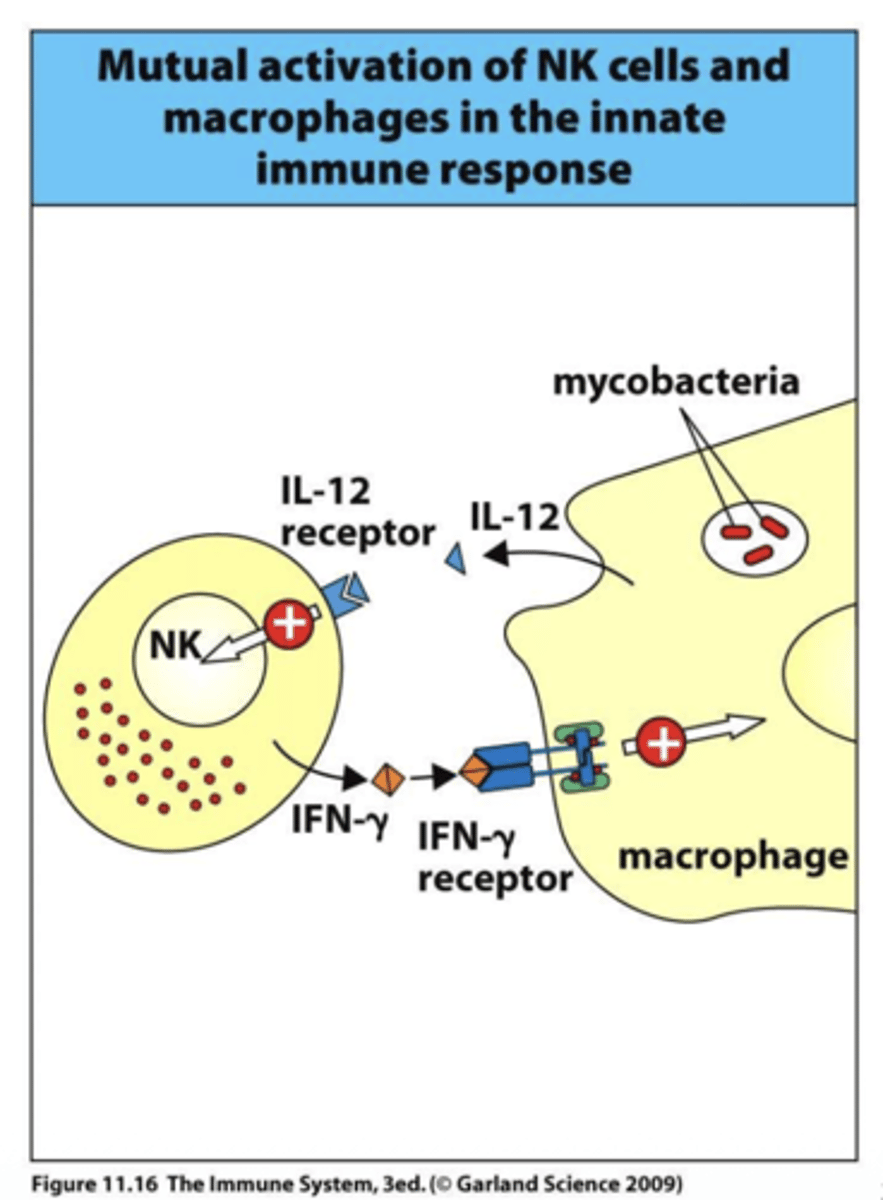
Q: What are the main functions of IFN-γ?
A: IFN-γ activates macrophages to enhance phagocytosis, stimulates the proteasome to process antigens, and interferes with viral replication to help control infections.
Q: What subunits make up the IFN-γ receptor?
A: The IFN-γ receptor is a dimer composed of IFN-γR1 and IFN-γR2, which work together to transmit signals inside the cell.
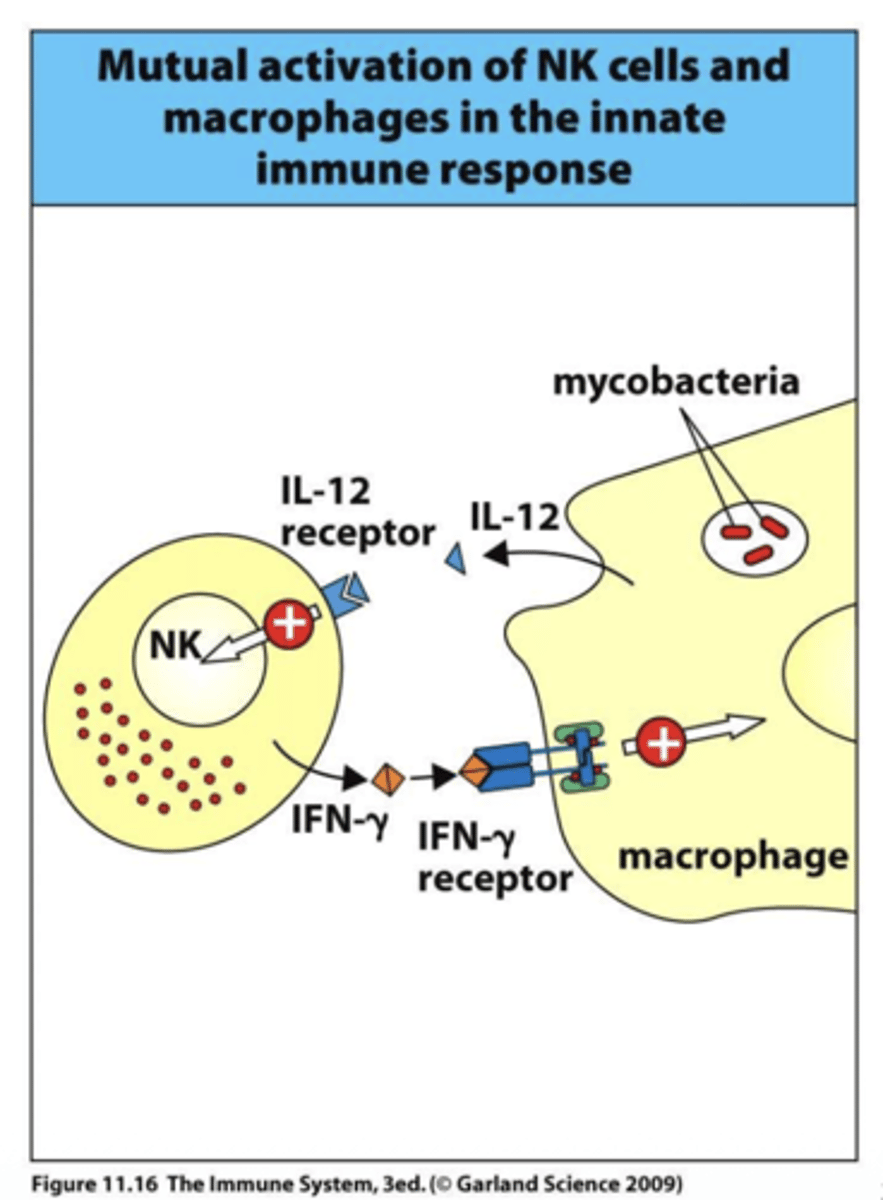
Q: Which kinases are associated with IFN-γR signaling?
A: IFN-γR associates with JAK-1 and JAK-2 kinases, which phosphorylate downstream targets to activate immune responses.
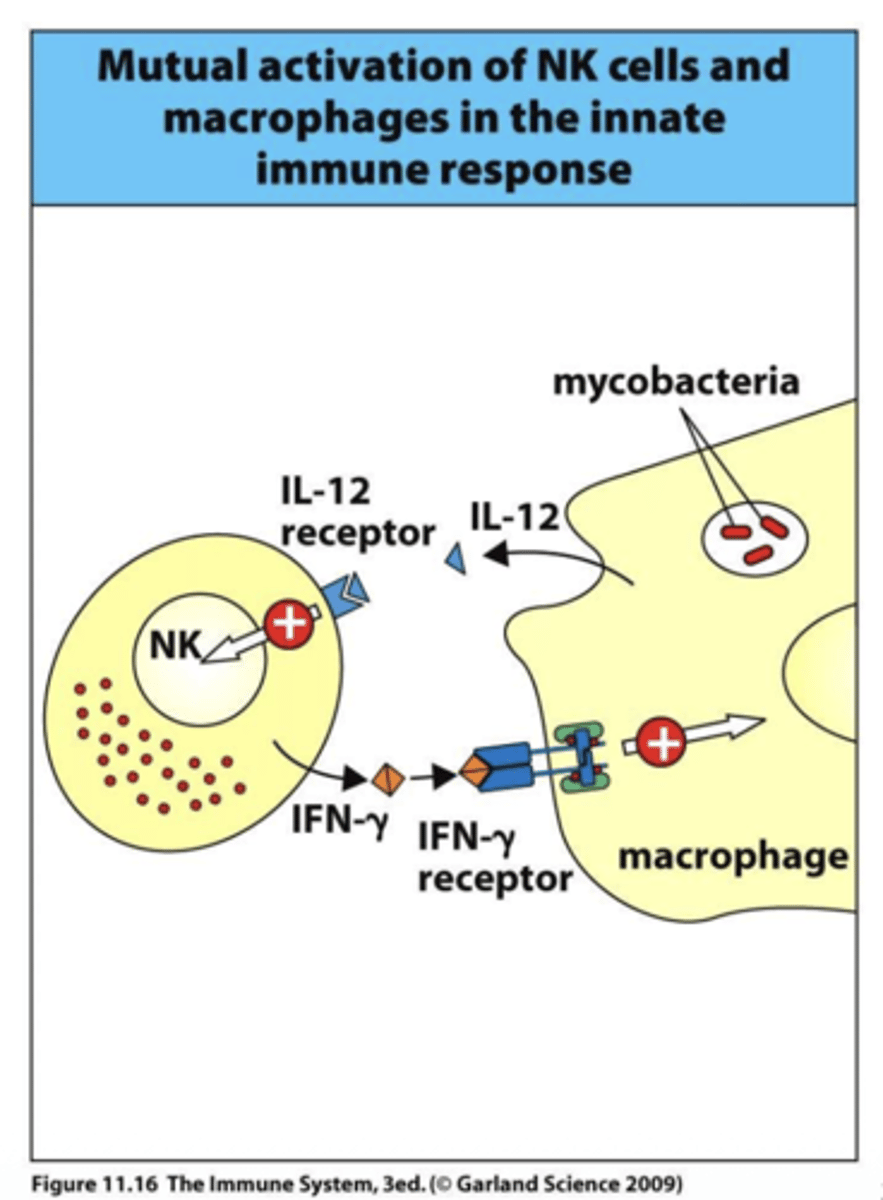
Q: How does IFN-γR deficiency affect macrophages?
A: Without IFN-γR, macrophages cannot be properly activated, reducing their ability to kill intracellular pathogens and present antigens effectively.
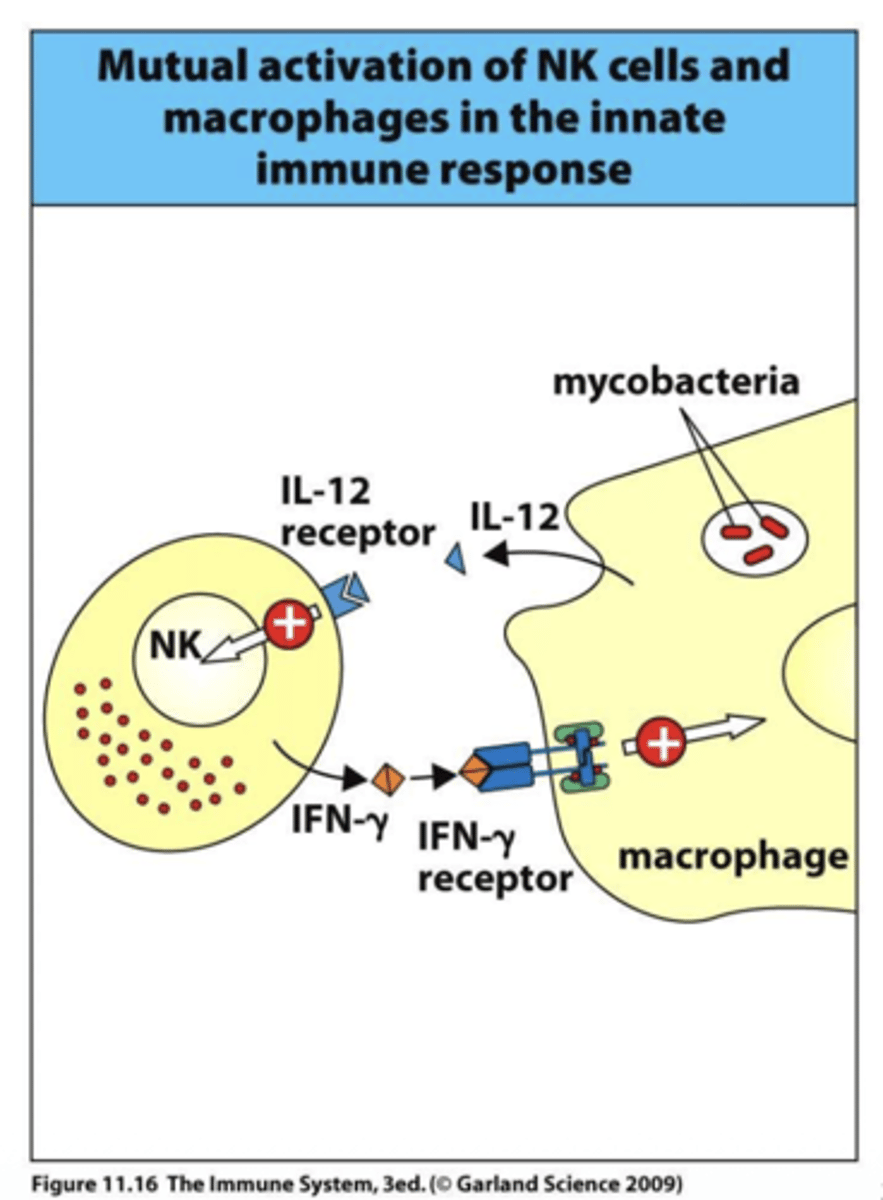
Q: Why is IFN-γ important for antiviral defense?
A: IFN-γ interferes with viral replication by activating antiviral pathways and enhancing antigen processing, helping the immune system control viral infections.
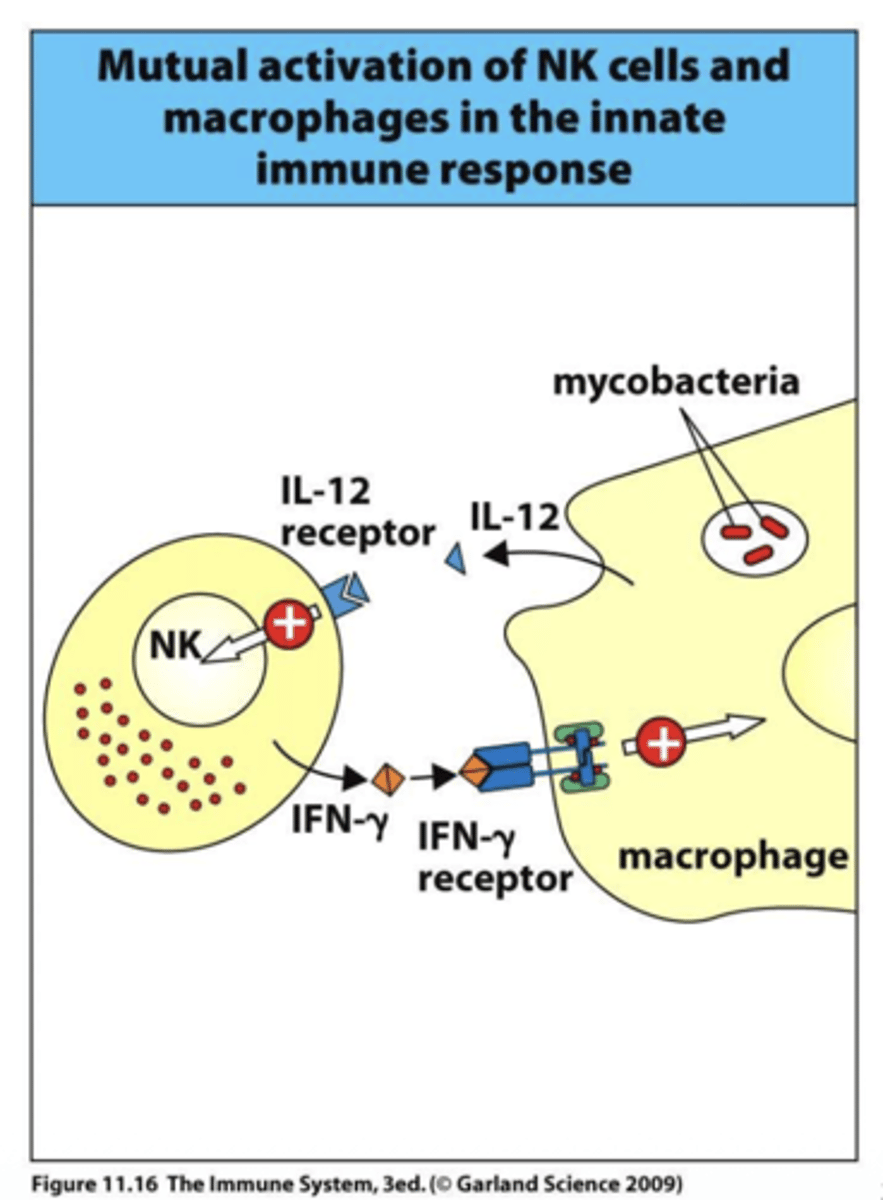
What is the effect of IL-12R deficiency on immune cells?
A: IL-12R deficiency mainly affects NK cells, reducing their ability to produce IFN-γ and respond to infections, particularly intracellular pathogens.
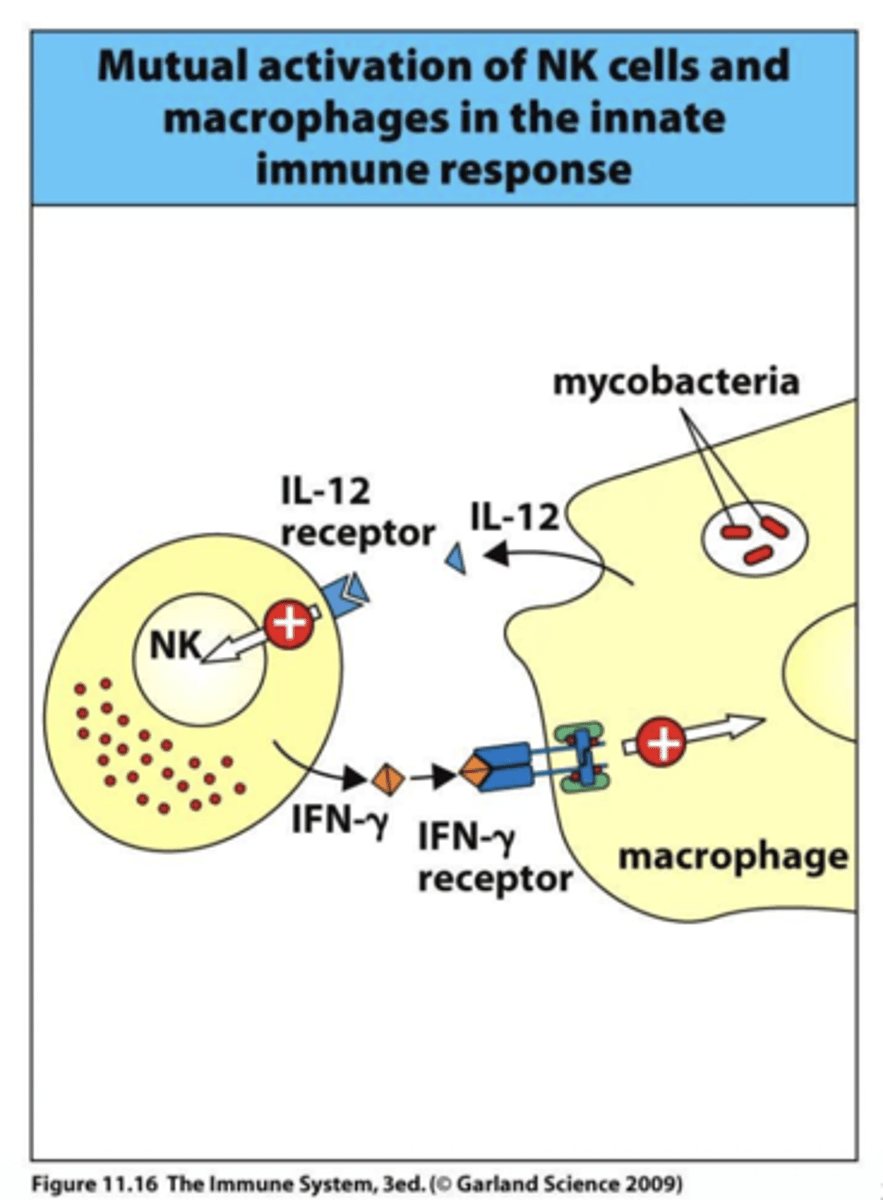
Q: How do IFN-γR and IL-12R deficiencies differ in immune dysfunction?
A: IFN-γR deficiency blocks macrophage activation and antiviral effects directly, while IL-12R deficiency mainly impairs NK cell activation and IFN-γ production, indirectly affecting immunity.
Q: What is the role of C3 in the complement system, and what happens if it is deficient?
A: C3 forms the alternative C3 convertase, the C5 convertase, and acts as an opsonin to mark pathogens for phagocytosis. Deficiency in C3 impairs these functions, leading to increased susceptibility to bacterial infections.
Q: What is the role of C5-C9 in the complement system, and what happens if these components are deficient?
A: C5 through C9 assemble to form the membrane attack complex (MAC), which creates pores in pathogen membranes to cause lysis. Deficiency in these components increases susceptibility to Neisseria infections.
Q: What is the main consequence of genetic defects affecting phagocytes?
A: Genetic defects in phagocytes impair their ability to engulf and kill pathogens, leading to persistent bacterial infections.
Q: What is a granuloma and how does it relate to phagocyte defects?
A: A granuloma is a collection of immune cells that forms to wall off pathogens the body cannot eliminate. In phagocyte defects, granulomas develop because bacteria persist and cannot be fully cleared.
its like a benign tumor, its an overgrowth of immune cells
its made of t cells and a pocked of macs, and the macs become giant cells by fusing and cant work correctly.
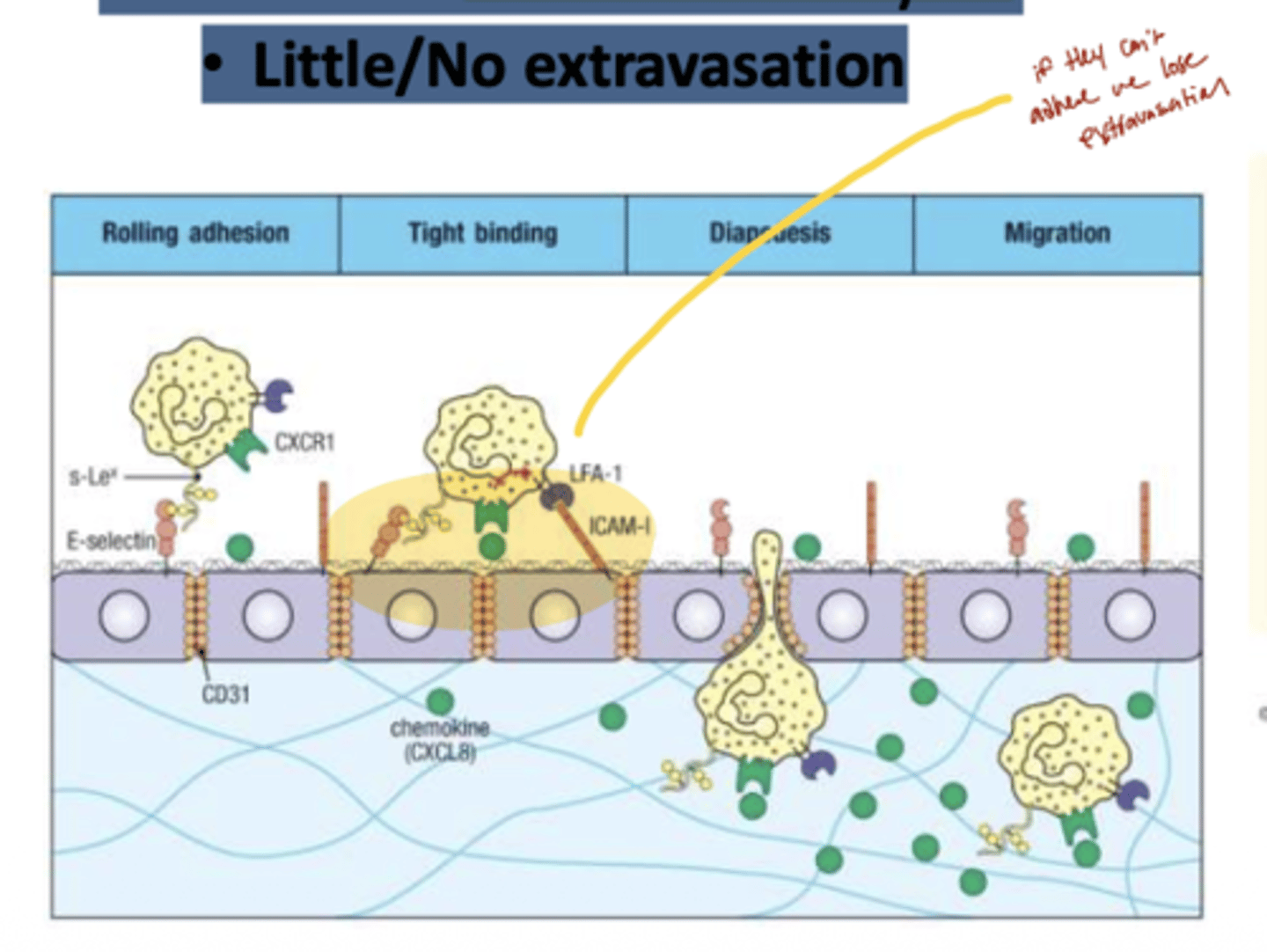
Q: What is the main defect in Leukocyte Adhesion Deficiency (LAD)?
A: LAD is caused by a failure of leukocytes to traffic properly, due to defects in adhesion molecules needed for migration.
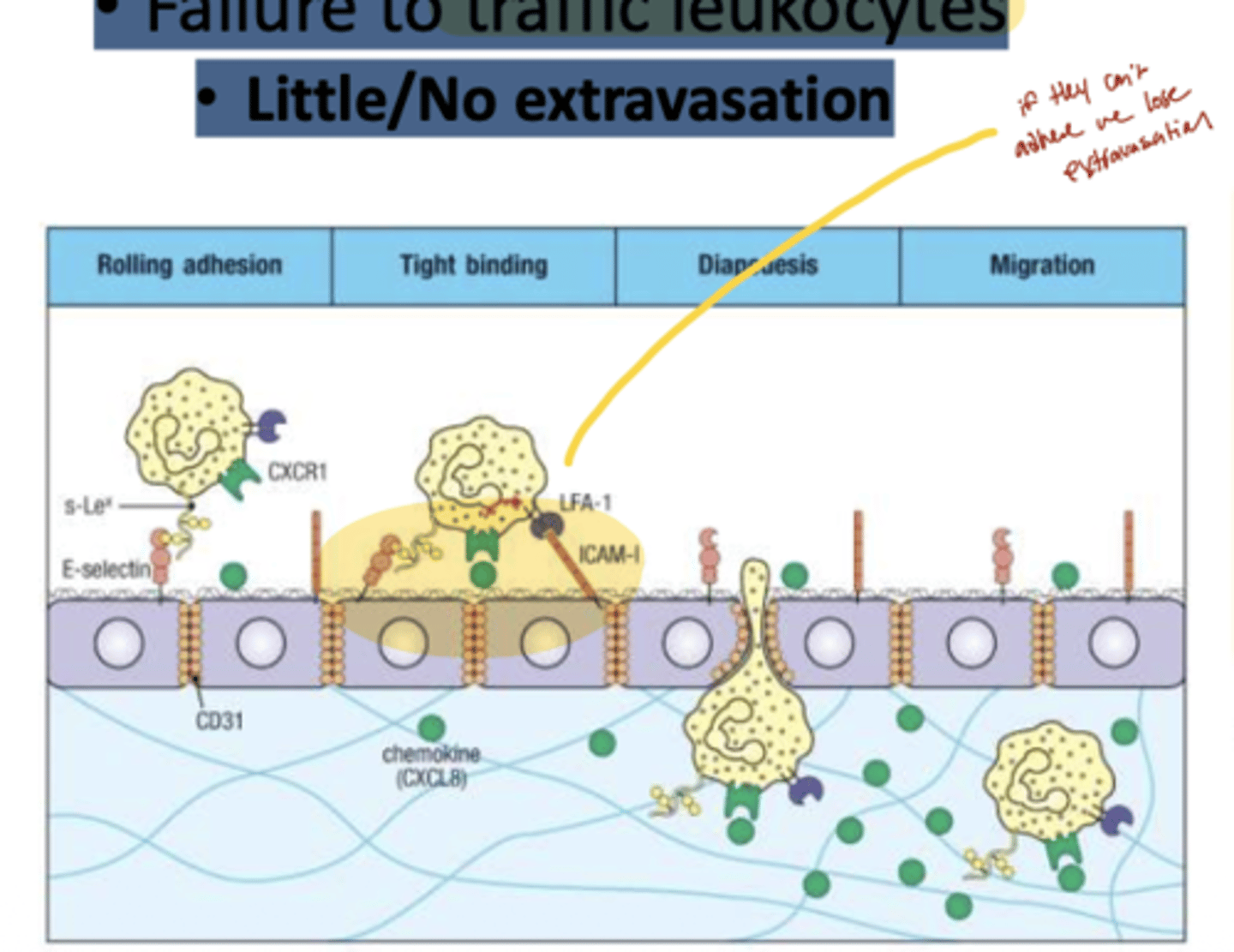
Q: How does LAD affect leukocyte extravasation?
A: In LAD, leukocytes show little or no extravasation(movement), meaning they cannot exit the bloodstream to reach sites of infection, leading to impaired immune response.
Q: What is the main defect in Severe Congenital Neutropenia?
A: SCN is caused by a lack of neutrophils because the bone marrow can’t fully make them from their precursor cells.
Q: When does Severe Congenital Neutropenia typically present?
It presents very early in life, often in infancy.
Q: What types of infections are common in Severe Congenital Neutropenia?
A: Patients experience frequent bacterial infections and have a high risk for fungal infections due to the absence of neutrophils.
Q: What is the main threat in antibody deficiency?
A: The major threat is encapsulated, pyogenic bacteria because antibodies are needed to recognize and target them for destruction.
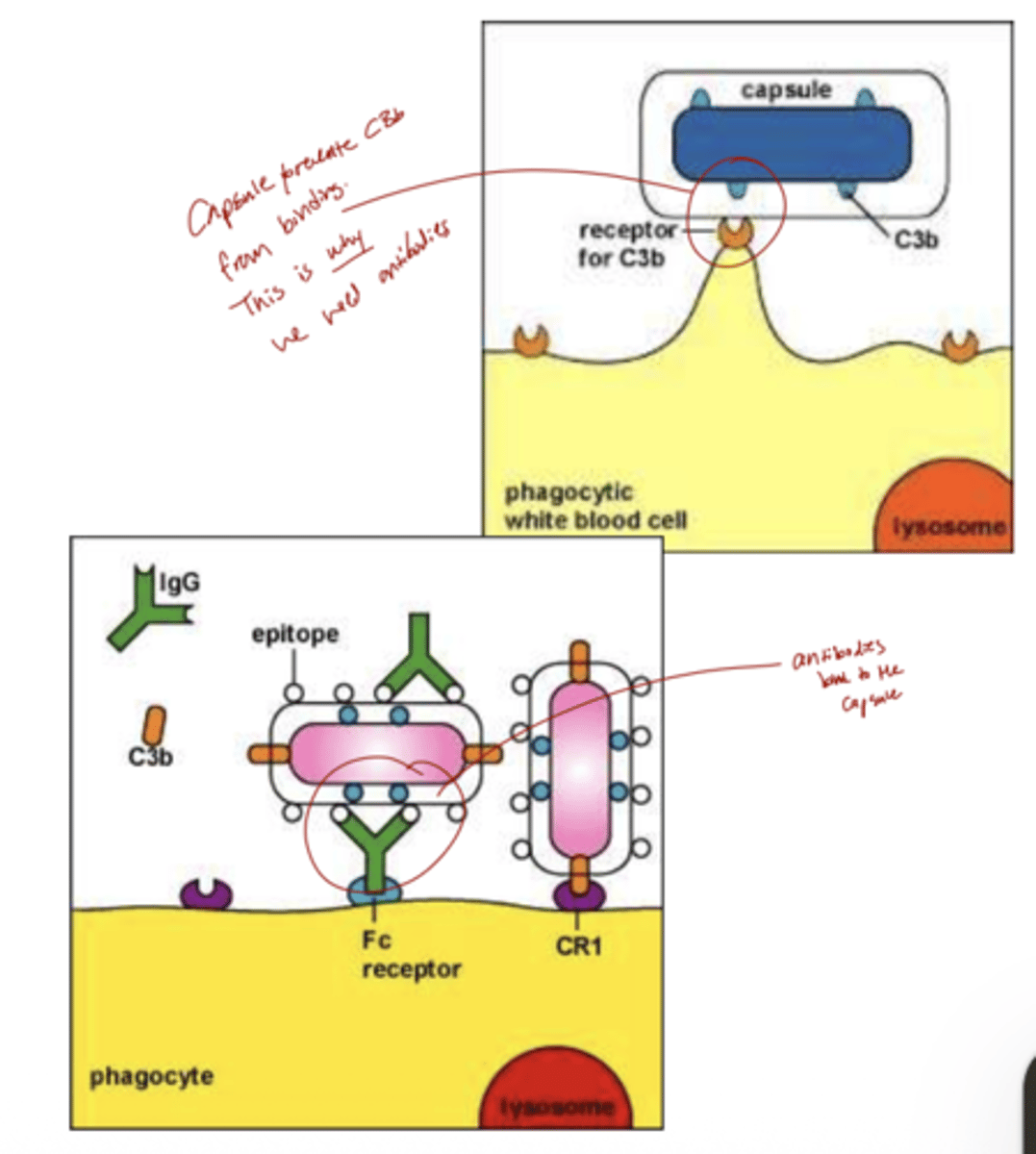
Q: Which bacteria are common causes of infection in antibody deficiency?
A: Common pathogens include Haemophilus influenzae, Streptococcus pneumoniae, Streptococcus pyogenes, and Staphylococcus aureus.
Q: Why are encapsulated bacteria especially dangerous in antibody deficiency?
A: Encapsulated bacteria are not easily recognized by phagocytes, allowing them to escape the innate immune system.
Q: How does the immune system normally defend against encapsulated bacteria?
A: Antibodies (through opsonization) and complement help phagocytes recognize and kill these bacteria, mounting an effective immune response.
Q: What is X-linked agammaglobulinemia (XLA) and why is it serious?
A: XLA is an antibody deficiency where patients have no functional antibodies, making them highly susceptible to infections; many children can die if all antibodies are missing.
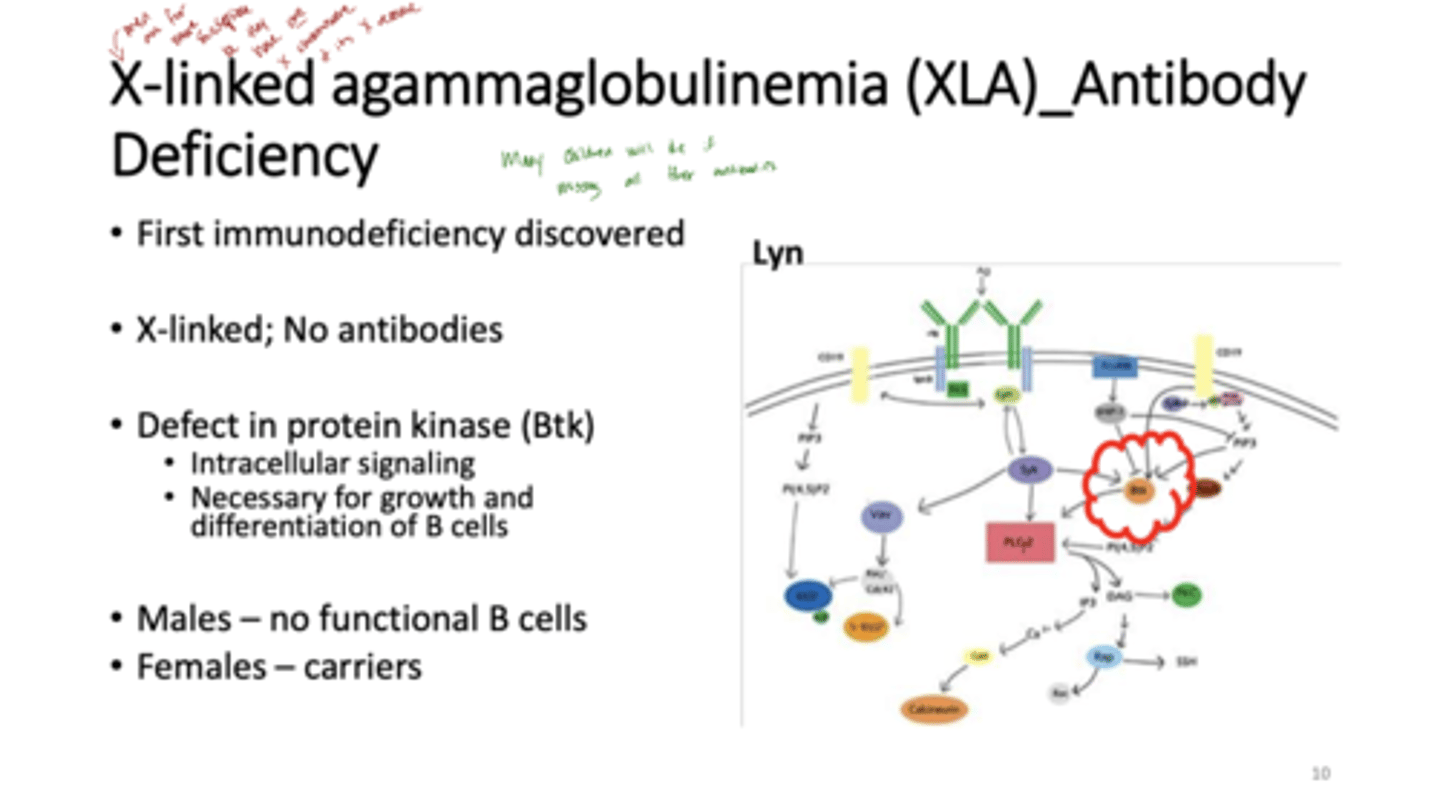
Q: What causes XLA?
A: It is caused by a mutation in the Btk (Bruton's tyrosine kinase) gene, which is essential for intracellular signaling needed for B cell growth and differentiation.
Q: How does XLA affect males and females differently?
A: Males have no functional B cells and therefore no antibodies, while females are usually carriers with normal B cell function.
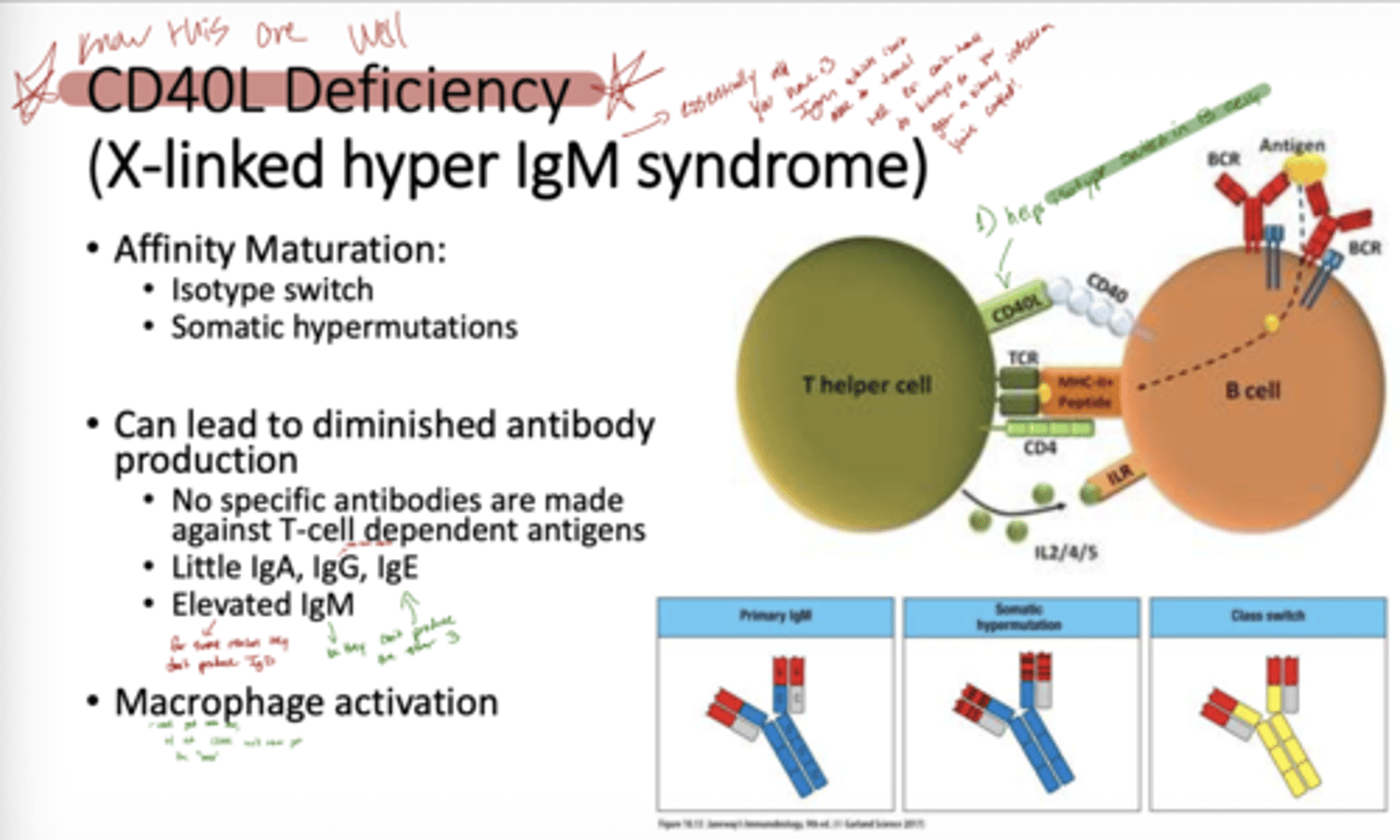
Q: What is CD40L deficiency and its other name?
A: CD40L deficiency, also called X-linked Hyper IgM Syndrome, is an antibody deficiency caused by a defect in the CD40 ligand on T cells.
Q: How does CD40L deficiency affect antibody responses?
A: It prevents isotype switching, affinity maturation, and somatic hypermutation, so little IgA, IgG, or IgE is made, while IgM is elevated. Patients cannot make antibodies against T-cell dependent antigens.
Q: How does CD40L deficiency affect macrophages?
A: Without CD40-CD40L signaling, macrophage activation is impaired, reducing deep tissue immune responses.
Q: What is the genetic cause of CD40L deficiency?
A: It is caused by a mutation in the CD40 ligand (CD40L) gene on T cells, which is X-linked, so primarily affects males.
Q: How does CD40L deficiency affect antibody production?
A: T cells cannot signal B cells via CD40-CD40L interaction, so B cells cannot undergo isotype switching, somatic hypermutation, or affinity maturation, resulting in very low IgG, IgA, and IgE, but normal or high IgM.
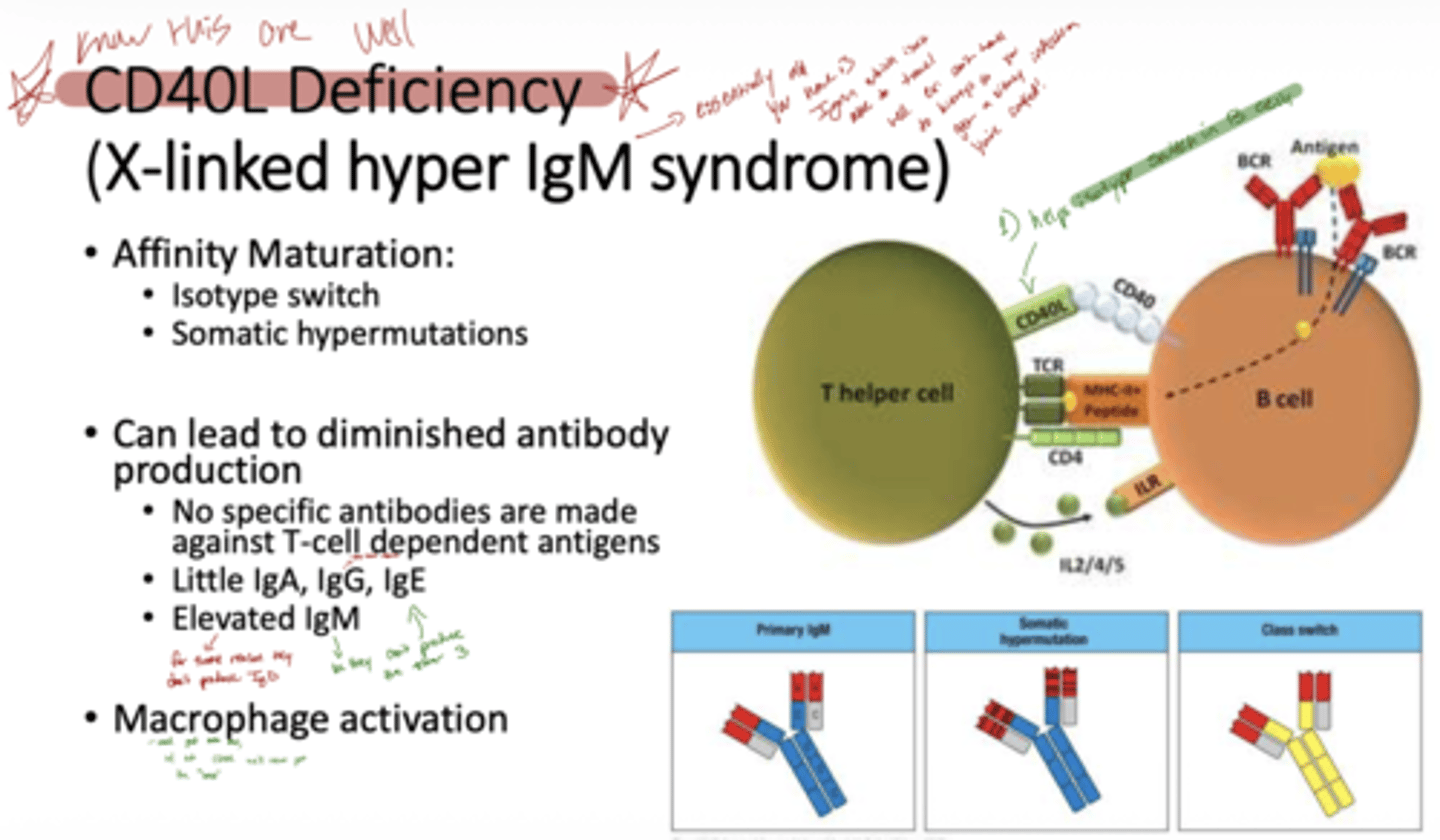
Q: Why are infections common in CD40L deficiency?
A: Patients cannot make antibodies against T-cell dependent antigens, leading to recurrent bacterial and opportunistic infections, especially in deep tissues.
essentially all they have is IgM which is not able to travel well.
Q: What causes X-linked Severe Combined Immunodeficiency (SCID)?
A: X-linked SCID is caused by mutations in cytokine receptors on lymphocytes, leading to no intracellular signaling and severely impaired T and sometimes NK cell function
Q: How is X-linked SCID treated?
A: Patients are usually kept in a pathogen-free environment and often require a bone marrow transplant to restore immune function.
Q: What is Wiskott-Aldrich Syndrome (WAS) and its main defect?
A: WAS is caused by a mutation in the WASP (Wiskott-Aldrich Syndrome Protein), leading to defects in protein trafficking, T cell function, and cytokine production.
Q: What are the main clinical features of Wiskott-Aldrich Syndrome?
A: Patients have impaired T cells, recurrent infections, eczema, and sometimes bleeding due to platelet defects.
Q: What causes autosomal SCID?
A: Autosomal SCID is caused by defects in gene rearrangement due to absence of RAG proteins, which are essential for developing functional T and B cell receptors.
if we have no somatic recomb we have no adaptive immunity (t or b cells) these people die early on
Q: How does the absence of RAG proteins affect the immune system?
A: Without RAG proteins, T and B cells cannot rearrange their receptors, so patients have no functional T or B cells, leading to severe immunodeficiency.
rag proteins recognize RSS between segments w/out RG theres no recognition
means what your born with is all you have for life, and even then it might not be function. children die very early on bc of this.
Q: What causes X-linked SCID and how does it affect immune function?
A: X-linked SCID is caused by mutations in cytokine receptors, which block intracellular signaling. This results in severely impaired T (and sometimes NK) cell function, leading to recurrent infections.
Q: What is a TAP defect and its consequence?
A: TAP (Transporter Associated with Antigen Processing) defects prevent peptides from entering the endoplasmic reticulum for MHC I presentation, causing loss of CD8 T cells and impaired cytotoxic T cell responses.

Q: What is Bare Lymphocyte Syndrome and how does it affect immunity?
A: Bare Lymphocyte Syndrome is caused by a lack of HLA class II expression, preventing CD4 T cell development and compromising nearly all aspects of adaptive immunity.
Q: Why must donor bone marrow cells be recognized by the recipient’s immune system?
A: Donor cells, including hematopoietic cells, must be recognized by the recipient’s T cells; otherwise, the transplant can fail or be rejected.
Q: What can happen if the recipient’s immune system attacks the transplant?
A: Dendritic cells and T cells may attack the transplanted bone marrow, causing graft rejection or graft-versus-host disease (GVHD).
Q: Which immunodeficiencies can be treated with bone marrow transplant?
A: Many immunodeficiencies that originate in hematopoietic cells, such as SCID or WAS, can be corrected by transplanting healthy hematopoietic cells.
Q: Why are immunosuppressive drugs needed after a bone marrow transplant?
A: Immunosuppressive drugs help prevent rejection of the donor marrow and reduce the risk of graft-versus-host disease.
What are the 4 types of hypersensitivity reactions?
A:
Type I (Immediate): IgE, only allergies (mast cells, basophils, eosinophils)
Type II (Cytotoxic): IgG, allergies/aids (type of deaminsase, not actually aids diseases),
-IgG binds to a cell
Type III (Immune Complex): -
-uses IgG
-small protein-protein interactions which gets lodged in blood vessels
Type IV (Delayed): Anything with a t cell, even if it also includes any of the other 3 types its considered type 4 if it has t cells