Intermolecular forces
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What is the overall effect of the frequent changing dipoles?
The temporary dipoles are attracted to each other
What is a prime example of permanent dipole-dipole forces?
Any hydrogen halide, eg. hydrogen chloride
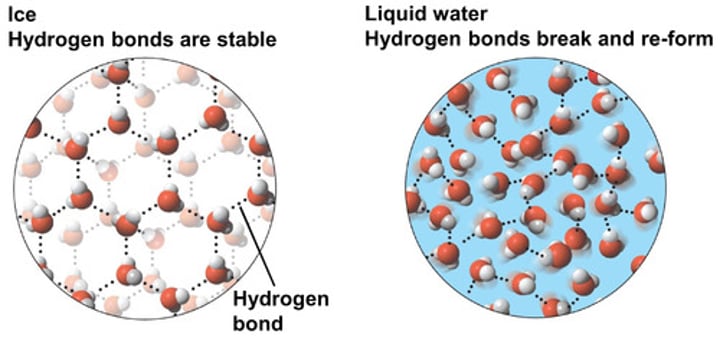
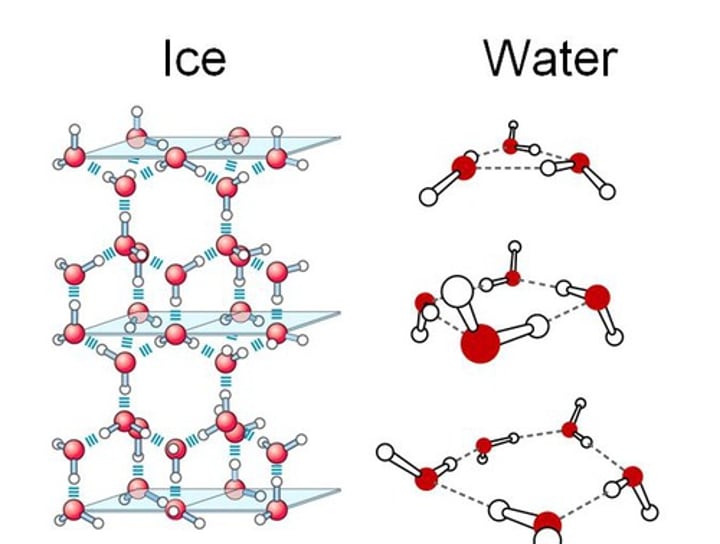
Describe the structure of ice. Why is it formed this way?
A regular 3D lattice that resembles diamond. This is formed because the hydrogen bonds hold the molecules in fixed positions
What are the three types of intermolecular forces?
Van der Waals forces, permanent dipole-dipole forces and hydrogen bonding
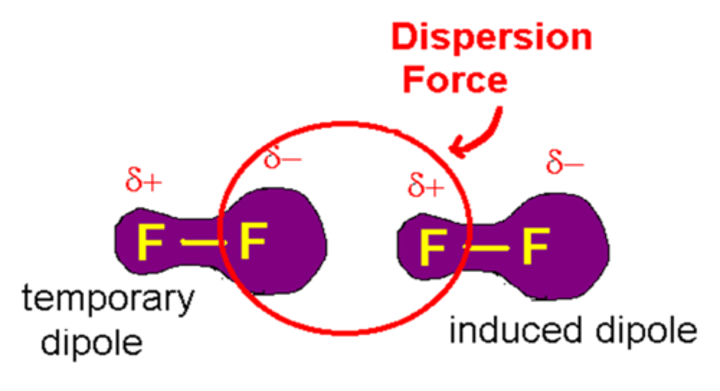
What are van der Waals forces also called? (2)
Induced dipole-dipole forces or London dispersion forces
Strength of intermolecular forces (weakest to strongest)

Do van der Waals forces occur in all molecules?
Yes
Are the electrons in molecules static or constantly moving?
Constantly moving
Is the electron density of a same-element molecule, eg. I₂, always equal?
No, because the electrons in electron clouds are constantly moving the electron density of I² can become uneven as more electrons are on one side
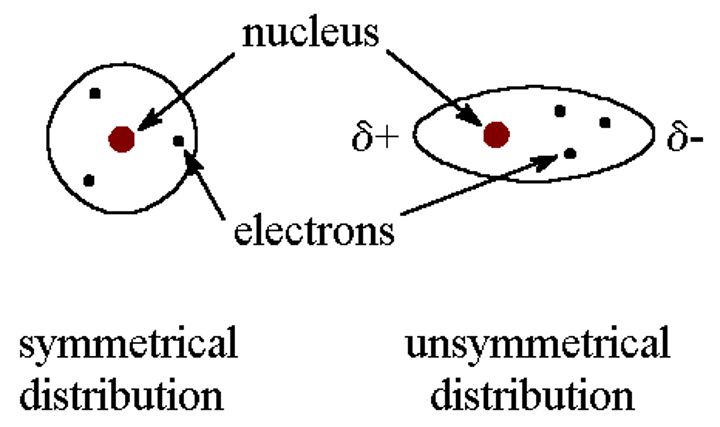
What is created by the uneven electron density?
An temporary dipole
What is a dipole?
A difference in charge between two atoms caused by a shift in electron density in a bond
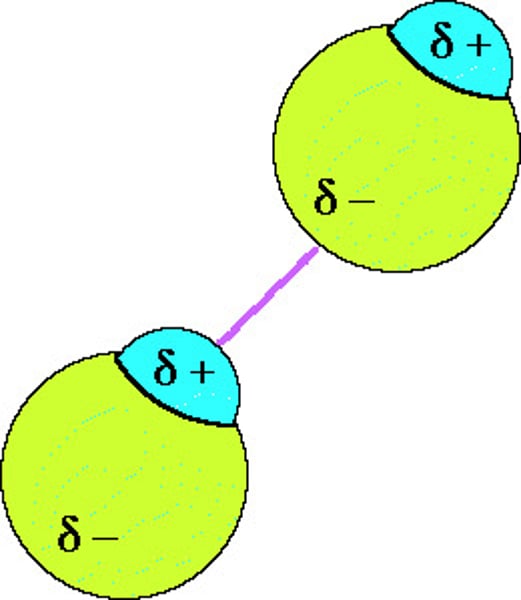
In an instance where there are lots of molecules, how do the temporary dipoles behave?
There is attraction between the positive and negative poles of the atoms which then induces a temporary dipole in another atom
What is the strength of van der Waals forces dependent on? (2)
The size of the molecule - the number of electrons present.
Molecule shapes
Why do larger molecules have stronger van der Waals forces?
Because they have larger electron clouds and so have larger fluctuations in electron density, leading to larger temporary dipoles
How do van der Waals forces change as you go down the group of noble gases? How do the boiling points change and why?
Forces increase as the number of electrons increase going down. Boiling points also increase as more energy is required to overcome the van der Waals forces

Why are dipole-dipole forces called permanent?
The delta negative and delta positive dipoles are always present
Where are the electrons going to lie closer to in hydrogen chloride?
The chlorine atom
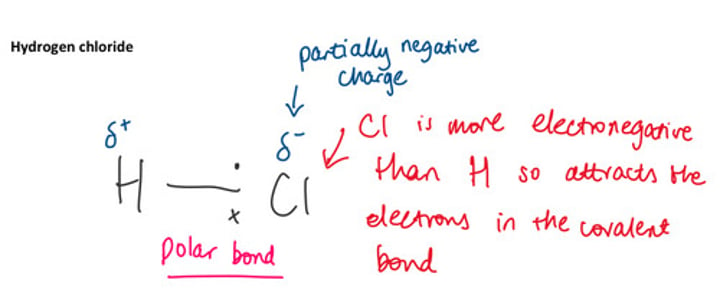
Will the partial charges ever switch in hydrogen chloride?
No because of chlorine is far more electronegative so the electrons will always lie closer
Permanent dipole-dipole forces
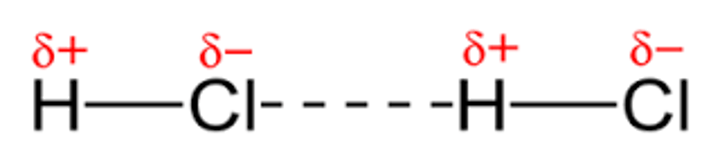
What type of molecules are permanent dipole-dipole interactions possible in?
Polar molecules - molecules that contain a permanent dipole
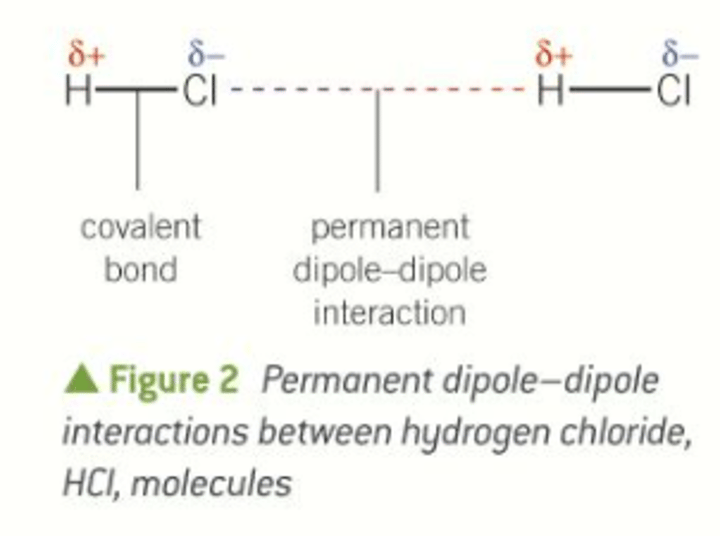
How can permanent dipole-dipole forces be overcome?
Through supplying the molecules with enough energy - melting/boiling points
Where are hydrogen bonds found?
When a hydrogen atom is bonded to either oxygen, nitrogen or fluorine
Hydrogen bonds in water
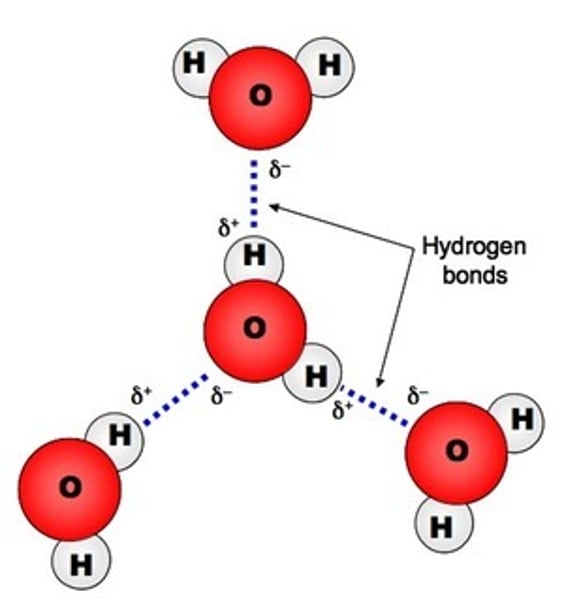
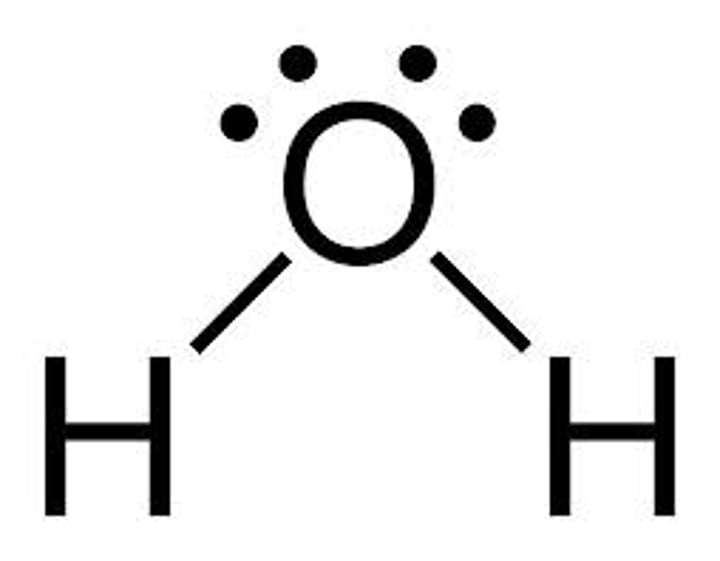
What does oxygen, nitrogen and fluorine atoms all have when bonded to hydrogen?
Lone pairs of electrons
Lone pairs on water molecules
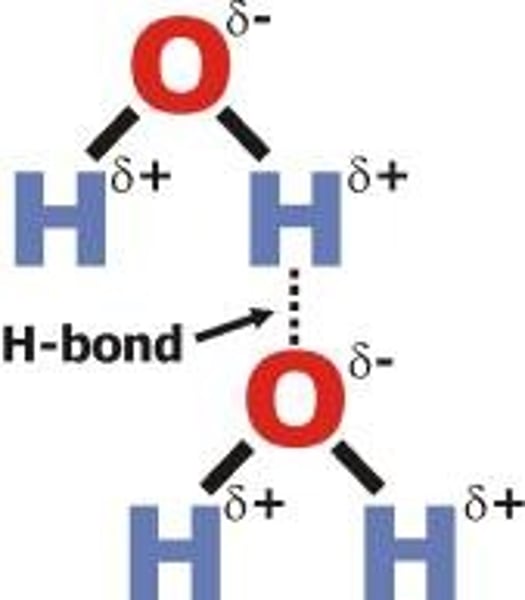
Which is more electronegative, oxygen or hydrogen?
Oxygen
What do the lone pairs on oxygen create in water molecules?
The delta negative region
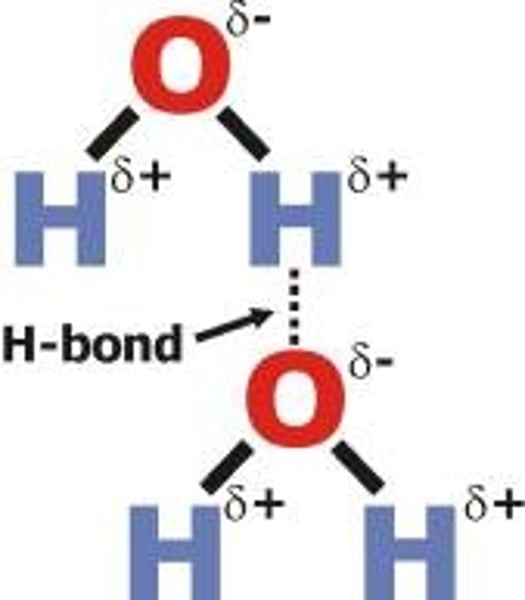
What does hydrogen develop when it bonds to a very electronegative element such as oxygen?
A strong δ+ charge
Hydrogen bonding is a very strong type of what?
Permanent dipole-dipole interaction
How are hydrogen bonds usually represented?
By dashes
Which is less dense - ice or liquid water?
Ice - that is why it floats!
Why is ice less dense than water?
The molecules in ice are slightly less closely packed than in liquid water, causing the volume to expand as there are more hydrogen bonds